Clear Sky Science · en
A synthetic system for RNA-responsive pyroptosis based on type III-E CRISPR nuclease-protease
Turning Deadly Cell Bursts into Targeted Tools
Our bodies sometimes defend themselves by making infected or damaged cells explode in a fiery kind of self-destruct called inflammatory cell death. This blast not only removes dangerous cells but also summons the immune system. The new study introduces a synthetic gene circuit, called DAMAGE, that can read the RNA messages inside a cell and decide whether that cell should be destroyed. By wiring a microbial defense system to our own cell-death machinery, the researchers show a way to selectively wipe out virus‑infected, cancerous, or aged cells while sparing healthy neighbors. 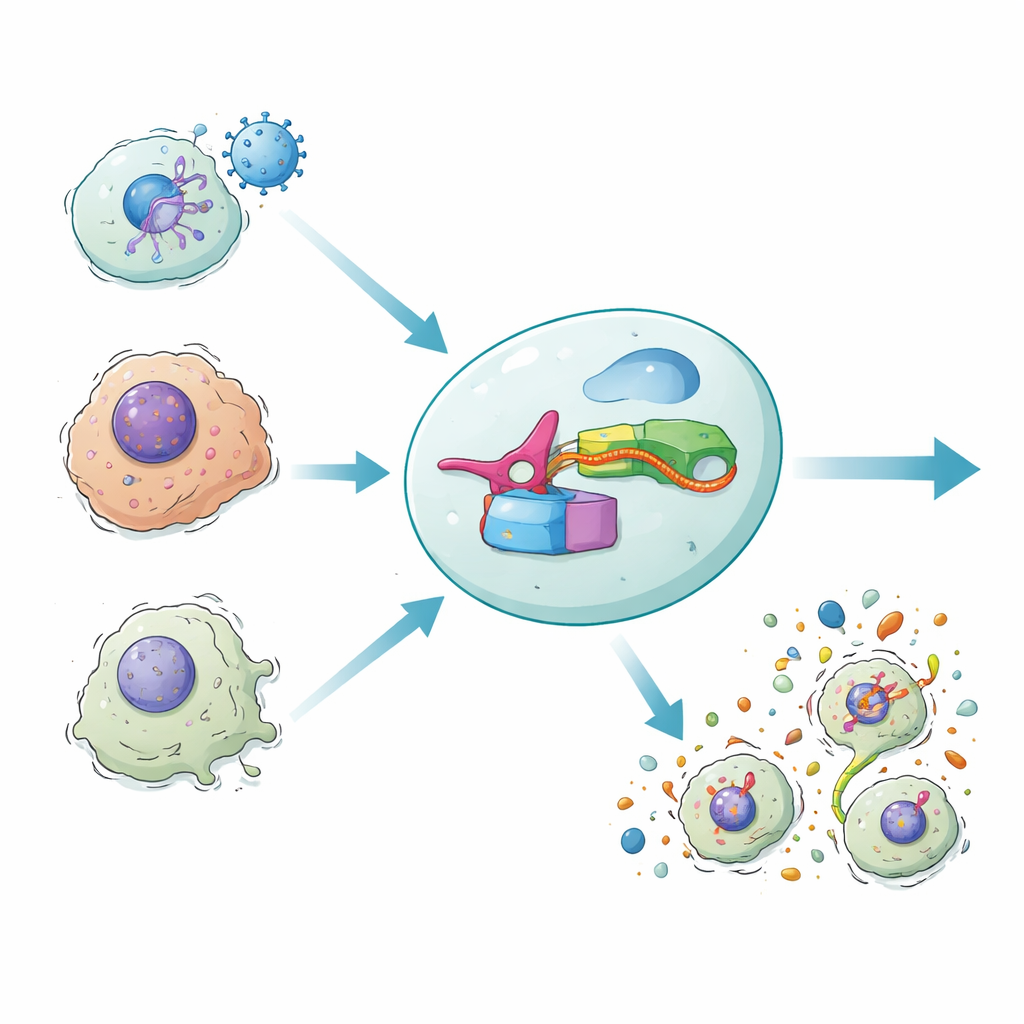
How Cells Normally Blow Themselves Up
In inflammatory cell death, a family of proteins known as gasdermins sit inside cells like loaded charges. Each gasdermin has a “killer” front half that can punch holes in cell membranes, and a “safety cap” back half that keeps the killer in check. When immune sensors detect danger, enzymes cut gasdermins in two, freeing the front half to assemble pores in the outer membrane. The cell swells, bursts, and spews out alarm signals that rally immune cells. This powerful response helps fight infections and tumors, but its natural control network is tangled and hard to rewire for therapy.
Borrowing Bacterial Defenses to Read RNA
Bacteria have evolved compact defense modules known today through CRISPR technology. One of these, the type III-E system, uses a protein called Cas7‑11 that can latch onto specific RNA sequences with the help of a short guide RNA. When Cas7‑11 finds a matching target RNA, it flips on a companion enzyme, