Clear Sky Science · en
Acid-base pair-mediated copolymerization of acid-sensitive epoxides and cyclic anhydride for synthesizing recyclable thermoplastics
Turning Everyday Plastics into Smarter Materials
From food containers to foam packaging, many of the plastics we rely on are tough to recycle because their molecular backbones are almost indestructible. This paper explores a new class of plastics designed to be strong in use but easier to un-build when we are done with them. By tweaking how certain small molecules are stitched together, and by using carefully chosen pairs of acids and bases as helpers, the researchers create sturdy materials that could one day stand in for common plastics like polystyrene—yet can be taken apart chemically and rebuilt.
Why Today’s Plastics Are Hard to Recycle
Mass-market plastics such as polyolefins are cheap, abundant, and mechanically robust because their chains are made from tightly bonded carbon atoms that resist breaking down. Unfortunately, this same strength makes them difficult to recycle back into their original ingredients. As a result, most mechanical recycling simply grinds and remelts old plastics into lower-value products. An appealing workaround is to build plastics from links that can, under the right conditions, be reversed. Polyesters, whose chains are held together by ester links, offer this possibility: under suitable chemical conditions, those links can be cut so the original building blocks reappear. The challenge is to make such polyesters both strong enough to rival common plastics and truly recyclable back to their starting molecules.
A New Way to Stitch Together Recyclable Chains
The study focuses on a versatile route called ring-opening copolymerization, in which two types of small ring-shaped molecules—epoxides and cyclic anhydrides—open up and connect alternately to form polyester chains. The anhydride, phthalic anhydride, is cheap and widely available, while the epoxides come from large-volume petrochemicals such as styrene, butadiene, and isobutylene. Earlier attempts to use these particular epoxides yielded only short, low-quality chains because the epoxides tend to rearrange into aldehydes in the presence of trace acids. Those aldehydes then act like chain stoppers or side-branch makers, capping growth and producing weak materials. The authors reasoned that if they could quietly remove these stray acids during the reaction, they could prevent the unwanted rearrangements and allow long chains to form.
How Acid–Base Pairs Tame a Runaway Side Reaction
To test this idea, the researchers paired bulky organic bases with gentle acids to form cooperative "acid–base pairs" that sit in the reaction mixture. The base component acts as a sponge for stray acidic species, including tiny amounts of phthalic acid and water-derived byproducts, which would otherwise trigger the troublesome rearrangement of epoxides into aldehydes. At the same time, the mild acid component helps activate the monomers so they still react quickly in the desired way. Through detailed control experiments, kinetic measurements, and analysis of chain ends, the team showed that this pairing interrupts a self-amplifying cycle in which acid creates aldehydes, aldehydes create more acids, and the reaction spirals into short, defective chains. With the cycle suppressed, the system instead channels most of the epoxides and anhydrides into long, well-behaved polyester chains.
Stronger Plastics with Built-In Second Lives
Using this strategy, the team produced several aromatic polyesters with molecular weights well above 100,000 units—high enough for demanding applications. These materials displayed tensile strengths above 50 megapascals and stiffness comparable to commercial polystyrene, meaning they resist stretching and bending under load. Yet they also process readily when melted and are more welcoming to water at their surfaces, which can be useful for coatings or blends. By subtly changing the side groups on the chains—phenyl, vinyl, or gem-dimethyl—the researchers tuned properties such as glass transition temperature, crystallinity, and how quickly chains can move past one another, linking molecular structure to performance in a systematic way.
