Clear Sky Science · en
MerTK-triggered TGFβ1 autocrine signal regulates microglial response to neurodegeneration
How the Brain’s Clean-Up Crew Shapes Disease
When nerve cells in the brain and optic nerve die, they leave behind a trail of debris. Specialized immune cells called microglia act as the brain’s clean-up crew, swallowing this waste. But microglia do more than tidy up: how they react can either protect nerve circuits or worsen diseases like Alzheimer’s. This study uncovers a hidden self-communication loop inside microglia that is switched on by debris from dying nerve fibers and helps determine how strongly these cells respond to damage.
A Trigger Hidden in Dying Nerve Fibers
To probe this process, the researchers used a mouse model in which the optic nerve is crushed, causing the long projections of eye neurons to degenerate without killing the cell bodies. This cleanly isolates the aftermath of axon breakdown. On the surface of these degenerating fibers, a fatty molecule flips outward, acting as an “eat me” cue to nearby microglia. Those signals are sensed by a receptor on microglia called MerTK, one of a small family of proteins that recognize debris. By selectively deleting MerTK in microglia, the team showed that without this receptor, microglia failed to mount their usual response to injury: they divided less, moved less, and turned on fewer genes linked to damage control and inflammation.
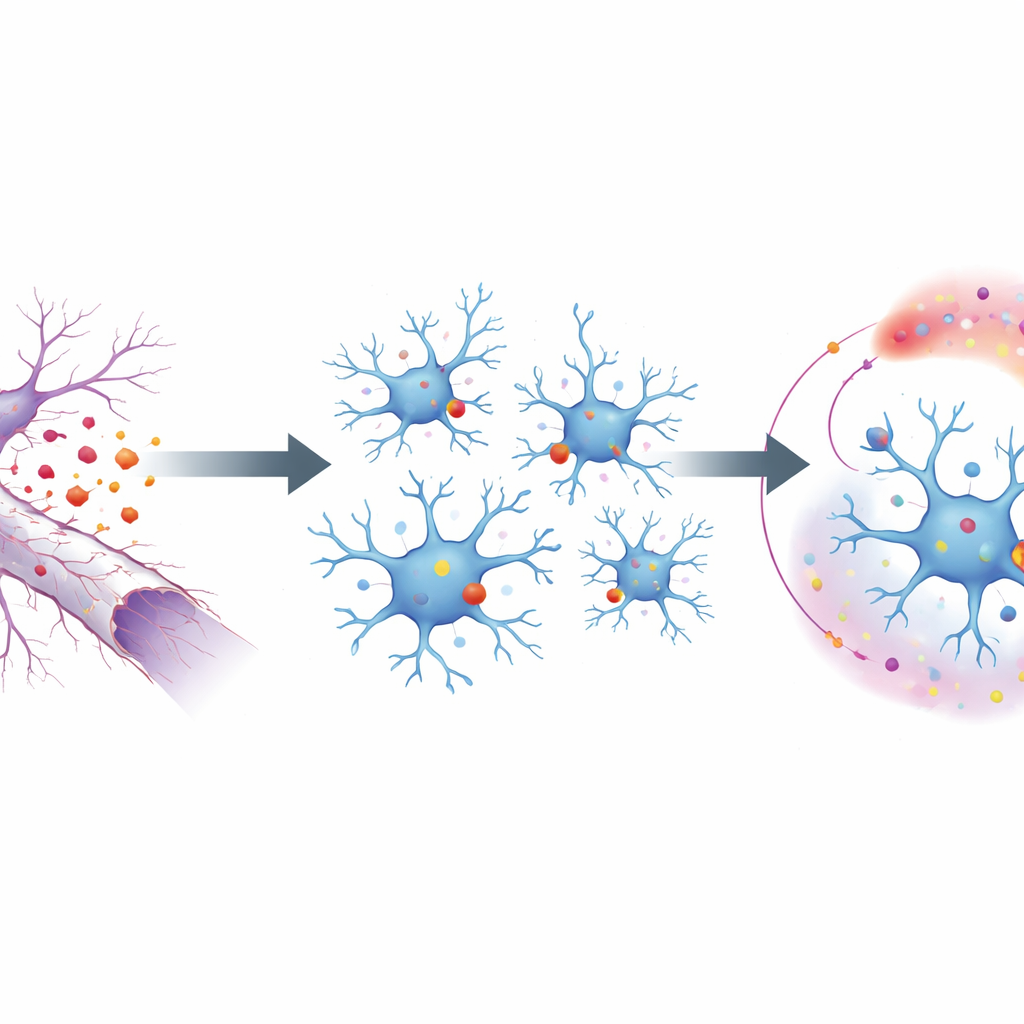
From Surface Signal to Genetic Reprogramming
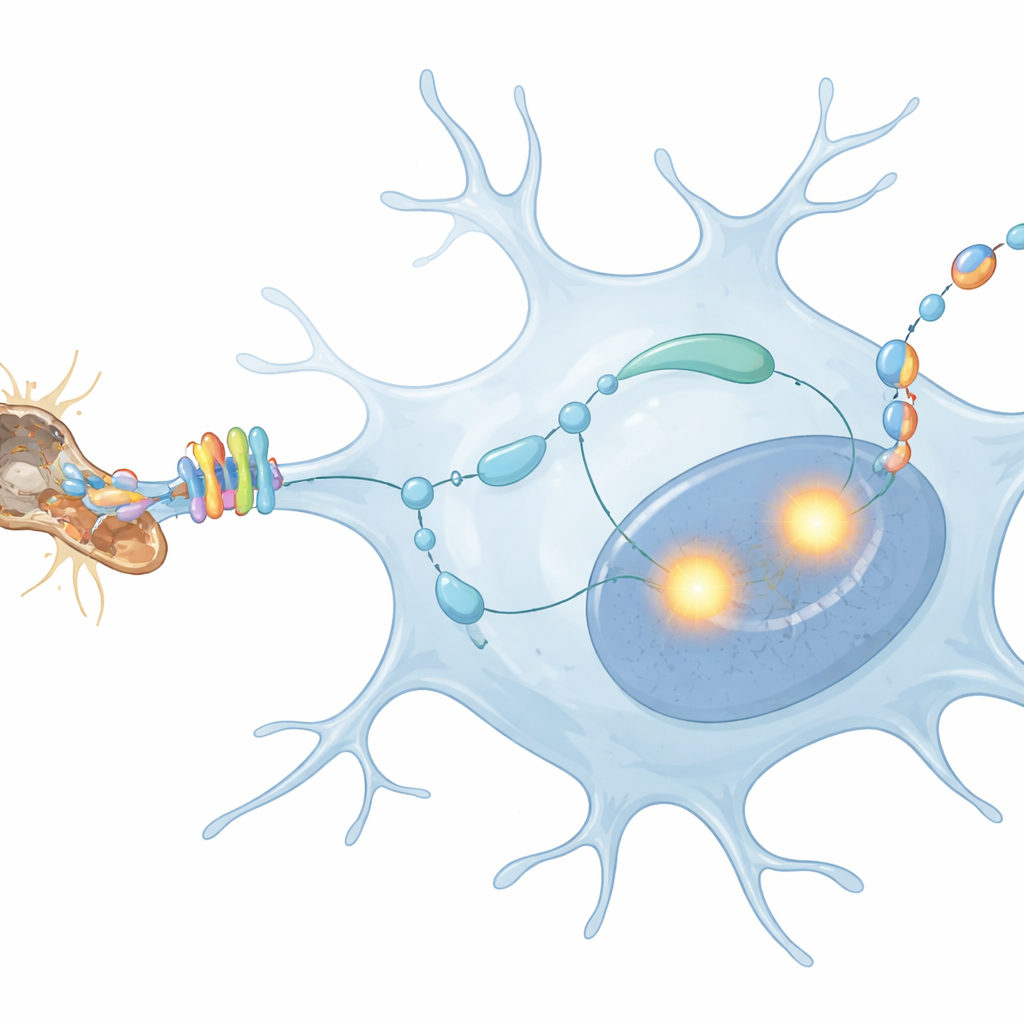
Looking inside microglia, the scientists traced how MerTK’s surface signal is converted into changes in gene activity. They found that MerTK activates a molecular relay called phospholipase C, which in turn boosts two master control proteins in the nucleus, PU.1 and IRF8. These factors act like foremen on the microglial assembly line, switching on many genes needed for an activated state. By using DNA-binding maps, the team discovered that PU.1 and IRF8 bind directly to a specific site in the gene that encodes TGFβ1, a powerful signaling protein. When this binding site was mutated so the foremen could no longer dock there, microglia in injured nerves could no longer ramp up TGFβ1 production, even though other baseline functions remained intact.
A Self-Feedback Loop in Microglia
