Clear Sky Science · en
Metabolic engineering of doxorubicin biosynthesis through P450-redox partner optimization and structural analysis of DoxA
Why a cancer drug’s origin story matters
Doxorubicin is one of the workhorse drugs of modern chemotherapy, used to treat cancers from leukemia to breast tumors. Yet despite decades of use, the way we make this medicine is surprisingly roundabout: industry usually builds it from a related compound rather than letting its native microbe finish the job. This study uncovers why the producing bacterium struggles to complete the final chemical steps and shows how re‑engineering its internal machinery can sharply boost fully formed doxorubicin, opening the door to more reliable and potentially cheaper supplies.
From soil bacteria to life‑saving medicine
Doxorubicin belongs to the anthracyclines, a family of natural molecules built by soil bacteria in the genus Streptomyces. These compounds share a rigid, flat carbon framework that slides between DNA base pairs, plus a sugar unit that nestles into DNA’s grooves. Together, these features jam the cell’s DNA‑handling machinery and ultimately trigger cell death – useful against fast‑growing cancer cells. The classic producer, Streptomyces peucetius, naturally makes mostly a precursor drug called daunorubicin, and only a modest amount of the more effective doxorubicin, which differs by just a single extra oxygen‑bearing group. That tiny structural change dramatically improves activity but turns out to be surprisingly hard for the microbe to achieve efficiently.
Finding the right electrical wiring inside the cell
The crucial enzyme that performs the last three oxidation steps on the drug scaffold is called DoxA, a member of the cytochrome P450 family. Like a miniature chemical factory, DoxA needs a steady flow of electrons to activate oxygen and install new oxygen atoms onto the drug molecule. Inside the bacterium, those electrons are supplied through a relay of helper proteins known as redox partners. The genome of S. peucetius contains several candidates, making it unclear which ones actually pair with DoxA. By comparing gene activity and metabolism in a normal strain, a daunorubicin‑rich mutant, and a doxorubicin‑rich mutant, the researchers pinpointed one ferredoxin (Fdx4) and one ferredoxin reductase (FdR3) as the natural partners. Rebuilding this trio in test‑tube reactions confirmed that DoxA works best when plugged into this specific electron‑transfer chain, much like matching the right power adapter to a device.
Relieving the enzyme’s self‑inflicted slowdown
Even with the correct electrical wiring, DoxA tends to stall at the final step that converts daunorubicin into doxorubicin. Earlier work hinted that the product itself may jam the enzyme. The team turned to a neighboring gene, dnrV, whose function had been mysterious. Biochemical tests showed that the DnrV protein tightly binds a range of anthracycline molecules, including doxorubicin, without chemically altering them. Adding DnrV to DoxA reactions greatly improved the flow of chemistry, allowing full conversion of precursors to the final drug while avoiding destructive side reactions. In practical terms, DnrV acts like an internal sponge that mops up freshly made doxorubicin, preventing it from clogging the enzyme or damaging DNA inside the producing cell.
