Clear Sky Science · en
Tissue-resident macrophage survival depends on mitochondrial function regulated by SerpinB2 in chronic inflammation
Why Fat’s Immune Cells Matter
Most people think of body fat as passive storage, but it is actually a bustling tissue filled with immune cells that help control inflammation and blood sugar. This study reveals how a specific group of long‑lived immune cells in deep belly fat act as guardians against type 2 diabetes—and how chronic inflammation in obesity quietly kills them off by damaging their tiny power plants, the mitochondria.
Two Kinds of Fat Guardians
Visceral adipose tissue—fat packed around the internal organs—contains two main types of immune cells called macrophages. One type is short‑lived and constantly renewed from the blood; these "inflammatory" macrophages tend to fuel swelling and insulin resistance. The other type is long‑term, tissue‑resident macrophages that are born early in life and normally help keep the tissue calm, support healthy fat storage, and promote whole‑body insulin sensitivity. By tracking these cells in mice and comparing gene activity, the researchers showed that resident macrophages are enriched in anti‑inflammatory and insulin‑sensitizing genes, whereas incoming monocyte‑derived macrophages express genes that worsen blood sugar control.
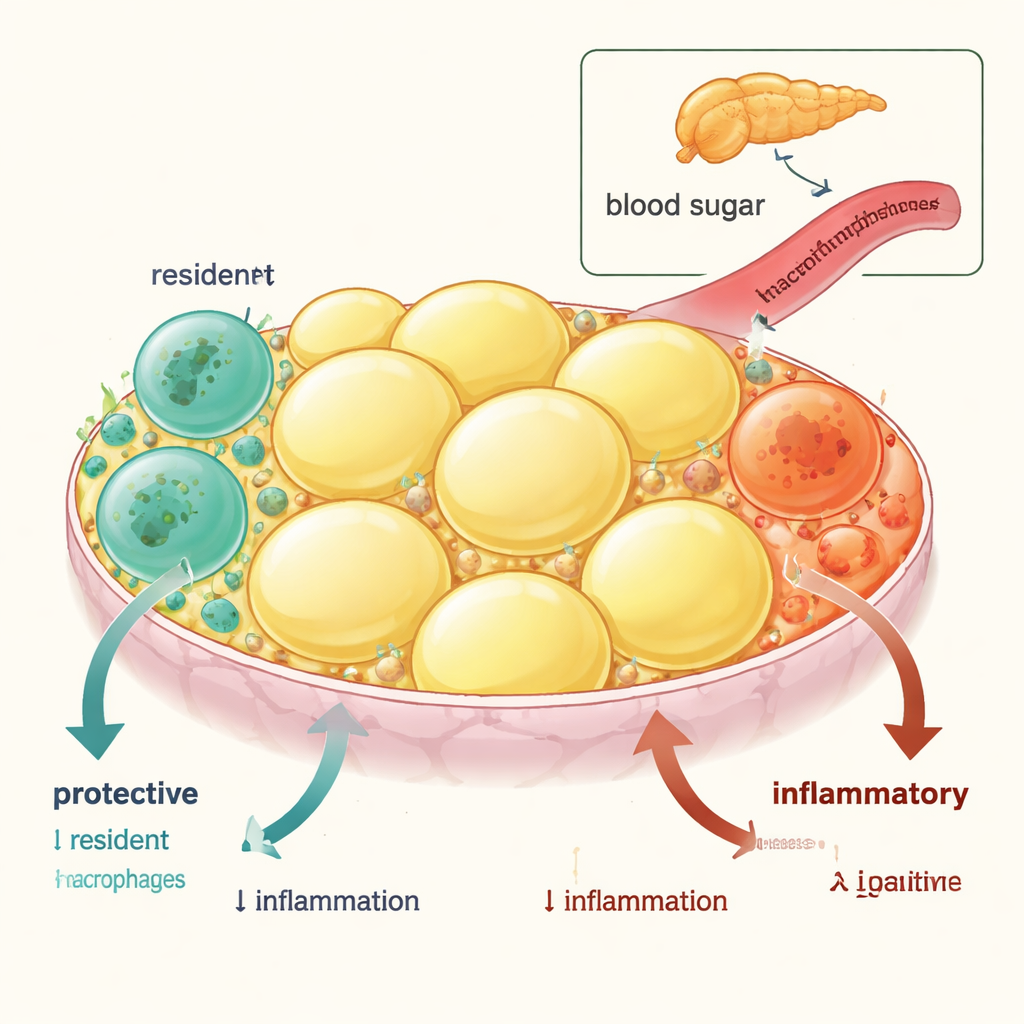
When Inflammation Kills the Good Guys
In obesity, this protective balance shifts. The team found that in both obese mice and obese or diabetic people, the number of resident macrophages in visceral fat falls, while inflammatory macrophages increase. Careful imaging and cell‑death markers revealed why: resident macrophages undergo more programmed cell death (apoptosis), whereas the inflammatory cells do not. Remarkably, when obese mice were taken off a high‑fat diet, the resident population slowly recovered through local proliferation, and metabolic health improved—showing that this loss is driven by the inflammatory state and can be reversed.
A Single Protein Links Mitochondria to Survival
A key clue was a protein called SerpinB2, best known for roles in blood clotting. The researchers discovered that SerpinB2 is produced at very high levels inside resident macrophages, but much less in inflammatory ones. In obese humans and mice, resident macrophages sharply reduced SerpinB2 expression, and higher body mass index was associated with fewer SerpinB2‑positive cells. Experiments in cells lacking SerpinB2 showed increased leakage of cytochrome c—a crucial protein—from mitochondria into the cytoplasm, a classic trigger of apoptosis. These SerpinB2‑deficient macrophages had fewer antioxidant defenses, more mitochondrial reactive oxygen species (ROS), and higher oxygen consumption, all signs of stressed, overworked mitochondria that push cells toward inflammatory behavior and death.