The next question was what TGFβ1 actually does in this context. The study shows that microglia make TGFβ1 and then respond to it themselves, forming an autocrine, or self-feedback, loop. When TGFβ1 or its receptors were genetically removed only from microglia, injury still caused axons to degenerate, but microglia no longer showed their full activation program. They proliferated less and expressed fewer damage-response and inflammatory genes, even though many homeostatic identity markers were still suppressed by other signals. A key downstream molecule of TGFβ signaling, called phosphorylated SMAD2, appeared specifically in microglial nuclei after injury and largely disappeared when TGFβ1 or its receptors were knocked out, confirming that the self-feedback loop was broken.
From Mouse Optic Nerve to Alzheimer’s Brains
To test whether this pathway matters beyond an artificial injury model, the authors examined a widely used mouse model of Alzheimer’s disease. In these animals, microglia clustered around amyloid plaques showed high levels of PU.1, IRF8, TGFβ1 and activated TGFβ signaling, echoing the optic nerve findings. The team then turned to human data from single-nucleus RNA sequencing of brain tissue from Alzheimer’s patients. There, microglia stood out as the main cells expressing the human counterparts of MerTK, PU.1, IRF8, and TGFB1. In postmortem brain sections from patients, microglia also showed strong signs of active TGFβ signaling, in contrast to tissue from control donors. Together, these observations argue that the same self-communication loop operates in human disease.
Why This Self-Talk Matters for Brain Health
This work reveals a chain of events in which debris from degenerating nerve fibers activates MerTK on microglia, which then boosts PU.1 and IRF8, which in turn drive microglia to produce TGFβ1 that feeds back onto themselves. This self-talk sharpens and sustains their response to neurodegeneration. For a layperson, the key message is that the brain’s janitorial cells are not just passive cleaners; they actively listen to their own signals, and this feedback loop may influence whether their actions help protect neurons or push diseases like Alzheimer’s forward. Understanding and possibly tuning this circuit could open new avenues to moderate harmful inflammation while preserving the beneficial clean-up that microglia provide.
Citation: Huang, Y., Deng, Z., Zhou, Z. et al. MerTK-triggered TGFβ1 autocrine signal regulates microglial response to neurodegeneration. Nat Commun 17, 2312 (2026). https://doi.org/10.1038/s41467-026-69189-3
Keywords: microglia, neurodegeneration, Alzheimer’s disease, brain inflammation, TGF-beta signaling