Clear Sky Science · en
Sequential-chain coupling over hierarchical click-sites enables highly selective urea electrosynthesis
Turning Waste into Plant Food
Urea fertilizer feeds crops around the world, but making it the traditional way burns huge amounts of fossil fuel and releases vast quantities of carbon dioxide. This study explores a different path: using electricity—from renewable sources when possible—to stitch together carbon and nitrogen from waste gases and polluted water into new urea molecules. By borrowing ideas from “click chemistry,” the authors design a smart catalyst that snaps these ingredients together cleanly and efficiently, pointing toward greener fertilizer and better use of industrial waste streams.

Why Rethink How We Make Urea?
Today’s urea production relies on century‑old, high‑temperature, high‑pressure processes that consume up to 2% of global energy and emit more than a ton of CO2 for every ton of urea produced. At the same time, power plants, factories, and wastewater streams release carbon dioxide and nitrate pollution that often go unused or cause environmental harm. Electrochemical urea synthesis offers a way to do two things at once: clean up CO2 and nitrate while making a valuable fertilizer at room temperature. The catch is that, at the tiny scales of molecules on a metal surface, carbon‑ and nitrogen‑containing fragments tend to go their own way, forming many by‑products instead of joining neatly into urea.
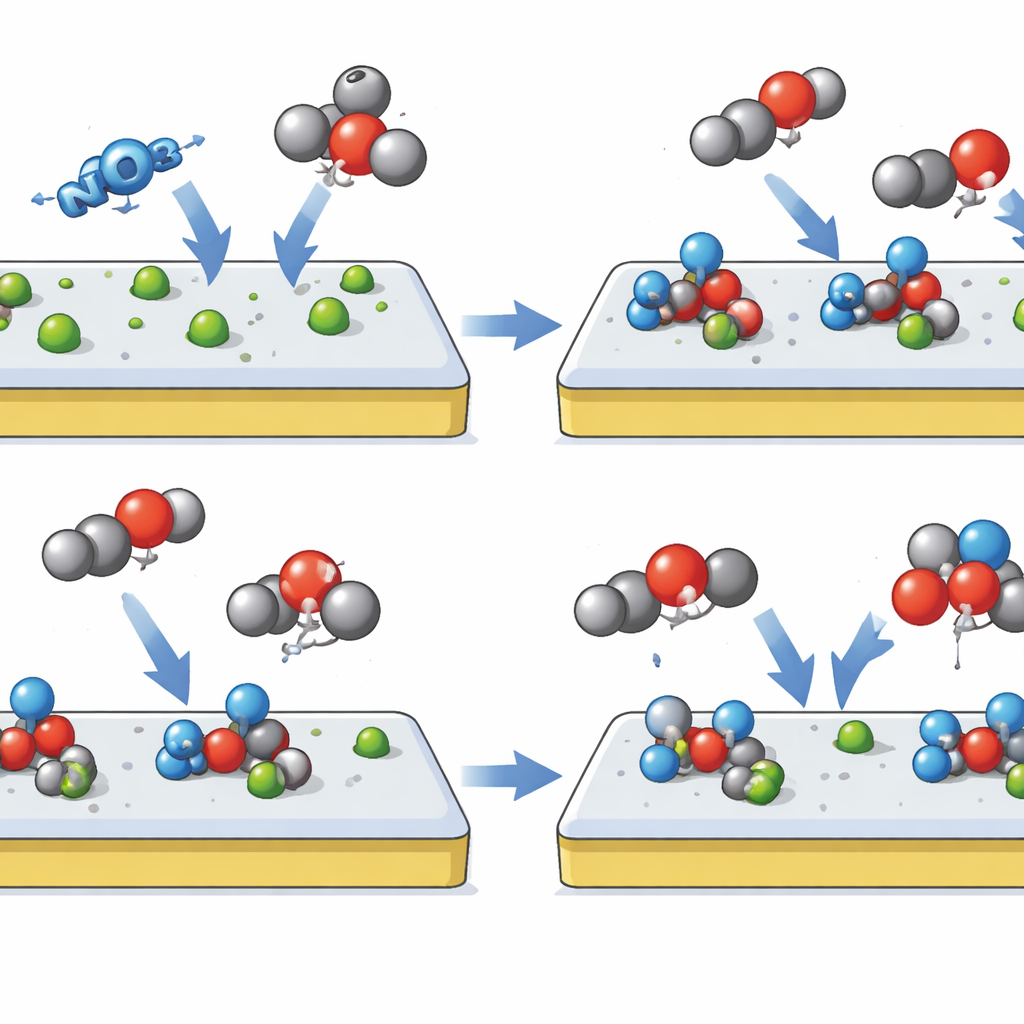
A Click-Inspired Molecular Assembly Line
The researchers take inspiration from click chemistry, a toolkit of reactions prized for snapping molecular pieces together quickly, selectively, and with minimal waste. They translate this idea to an electrode surface by building a “hierarchical click-site” catalyst based on indium oxide gently modified with the element selenium, called Se–InOx. The design creates two sequential stages on the same surface. In the first stage, the catalyst strongly prefers to grab nitrate from solution and convert it into a stable nitrite-like fragment, while deliberately refusing to bind incoming CO2. In the second stage, that anchored nitrogen fragment itself becomes a docking point that encourages CO2 to swing in and connect, forming a key carbon–nitrogen bonded intermediate that leads directly to urea.
How the Smart Surface Does Its Job