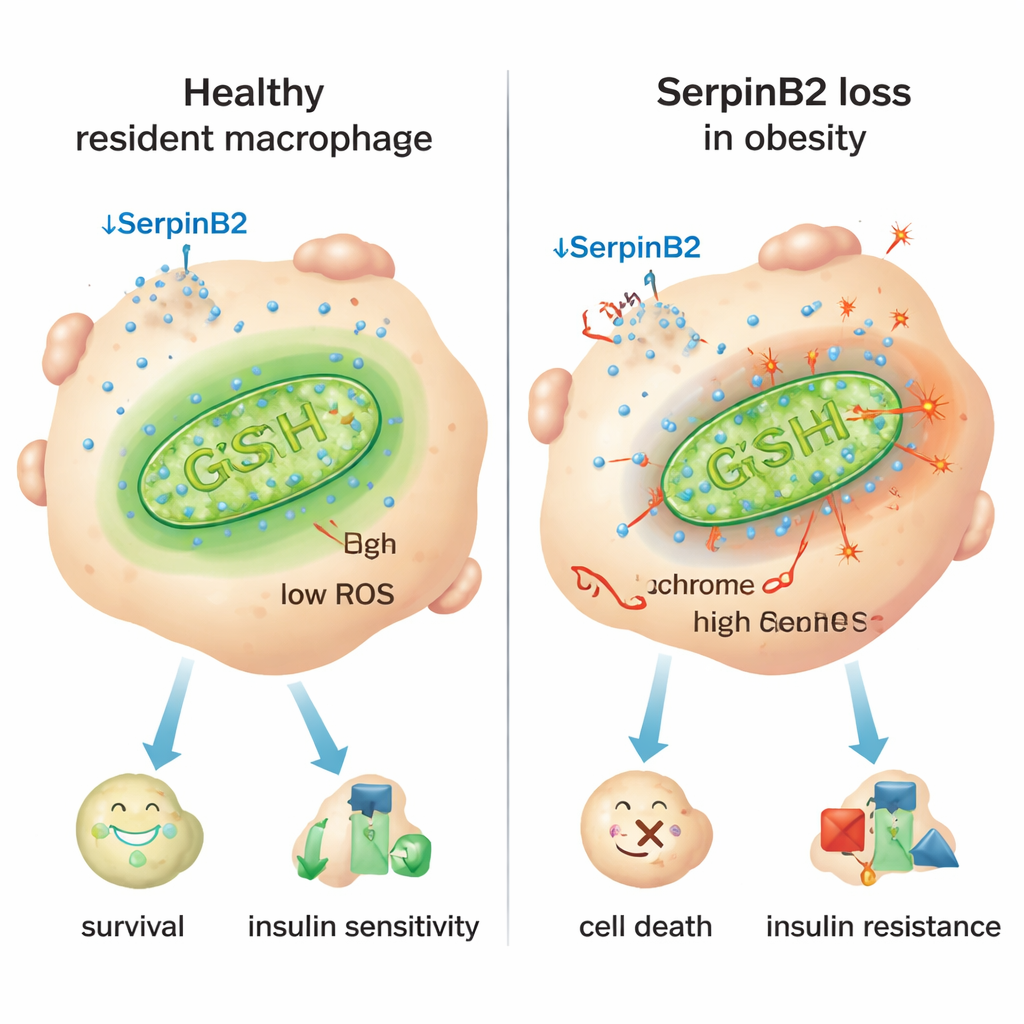
The Inflammatory Switch and a Possible Fix
The study traced this mitochondrial stress back to chronic inflammatory signals in obesity, especially the immune messenger interferon‑γ (IFN‑γ), which is produced in excess in fat tissue. IFN‑γ activates a transcriptional repressor called Ikaros that binds to the SerpinB2 gene’s control region and turns it down. In mice engineered so their myeloid cells could not respond to IFN‑γ, resident macrophages preserved SerpinB2, survived better, and the animals showed lower blood sugar, better insulin sensitivity, and smaller fat cells. Conversely, deleting SerpinB2 specifically in macrophages worsened glucose intolerance and insulin resistance, partly because fat tissue became more inflamed and expanded. Boosting antioxidant glutathione levels with the supplement N‑acetylcysteine (NAC) restored mitochondrial protection, rescued resident macrophage survival, shrank adipocytes, and improved metabolic measures even when SerpinB2 was missing.
What This Means for Metabolic Health
For a lay reader, the core message is that not all fat or fat‑resident immune cells are harmful. A specialized, long‑lived group of macrophages in visceral fat actually protects us from runaway inflammation and high blood sugar by keeping their mitochondria healthy through SerpinB2‑driven antioxidant defenses. In obesity, chronic inflammatory signals shut down SerpinB2, mitochondria become stressed, these protective cells die, and more damaging macrophages take over, contributing to insulin resistance. The work suggests that therapies aimed at preserving resident macrophages—by blocking the IFN‑γ/Ikaros brake on SerpinB2 or by supporting glutathione‑based antioxidant defenses—could help prevent or treat metabolic diseases linked to obesity.
Citation: Vasamsetti, S.B., Sadaf, S., Uddin, M.A. et al. Tissue-resident macrophage survival depends on mitochondrial function regulated by SerpinB2 in chronic inflammation. Nat Commun 17, 1493 (2026). https://doi.org/10.1038/s41467-026-69196-4
Keywords: visceral fat, macrophages, mitochondria, insulin resistance, SerpinB2