Clear Sky Science · en
Molecular mechanisms of flotillin complexes in organizing membrane microdomains
Hidden rooms on the cell's surface
Every cell in your body is wrapped in a thin membrane that looks simple at first glance but is actually divided into countless tiny "neighborhoods." These neighborhoods help control how signals are received, how nutrients enter, and how waste leaves. This study uncovers how a pair of long‑mysterious proteins, called flotillins, build miniature sheltered rooms on the inner side of the membrane—structures that may influence processes as diverse as cell signaling, cargo transport, and even cancer spread. 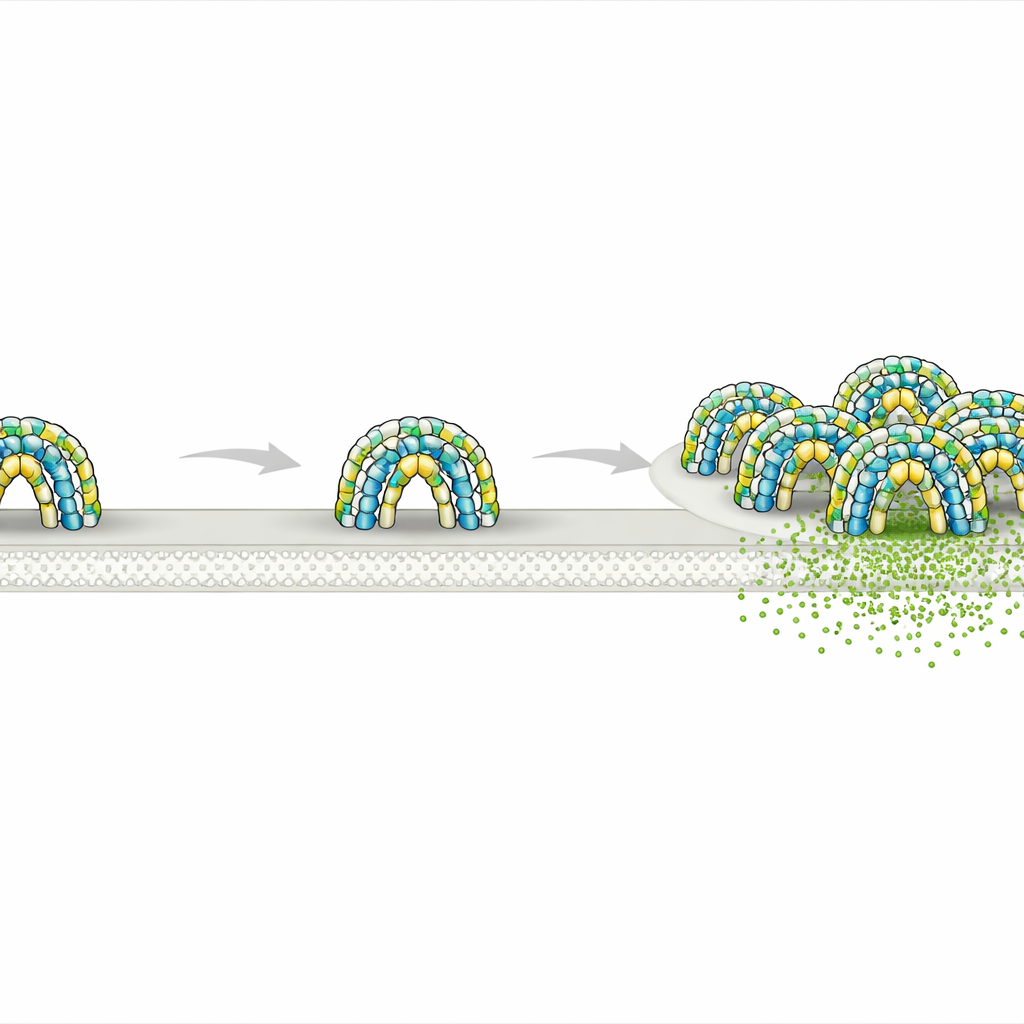
A closer look at tiny membrane neighborhoods
Biologists have long known that cell membranes are not uniform. Instead, lipids and proteins cluster into small patches that act like pop‑up workstations for specific tasks such as communication or cargo sorting. Flotillin‑1 and flotillin‑2 are two proteins that always seem to mark such patches, forming small bright spots on the cell surface and taking part in endocytosis—the process by which cells pull material inward—as well as signaling and protein trafficking. Yet nobody knew what shape flotillin structures actually take, or how they carve out their own territory on the membrane.
Domes that fence off circles of membrane
Using high‑resolution cryo‑electron microscopy on purified human proteins, the researchers discovered that flotillin‑1 and flotillin‑2 assemble into a remarkably large complex made of 44 protein copies, alternating between the two types. Together they form a rigid dome sitting on the inner face of the membrane. The base of this dome is built from membrane‑anchoring segments rich in greasy amino acids and lipid attachments that sink partway into the membrane. Above that, long helical stalks form a tightly packed barrel‑like wall, and a roof of interlocking protein segments caps the structure. The ring of flotillin at the base defines a circular patch of membrane about 30 nanometers across—essentially fencing off a tiny disk of membrane and the space just above it.