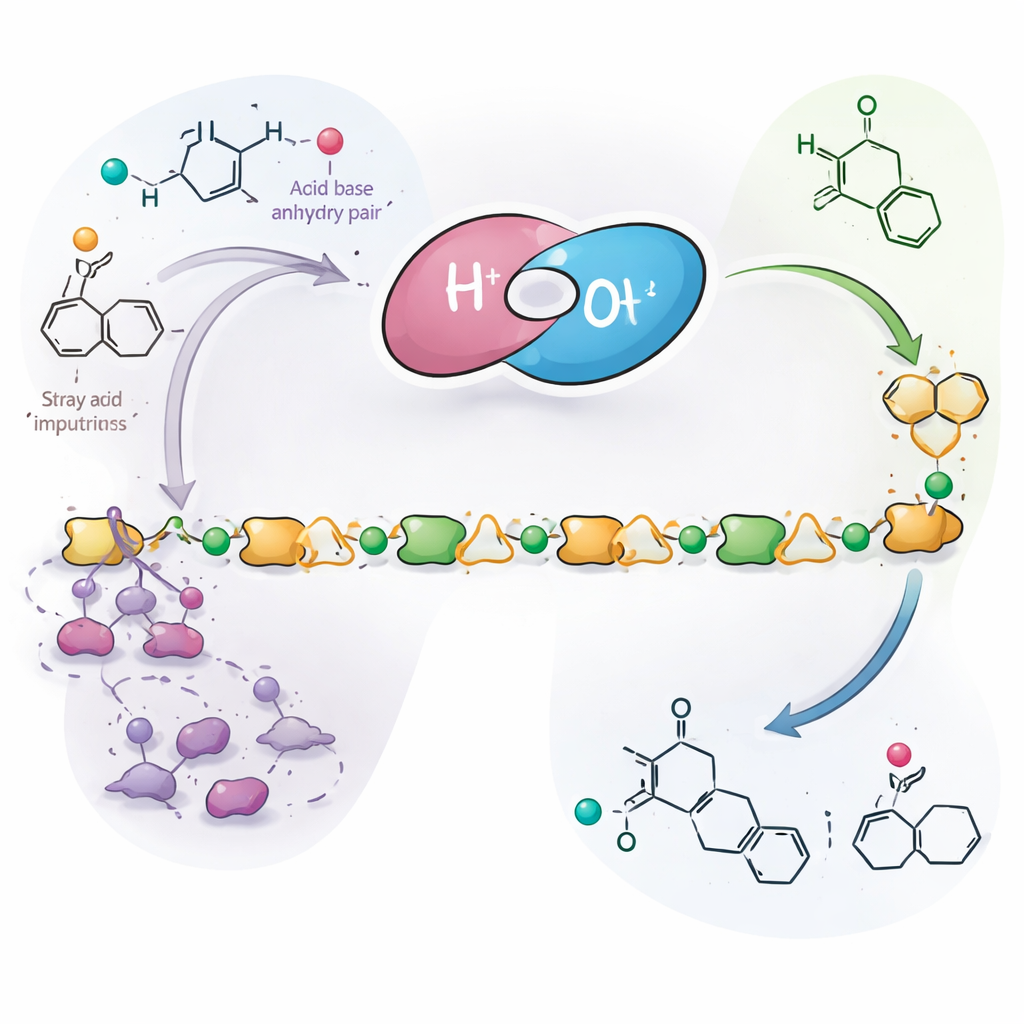
Unbuilding Plastics Back to Their Building Blocks
A key test of this approach is whether the new polyesters can truly be "unmade". The authors showed that, under relatively mild heating with simple acid catalysts such as sulfonic acids or zinc chloride, the chains can be driven to fall apart back into phthalic anhydride and the corresponding aldehydes. For one representative polyester, they recovered more than ninety percent of the anhydride and a large fraction of the aldehyde. Those small molecules are reactive starting points that can be used again to make fresh polymer or other products. In plain terms, the work demonstrates plastics that are strong enough to replace everyday materials like polystyrene but whose chemical zipper can be undone on demand, pointing toward a future in which plastics are designed from the outset for both performance and circularity.
Citation: Xie, Z., Yang, Z., Hu, C. et al. Acid-base pair-mediated copolymerization of acid-sensitive epoxides and cyclic anhydride for synthesizing recyclable thermoplastics. Nat Commun 17, 2668 (2026). https://doi.org/10.1038/s41467-026-69201-w
Keywords: recyclable plastics, polyesters, ring-opening copolymerization, acid-base catalysis, circular polymer economy