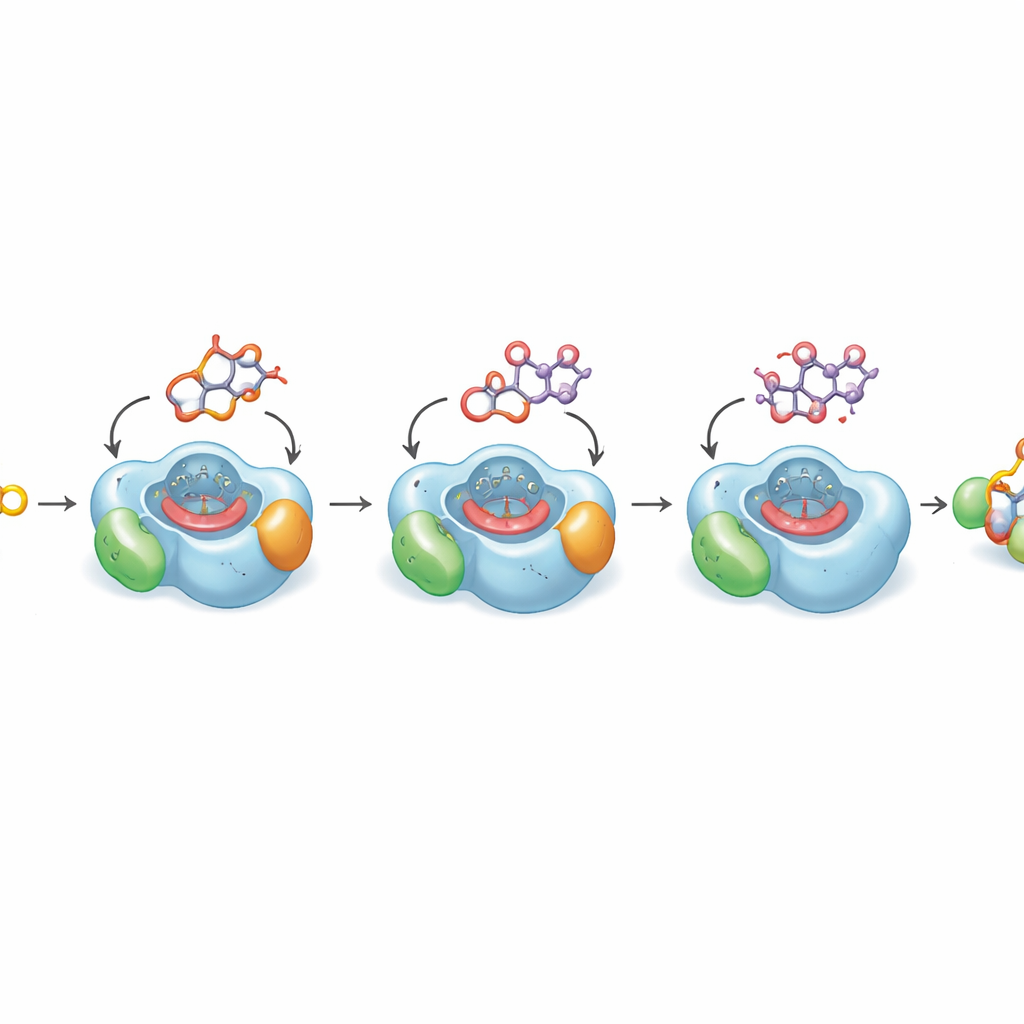
Seeing why the last step is so slow
To understand why the key last oxidation is inherently sluggish, the authors solved high‑resolution crystal structures of DoxA bound to three successive intermediates. These structures reveal how the flat drug core and its attached sugar nestle into a snug pocket above the enzyme’s heme group, the reactive metal center that performs oxidation. For the earlier steps, the carbon atom that must be modified sits close to the heme in an ideal position for reaction. But in the final substrate, daunorubicin, the portion of the molecule that needs to be hydroxylated bends away from the heme into a small oily cavity, an arrangement that quantum calculations show is energetically favored. Computer simulations over microsecond timescales confirm that the molecule only occasionally visits a productive pose for that last oxygen insertion. This structural bias explains why the conversion to doxorubicin is naturally inefficient.
Building a better doxorubicin‑making strain
Armed with these insights, the researchers rationally redesigned the bacterium rather than relying on random mutagenesis. They introduced a DNA repair gene to stabilize production, then installed extra copies of doxA, the optimal redox partners fdx4 and fdr3, and the helpful binder dnrV under carefully chosen promoters to balance their amounts. They also fine‑tuned culture conditions and used special resins to soak up toxic and sticky intermediates. In laboratory flasks and a 20‑liter bioreactor, the best engineered strain produced 336 milligrams of doxorubicin per liter of culture with an 81:19 ratio of doxorubicin to daunorubicin—an increase of about 180% over the starting strain and a far cleaner product mixture.
What this means for future cancer drug supply
By dissecting both the electrical support system and the 3D structure of a key enzyme, this work explains why a medically vital microbe underperforms at making our most prized anthracycline. Matching DoxA with its natural redox partners, adding a drug‑binding helper protein, and re‑balancing gene expression turned a reluctant producer into a robust one. While further enzyme tuning could accelerate the final chemical step even more, the study already brings fully biological doxorubicin manufacturing much closer to industrial reality. For patients and health systems, such advances could translate into more secure, scalable, and cost‑effective access to a staple chemotherapy drug.
Citation: Koroleva, A., Artukka, E., Yamada, K. et al. Metabolic engineering of doxorubicin biosynthesis through P450-redox partner optimization and structural analysis of DoxA. Nat Commun 17, 2358 (2026). https://doi.org/10.1038/s41467-026-69194-6
Keywords: doxorubicin production, anthracycline biosynthesis, enzyme engineering, microbial drug manufacturing, cytochrome P450