Clear Sky Science · en
Strain-stabilized interfacial polarization tunes work function over 1 eV in RuO2/TiO2 heterostructures
Why tiny shifts in atoms can matter for future devices
Metals are usually thought of as electrically uniform blocks where electric fields are quickly neutralized. This study overturns that simple picture for a common metal oxide, showing that when it is grown in ultrathin layers on another oxide, subtle shifts of atoms at their boundary create a built-in electric effect. That hidden effect lets the researchers change how tightly the metal holds on to its electrons by more than one electron volt—a huge swing for technologies that depend on moving charge efficiently, from sensors and catalysts to quantum devices.
A hidden electric effect at a metal boundary
In many modern electronics made from oxides, engineers deliberately build up electric charge at the meeting point between two different materials. This “interfacial polarization” has long been exploited in semiconductors and insulators to create conducting sheets of electrons or switchable electric dipoles. Metals, however, have been considered off-limits because their mobile electrons are expected to screen out any long-range electric fields. The authors challenge this view by examining a metallic oxide, ruthenium dioxide (RuO2), grown on titanium dioxide (TiO2) in a carefully controlled, atomically smooth stack. Their goal was to see whether a polar effect could survive at the buried interface and, if so, whether it would noticeably alter the metal’s electronic behavior.
Building atomically precise oxide sandwiches
To probe this question, the team used hybrid molecular beam epitaxy, a technique that lets them deposit materials one atomic layer at a time. They fabricated structures where a few-nanometer-thick RuO2 film is sandwiched between equally thin TiO2 layers on a TiO2 crystal substrate. Because the atomic spacings in RuO2 and TiO2 do not match perfectly, the RuO2 film is stretched and compressed in different directions—conditions known to trigger unusual phases in oxides. X-ray measurements and atomic force microscopy confirmed that the layers were extremely flat, crystalline, and well controlled in thickness, with the RuO2 film remaining strained below about 4 nanometers.
Seeing atoms shift and dipoles form
To find out what happens to atoms at the buried boundary, the researchers turned to a cutting-edge imaging method called multislice electron ptychography. This approach reconstructs the positions of both heavy metal atoms and lighter oxygen atoms with picometer precision. The images revealed that, near each RuO2/TiO2 interface, the metal ions shift slightly relative to their surrounding oxygen cages in a direction perpendicular to the layers. These tiny displacements point from the TiO2 into the RuO2 and are opposite at the top and bottom interfaces, forming mirror-symmetric regions with built-in electric dipoles. The effect extends a couple of atomic layers into the metallic RuO2, demonstrating that a polar distortion can coexist with good electrical conductivity in this rutile-structured system.
Turning buried polarization into a tunable surface barrier
The team then asked how this hidden polarization influences the surface where electrons actually leave or enter the metal. Using Kelvin probe force microscopy, they mapped the local surface potential of RuO2 films of different thicknesses and converted it into the surface work function—the energy barrier an electron must overcome to escape. Instead of changing smoothly with thickness, the work function rose sharply as the RuO2 layer approached about 4 nanometers, peaked more than 1 electron volt above the thin-film and substrate values, and then decreased again as the film grew thicker and relaxed its strain. This nonmonotonic behavior is inconsistent with simple band alignment between RuO2 and TiO2 alone. It points to an extra internal electric field created by the interfacial polarization, which boosts the surface barrier most strongly when the film is thin and fully strained.
