Clear Sky Science · en
Whole-genome sequencing analysis of anthropometric traits in 672,976 individuals reveals convergence between rare and common genetic associations
How DNA Shapes Everyday Body Differences
Why are some people tall and lean while others are shorter or carry weight around the middle? We know that family genes matter a lot, but scientists have long struggled to find all the DNA changes that add up to traits like height and body shape. This study uses a powerful technique called whole-genome sequencing in nearly 700,000 people to search every corner of the genome—both well-known and rarely explored regions—to see how tiny differences in DNA, whether common or extremely rare, work together to shape our bodies.
Looking Across Nearly 700,000 Genomes
The researchers analysed complete DNA sequences from 447,000 participants in the UK Biobank and 225,000 people in the US All of Us program. Instead of only scanning common DNA changes or just the parts of genes that make proteins, they examined the entire genome, including the vast “non-coding” stretches that help control when and where genes are switched on. They focused on three basic body measurements: height, body mass index (BMI, a rough measure of weight for a given height), and waist–hip ratio adjusted for BMI, which reflects where fat is stored on the body. Using advanced statistical tools, they tested both individual DNA changes and groups of rare changes within the same gene or regulatory region.
Rare DNA Changes with Big Effects
Compared with common variants, rare DNA changes are carried by very few people but can have surprisingly large effects. The team found 90 rare or low-frequency single DNA variants and 135 clusters of rare protein-altering variants that were linked to the three traits. Some had striking impacts: rare damaging variants in the UBR3 gene raised BMI by almost 3 units, and a rare change in a small RNA gene called MIRNA497 shifted height by nearly 4 centimeters per copy. They also discovered groups of rare non-coding variants—changes that do not alter proteins directly but influence how genes are regulated—that were strongly tied to body size and shape.
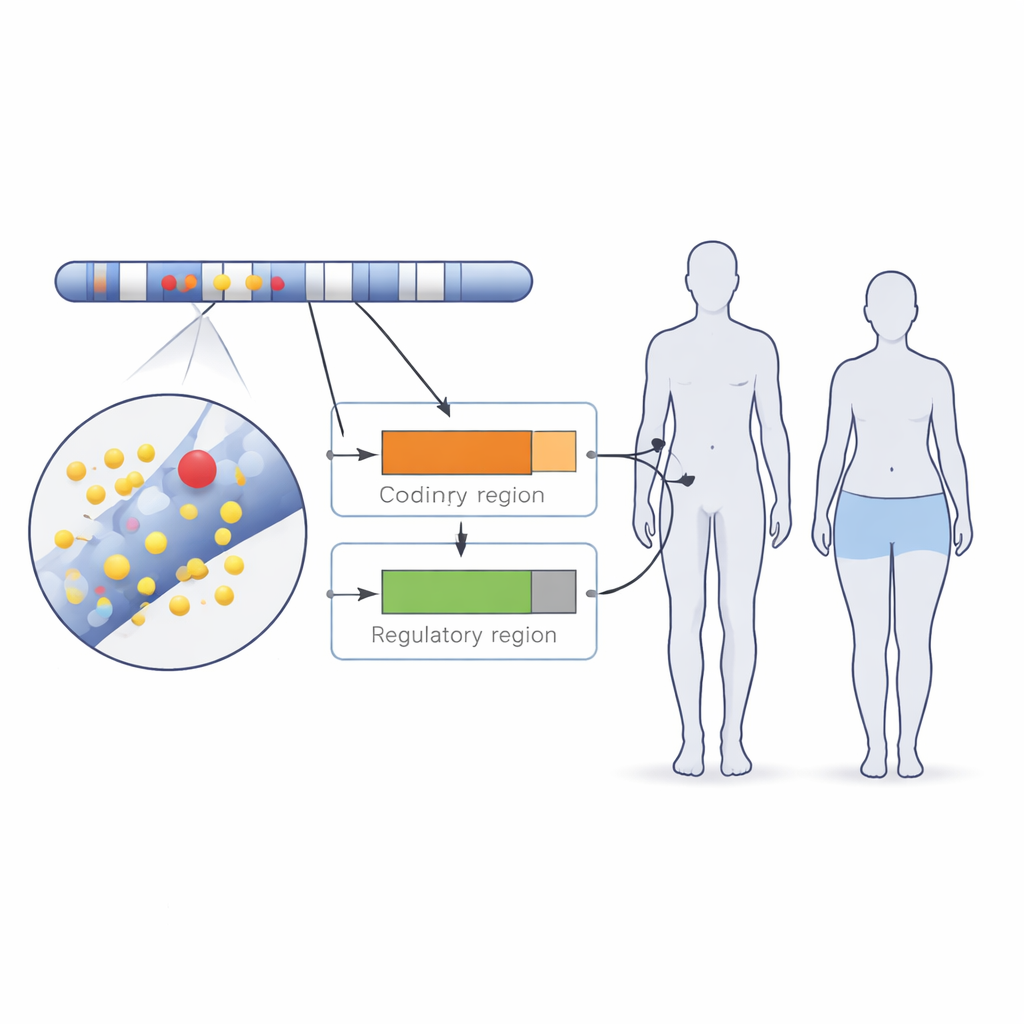
Hidden Control Switches Outside Genes
A key advance of whole-genome sequencing is the ability to find important control switches outside traditional genes. The study uncovered 51 significant clusters of rare variants in non-coding regions. One of the clearest examples involves FGF18, a gene important for bone growth. Here, rare changes in the 5′ untranslated region—a segment that helps control how much of the protein is made—altered height by as much as 6 centimeters, while protein-changing variants in the same gene showed no clear effect. This pattern suggests that, for some genes, fine-tuning their activity through regulatory DNA may be more compatible with healthy development than disrupting the protein itself.
Rare and Common Variants Point to the Same Genes
One of the strongest messages from the study is that rare and common genetic variants tend to cluster in the same genomic neighborhoods. For height in particular, about 97% of rare variant signals lay very close to locations already highlighted by large studies of common variants. Similar patterns appeared across different ancestry groups in the All of Us cohort, indicating that the same underlying genes and pathways shape body size worldwide. When the researchers estimated how much of each trait’s heritability (the portion explained by genetic differences) comes from various parts of the genome, they found that the contribution of rare variants is small compared with common ones—but heavily concentrated near known common-variant signals.
What This Means for Understanding Body Shape
For non-specialists, the take-home message is that the genes and pathways influencing height and body fat distribution are broadly the same whether the DNA changes involved are frequent and subtle or extremely rare and powerful. Whole-genome sequencing reveals both: it exposes rare variants with outsized effects and uncovers hidden regulatory switches that earlier methods missed. By showing that rare and common variants converge on the same regions of the genome—and that this holds across different ancestries—the study helps narrow the search for truly causal genes and mechanisms. In the long run, this sharper view of how DNA shapes our bodies could guide more precise risk prediction and point to biological pathways that might be targeted to improve health.
Citation: Hawkes, G., Wright, H.I.W., Beaumont, R.N. et al. Whole-genome sequencing analysis of anthropometric traits in 672,976 individuals reveals convergence between rare and common genetic associations. Nat Commun 17, 2432 (2026). https://doi.org/10.1038/s41467-026-69208-3
Keywords: whole-genome sequencing, rare genetic variants, human height, body fat distribution, genetic heritability