Clear Sky Science · en
Atypical protein kinase C activation drives intestinal glucose excretion in diabetes mellitus
Turning the Gut into a Sugar Valve
People with diabetes live with too much sugar circulating in their blood. Bariatric surgeries like gastric bypass can normalize blood sugar surprisingly quickly, even before much weight is lost, but how this happens has been a mystery. This study uncovers a gut-based "sugar valve"—a way for the intestine to pull sugar out of the blood and flush some of it back into the gut—suggesting a new drug target that could mimic the benefits of surgery without the operation.
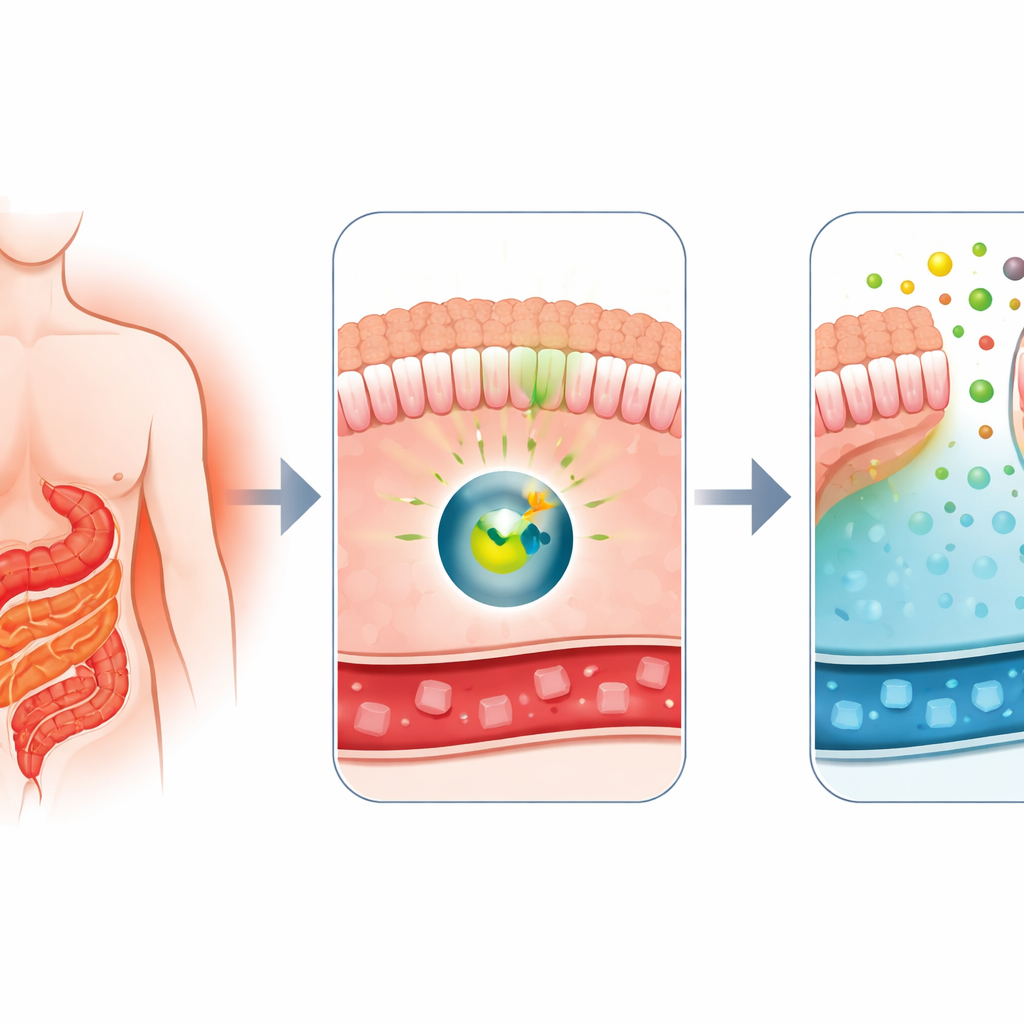
Why Surgery Changes Blood Sugar So Fast
Doctors have long known that Roux-en-Y gastric bypass, a common weight-loss surgery, is also one of the most powerful treatments for type 2 diabetes. Soon after surgery, patients’ blood sugar often improves dramatically, even before they shed many pounds. Earlier work from this group showed that, after bypass, parts of the small intestine begin to act like a sponge, soaking up glucose from the bloodstream and releasing some of it into the gut’s inner space. That odd behavior, called intestinal glucose excretion, appeared to rely on a transporter protein called GLUT1. The big question was: what molecular switch tells the intestine to flip into this sugar-clearing mode?
Using Big Data to Find a Molecular Switch
To hunt for that switch, the researchers used a large-scale computational drug-discovery system built on the Connectivity Map, a database of how thousands of chemicals change gene activity in cells. They compared gene expression patterns from five situations where intestinal glucose excretion is high—including tissue from bypass-operated rats and cells treated with an intestinal growth signal—to patterns produced by known drugs. One class of compounds consistently stood out: activators of protein kinase C (PKC), a family of enzymes that relay signals inside cells. When they dug deeper, the team homed in on the “atypical” subgroup of PKC enzymes, especially one called PKCζ, whose activity in intestinal cells most closely matched the bypass-like, glucose-excreting state.
The Gut Cells’ Sugar Pump in Action
Zooming in on individual intestinal cells using single-cell RNA sequencing, the authors found that bypass surgery boosts both PKCζ and GLUT1 in specific nutrient-absorbing cells lining the distal small intestine. In cell cultures and human intestinal organoids, artificially increasing PKCζ or treating with prostratin—a plant-derived compound that safely activates these atypical PKC enzymes—drove GLUT1 to the cell surface. There, GLUT1 acted like a two-way pump: it drew more glucose out of the blood-facing side of the cells and allowed excess glucose to exit into the gut space. Importantly, this rerouting of sugar did not significantly accelerate the cells’ own sugar-burning (glycolysis), indicating that the main effect was disposal of glucose, not extra energy production.
