Clear Sky Science · en
Can ferric-oxyl excited states explain elongated iron-oxygen bonds in heme peroxidase catalytic intermediates?
Why iron and oxygen bonds in enzymes matter
Inside our cells, special proteins called enzymes use oxygen to safely carry out powerful chemical reactions. Among them, heme peroxidases rely on an iron–oxygen pair at their core to break down hydrogen peroxide, a reactive, potentially damaging molecule. For decades, scientists have disagreed about the exact nature of this iron–oxygen bond: is it more like a tight double bond or a looser single bond—and what does that mean for how these enzymes work? This study tackles that mystery using ultrafast X-ray methods and advanced calculations, revealing that the answer lies in fleeting excited states of the iron–oxygen unit itself.
Following an enzyme in real time
The researchers focused on a bacterial dye-decolourising peroxidase, a heme enzyme that normally cycles through two key high-energy forms, known as Compound I and Compound II. These forms both feature an iron atom bound to oxygen and are central to how the enzyme processes hydrogen peroxide and oxidizes other molecules. Previous experiments on similar enzymes produced puzzlingly long iron–oxygen bond lengths, which some scientists interpreted as evidence that the supposedly highly reactive iron–oxygen unit had either been altered by X-rays or had picked up an extra proton, changing its character. To avoid such artifacts, the team used time-resolved serial femtosecond X-ray crystallography at an X-ray free-electron laser, capturing diffraction and X-ray emission signals from thousands of tiny protein crystals at room temperature, all within tens of femtoseconds—faster than damage can occur.
Watching the chemistry unfold inside crystals
In their setup, microcrystals of a slightly modified version of the enzyme were mixed with hydrogen peroxide directly on a moving tape and then probed after delays ranging from half a second to tens of minutes. Early time points favor the formation of Compound I, while later ones are dominated by Compound II. Structural data showed that, in both intermediates, the iron atom sits next to a single oxygen atom in the heme pocket, and that protective loop regions of the protein shift to shield this highly oxidizing center. Importantly, precise measurements revealed that the iron–oxygen bond length remained around 1.83 angstroms across all time points—longer than expected for a classic double-bonded ferryl (Fe(IV)=O) species and closer to a single bond, yet the spectral signatures from X-ray emission and optical absorption clearly indicated high oxidation states consistent with Compounds I and II.
Ruling out simple explanations
Because the experiments were carried out with ultrashort pulses at room temperature, the usual suspects for distorted bond lengths—X-ray-induced reduction and cryogenic artifacts—could be largely dismissed. The team also tested whether the oxygen bound to iron had become protonated, turning the double bond into a hydroxide-like single bond. However, known acid–base properties of similar heme centers, together with previous chemical studies, argue strongly against such protonation in this type of enzyme. The spectroscopic data further showed that the iron remained in a high oxidation and low-spin state after reaction with hydrogen peroxide, just as expected for bona fide ferryl intermediates, reinforcing the idea that the unexpectedly long bond must arise from more subtle electronic effects rather than a simple change in chemical form.
