Clear Sky Science · en
Early diagnosis of pancreatic ductal adenocarcinoma by signal-enhanced lateral flow immunoassay: SELFI
Why catching this cancer early matters
Pancreatic cancer is one of the deadliest cancers largely because it is usually found too late for effective treatment. Early tumors often cause no clear symptoms, and the pancreas sits deep inside the body where it is hard to see with standard scans. This study introduces a new kind of rapid blood test, designed to spot pancreatic ductal adenocarcinoma (PDAC) at an earlier and more treatable stage, using a simple strip-based assay similar in convenience to a home pregnancy test.
A quiet disease that is hard to see
PDAC has a five‑year survival rate below 10%, in large part because current tools struggle to detect it early. Imaging methods like CT and MRI are expensive, can be invasive, and are not particularly sensitive for small, early tumors. Blood‑based tests look for molecules shed by tumors into the bloodstream. For PDAC, the only widely used marker is a sugar‑coated protein called CA19‑9. Today, CA19‑9 is usually measured by an enzyme‑linked immunosorbent assay (ELISA), a multi‑step laboratory procedure that requires specialized equipment and more than four hours, making it impractical as an easy screening tool.
Turning a strip test into a precision tool
Lateral flow immunoassays—the same basic format as many rapid at‑home tests—offer speed and simplicity, but their color bands can be too faint when the target molecule is present at very low levels. The authors set out to keep the friendliness of a strip test while matching or beating the sensitivity of ELISA. They created SELFI (signal‑enhanced lateral flow immunoassay), which still captures CA19‑9 on a test line but uses specially engineered nanoparticles to make the line much darker for the same amount of marker in blood. The goal is a 15‑minute test that can reliably pick up PDAC‑related CA19‑9 levels that older strip tests would miss.
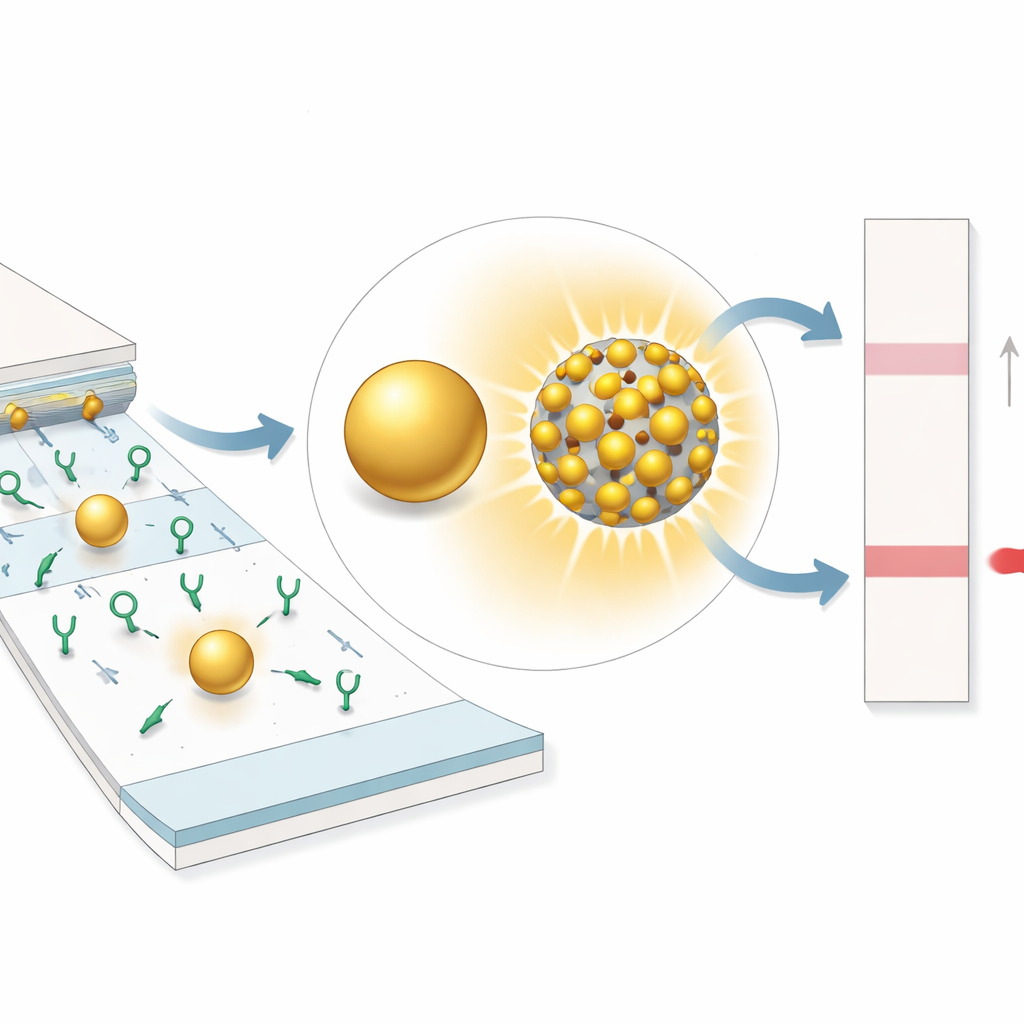
Tiny engineered particles that shine brighter
At the heart of SELFI are composite nanoparticles built from a silica sphere coated with many small gold particles separated by nanometer‑scale gaps. When light hits this structure, the electric field concentrates strongly in the gaps—"hotspots" that boost how much light the particle absorbs and scatters. Using computer simulations, the team showed that shrinking the gaps is more important than simply making the gold particles bigger: as the gaps narrow, the hotspots intensify, dramatically increasing the overall optical signal. They then fabricated a series of these particles with different gap sizes and confirmed experimentally that those with the tightest gaps produced the deepest color on test membranes, far outshining conventional single gold particles.
A faster and far more sensitive blood test
Using these bright nanoprobes, SELFI measures CA19‑9 in serum within 15 minutes, compared with roughly 285 minutes for ELISA. In controlled samples, SELFI could detect CA19‑9 at concentrations more than ten thousand times lower than a standard gold‑particle strip test and about 25 times lower than a commercial ELISA kit. The same nanoparticle design also improved detection of other cancer‑related proteins, suggesting the platform is adaptable beyond pancreatic cancer. When applied to blood samples from 150 people—healthy volunteers and patients with early and late‑stage PDAC—SELFI’s ability to distinguish early‑stage patients from healthy controls was slightly but significantly better than ELISA and much better than ordinary strip tests, as measured by standard diagnostic accuracy curves.
What this could mean for patients
This work shows that by carefully engineering how light interacts with nanoparticles on a simple strip, it is possible to create a rapid, low‑cost blood test that approaches laboratory‑grade sensitivity. SELFI does not introduce a new biomarker; instead it makes far better use of the long‑known CA19‑9 signal and could, in principle, be expanded to panels of markers for many diseases. If further studies confirm its performance in larger and more diverse populations, this technology could enable more routine screening and earlier diagnosis of pancreatic cancer, offering patients a better chance at timely surgery and effective treatment.
Citation: Jang, S., Shin, M., Han, J. et al. Early diagnosis of pancreatic ductal adenocarcinoma by signal-enhanced lateral flow immunoassay: SELFI. Nat Commun 17, 2768 (2026). https://doi.org/10.1038/s41467-026-69204-7
Keywords: pancreatic cancer, early detection, lateral flow test, nanoparticles, biomarkers