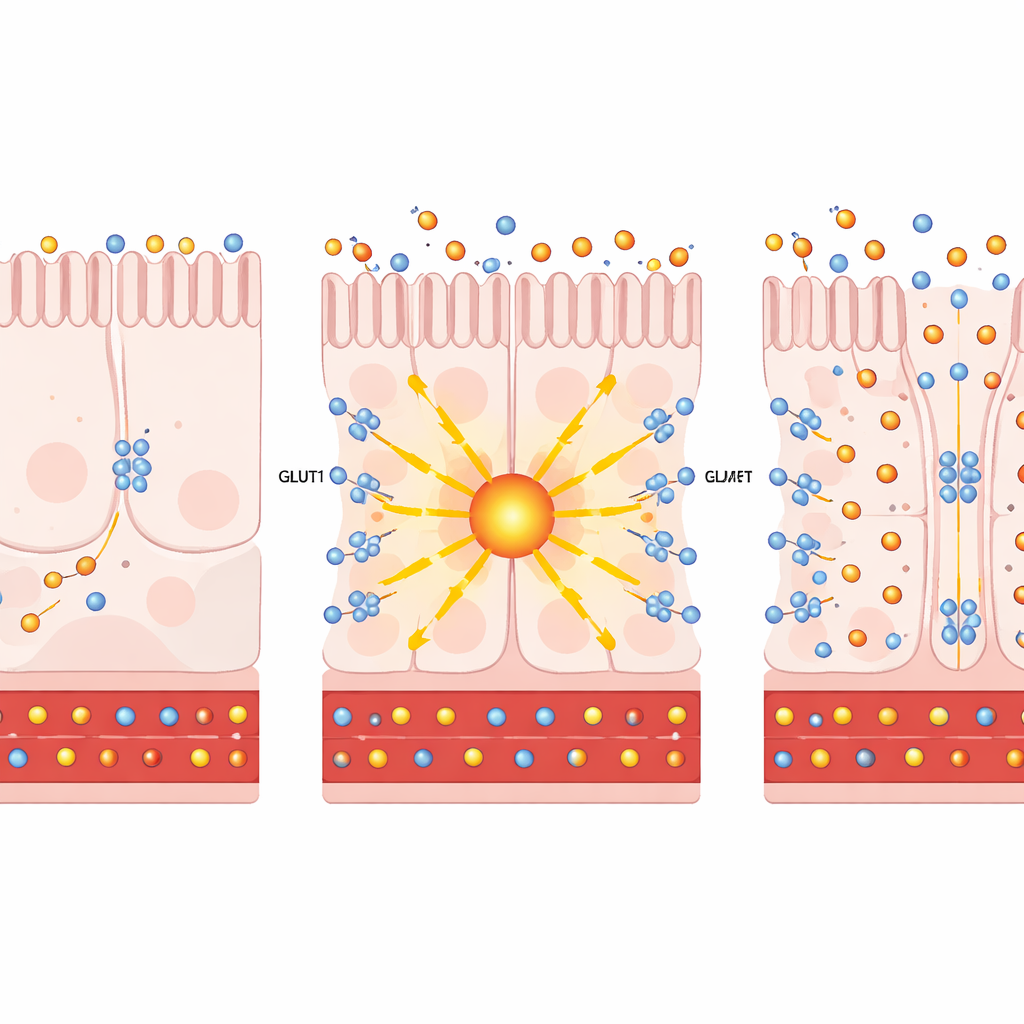
From Cells to Mice: Mimicking Surgery Without the Scalpel
The team then tested whether activating this pathway in live animals could improve diabetes. In obese, insulin-resistant mice, delivering extra PKCζ specifically to the intestine led to slower weight gain, lower fasting blood sugar, and better glucose tolerance. Radioactive sugar tracers showed more sugar being taken up by the distal intestine and washed out into the gut lumen. A similar picture emerged in diabetic mouse models treated with prostratin: they gained less weight, handled sugar loads better, and shunted more glucose into the intestinal tract, all without changes in insulin levels, food intake, or gut barrier integrity. In tissue slices, GLUT1 appeared on both the blood-facing and gut-facing sides of the cells, perfectly positioned to pull sugar from the circulation and push it into the gut.
What This Could Mean for Future Diabetes Care
To a non-specialist, the key message is that the intestine can be turned into a controlled escape route for excess blood sugar. By activating a specific enzyme inside intestinal cells—atypical PKC—the researchers were able to coax cells to move more GLUT1 transporters to their surface and behave like a reversible drain, pulling sugar from the blood and dumping some of it back into the gut. In mice, this gut-driven sugar disposal improved blood sugar and limited weight gain without overstimulating insulin, changing appetite, or damaging the intestine. Although prostratin and related compounds are not yet approved diabetes drugs, this work points to the aPKC–GLUT1 pathway as a promising target for future medicines that could partly reproduce the metabolic benefits of gastric bypass surgery without requiring an operation.
Citation: Kang, C.W., Hong, ZY., Oh, J.H. et al. Atypical protein kinase C activation drives intestinal glucose excretion in diabetes mellitus. Nat Commun 17, 2417 (2026). https://doi.org/10.1038/s41467-026-69193-7
Keywords: intestinal glucose excretion, type 2 diabetes, GLUT1 transporter, atypical protein kinase C, prostratin