Flexible domes seen inside living cells
To test whether these domes exist in real cells, the team engineered human T cells to produce fluorescently tagged flotillin‑1 and flotillin‑2 and then imaged them by a combination of light microscopy and cryo‑electron tomography. They observed hollow dome‑shaped structures attached to the inner surface of the plasma membrane, closely matching the shape determined in purified samples. Many domes appeared slightly distorted or partially opened, and some contained extra densities inside, likely representing other proteins temporarily trapped under the roof. In some regions, multiple domes clustered together on flat or curved membranes, including on endosomes and small extracellular vesicles, hinting that single domes can join to form larger membrane platforms. 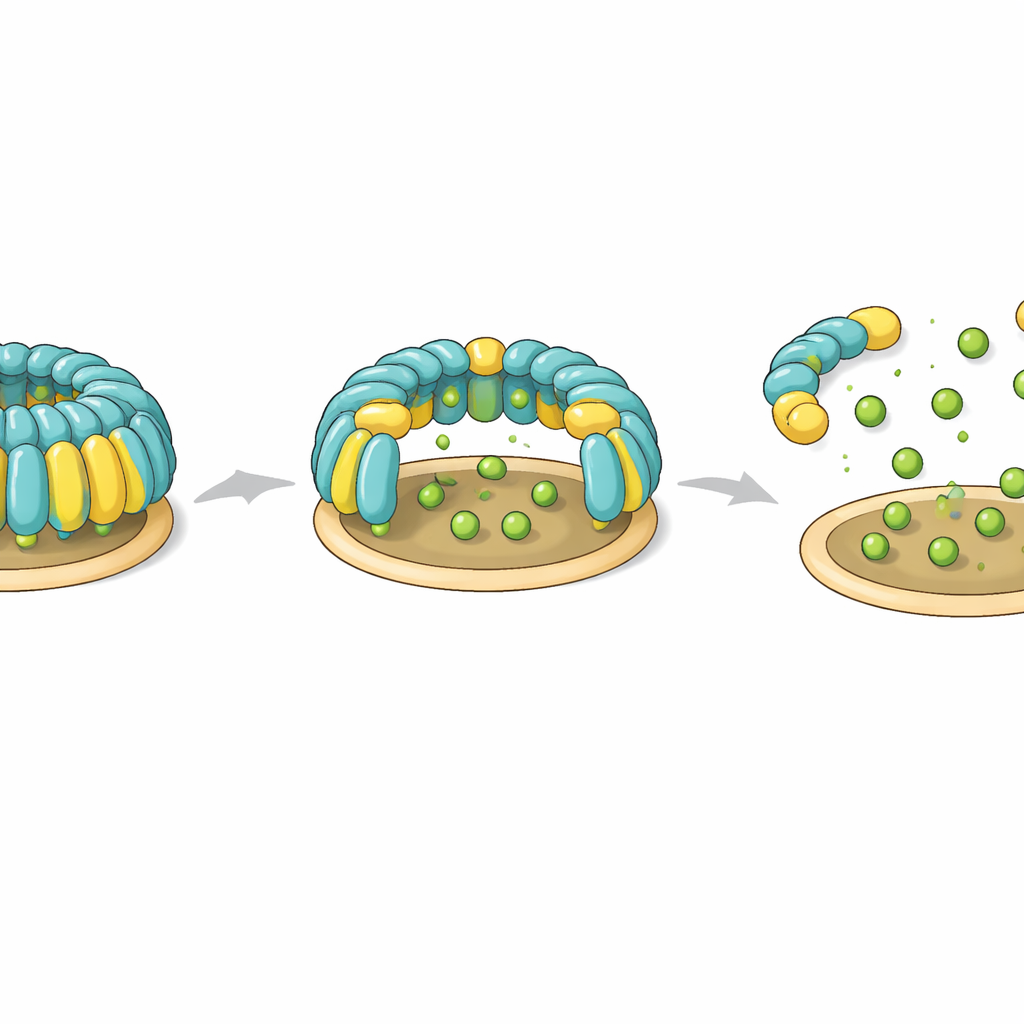
A molecular switch for building and taking apart domes
The study also identifies a potential control knob that cells might use to assemble or dismantle flotillin domes. Two specific tyrosine residues—one on each flotillin protein—sit at hinge‑like positions that connect the membrane‑anchoring base to the helical wall. In the intact complex, these residues help form a snug hydrophobic core that locks the orientation of domains. When the researchers introduced mutations that mimic adding a negative charge, as would happen when enzymes phosphorylate these tyrosines, the entire dome failed to assemble. Non‑charged mutations left assembly intact. These observations suggest that phosphorylation at these hinge points could act as a reversible switch that destabilizes the dome, opening it or breaking it apart during processes such as flotillin‑dependent endocytosis.
Redefining how these domains shape the membrane
Flotillin patches were once thought of mainly as “lipid rafts” rich in cholesterol and related fats. Surprisingly, when the team examined giant membrane bubbles peeled from cells, flotillins favored the more fluid, non‑raft regions rather than the rigid, cholesterol‑dense ones. Lipid analysis showed only a modest enrichment of one particular molecule, sphingosine, within flotillin complexes, while other raft‑associated lipids were not concentrated. This points to a different organizing principle: instead of being passengers on pre‑existing lipid rafts, flotillin domes themselves act as structural containers that laterally segregate pieces of membrane, capturing certain proteins and lipids within their fenced‑off area.
Why this tiny architecture matters
In everyday terms, flotillin complexes behave like modular canopies that cells can deploy on their inner surface to cordon off small circular plots of membrane. Each dome can host a distinct mix of partner proteins and lipids and, by clustering with others, can build larger functional zones for signaling, cargo sorting, or vesicle formation. Because the domes are flexible and appear to open and close, and because their assembly is sensitive to phosphorylation, cells may dynamically remodel these structures in response to cues. This work therefore recasts flotillins from vague “raft markers” into concrete architectural elements that help sculpt the membrane into specialized micro‑workspaces.
Citation: Lu, MA., Qian, Y., Ma, L. et al. Molecular mechanisms of flotillin complexes in organizing membrane microdomains. Nat Commun 17, 2541 (2026). https://doi.org/10.1038/s41467-026-69197-3
Keywords: flotillin, membrane microdomains, cryo-electron microscopy, endocytosis, lipid rafts