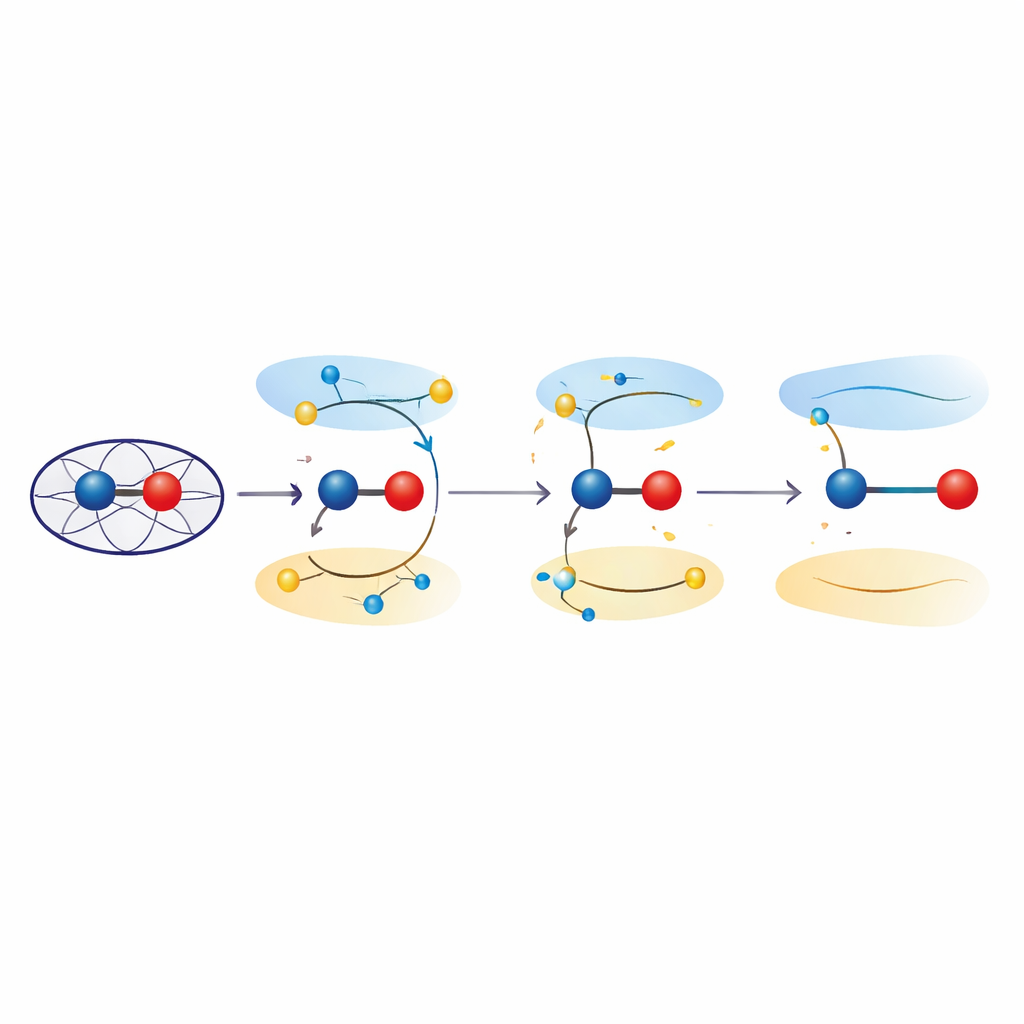
Excited states that stretch bonds
To probe those effects, the researchers turned to quantum mechanical calculations on both simplified models and the full protein environment. Using time-dependent density functional theory and combined quantum mechanics/molecular mechanics approaches, they examined how promoting electrons from bonding to antibonding orbitals in the iron–oxygen unit changes the preferred bond length. These excited states, which are energetically close to the ground ferryl state, consistently produced iron–oxygen distances in the 1.8–1.9 angstrom range—matching the crystallographic observations. Analysis of the electron distribution showed that in these states the iron–oxygen pair no longer behaves like a pure Fe(IV)=O double bond, but instead takes on “ferric–oxyl” character, with properties akin to Fe(III) bound to an oxygen-centered radical. Quantum refinement of the experimental structures confirmed that such excited-state descriptions fit the data at least as well as conventional ground-state models.
What this means for understanding enzyme power
Put simply, the work suggests that long iron–oxygen bonds observed in these heme peroxidases do not require invoking damage, reduction, or hidden protons. Instead, they can arise naturally when the ferryl unit briefly accesses low-lying excited states that weaken the bond and impart ferric–oxyl character. For non-specialists, this means that the “business end” of many oxygen-activating enzymes may be more dynamic and electronically flexible than once thought, with subtle shifts in electron placement changing bond strength and reactivity without altering the overall chemistry. Recognizing these excited states could reshape how scientists interpret structural data on powerful biological oxidants and may guide the design of artificial catalysts that mimic, or deliberately tune, this delicate electronic balancing act.
Citation: Williams, L.J., Kamps, J.J., Brânzanic, A.M.V. et al. Can ferric-oxyl excited states explain elongated iron-oxygen bonds in heme peroxidase catalytic intermediates?. Nat Commun 17, 2324 (2026). https://doi.org/10.1038/s41467-026-69192-8
Keywords: heme peroxidase, ferryl intermediate, iron-oxygen bond, excited electronic states, X-ray free-electron laser