To make this behavior possible, the team subtly reshapes the electronic landscape of indium oxide by swapping a small fraction of its oxygen atoms for selenium. This tweak increases the local electron density and distorts the crystal lattice just enough that CO2 finds the surface unattractive, while nitrate and its nitrite‑like offspring bind more strongly. Computer simulations show that, on this tuned surface, forming the crucial carbon–nitrogen bond requires less energy and outcompetes side reactions that would otherwise turn nitrate into ammonia or CO2 into formic acid. Sophisticated in‑situ measurements—probing the surface with infrared light and magnetic resonance while the reaction runs—directly detect the expected intermediates, including the joined carbon–nitrogen species, confirming the step‑by‑step assembly line the designers envisioned.
Cleaner Output and Strong Performance
When tested in a flowing electrochemical cell, the Se–InOx catalyst delivers urea with both high speed and high purity. Under optimized conditions, it achieves a urea production rate of about 255 millimoles per hour per gram of catalyst and converts nearly 79% of the electrical charge into urea, with more than 85% of the nitrogen and essentially 100% of the carbon in the products ending up in urea rather than side chemicals. Competing pathways that generate ammonia, formic acid, or hydrogen are strongly suppressed. The catalyst maintains its structure and activity over repeated cycles and in a scaled‑up 5 × 5 cm cell that runs continuously for 20 hours, yielding over a gram of solid urea that passes nuclear magnetic resonance purity checks.
Costs, Climate Impact, and Future Promise
Beyond lab performance, the authors evaluate whether this approach could make economic and environmental sense at larger scale. Their analysis suggests that, if powered by inexpensive renewable electricity and further improved to modestly higher efficiency, electrochemical urea could approach or even undercut current market prices, especially when by‑products are also valued. A life‑cycle assessment shows that using low‑carbon electricity can cut greenhouse gas emissions per kilogram of urea below those of the conventional route. In simple terms, this work shows that carefully engineered “click-like” surfaces can choreograph how waste carbon and nitrogen meet, turning a pollution problem into a fertilizer solution and offering a blueprint for cleaner production of other complex chemicals.
Citation: Sun, Y., Tian, M., Wu, Q. et al. Sequential-chain coupling over hierarchical click-sites enables highly selective urea electrosynthesis. Nat Commun 17, 2388 (2026). https://doi.org/10.1038/s41467-026-69207-4
Keywords: electrochemical urea synthesis, carbon dioxide utilization, nitrate reduction, heterogeneous catalysis, green fertilizer