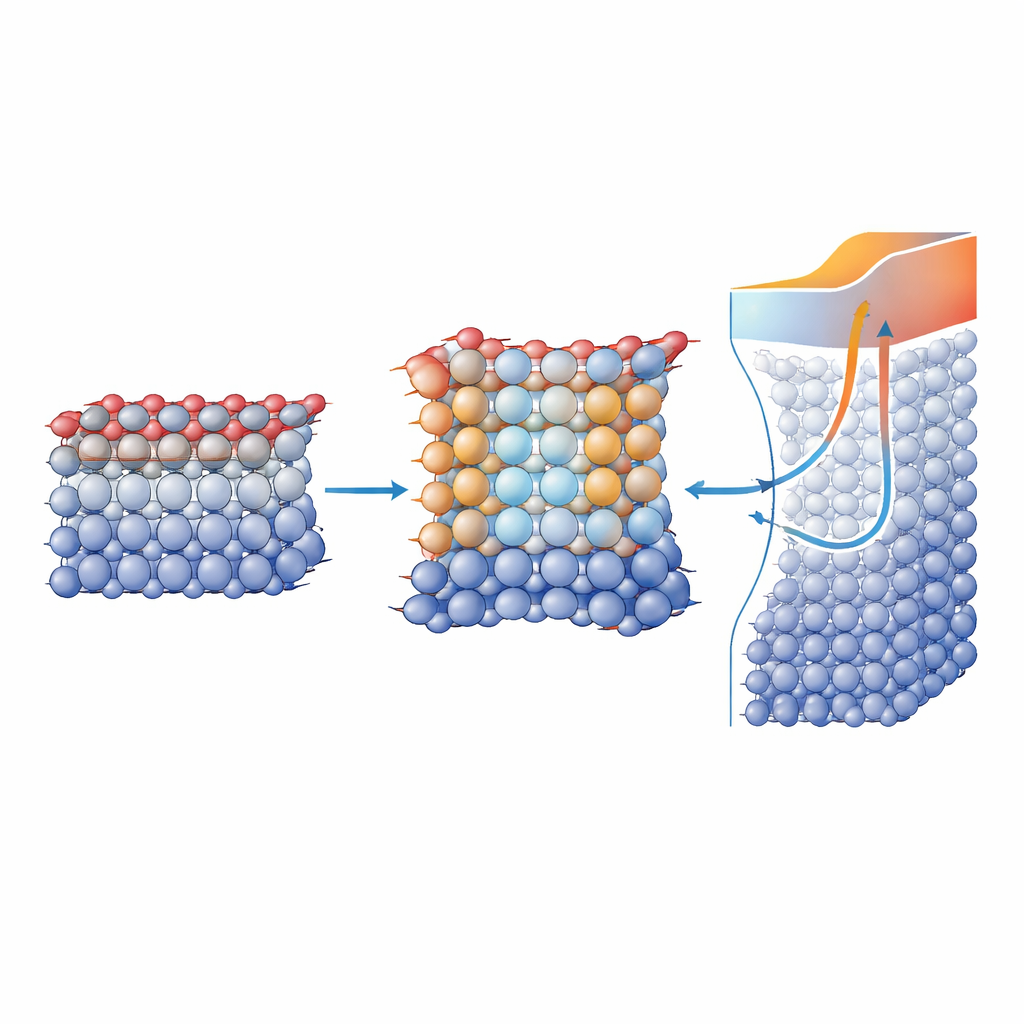
How charge transport reveals a special interfacial layer
Electrical measurements added another piece to the puzzle. By tracking the sheet conductivity, carrier density, and mobility of RuO2/TiO2 stacks as the RuO2 layer thickened, the authors showed that current flows through two channels in parallel: the bulk-like interior of the metal and a thinner region near the interface where conductivity is suppressed. Modeling these data indicates that this compensated interfacial layer is roughly 1.6 nanometers thick when RuO2 sits on TiO2, and shrinks to about 0.7 nanometers when RuO2 is capped symmetrically with TiO2 on both sides. Those thicknesses closely match the polarized region seen in the microscopy, and the reduced conductivity is consistent with other so-called polar metals. Together, the transport and imaging results show that the buried polarization is not just a structural curiosity—it directly shapes how electrons move.
What this means for future oxide technologies
By stabilizing interfacial polarization in a metallic oxide and linking it to a record-large, reversible change in surface work function, this work opens a new design strategy for oxide-based devices. Instead of relying on chemical treatments or adsorbed molecules to tune a metal’s electronic behavior, engineers could adjust layer thickness, strain, or stacking order to sculpt hidden polar regions that in turn control surface barriers and conductivity. Such strain-stabilized polar metals could offer tunable contacts for electronics, more active surfaces for catalysis, and new playgrounds for quantum phases that depend sensitively on electric fields at the nanoscale.
Citation: Jeong, S.G., Lin, B.Y.X., Jin, M. et al. Strain-stabilized interfacial polarization tunes work function over 1 eV in RuO2/TiO2 heterostructures. Nat Commun 17, 2516 (2026). https://doi.org/10.1038/s41467-026-69200-x
Keywords: polar metals, oxide heterostructures, work function tuning, interfacial polarization, RuO2 TiO2 thin films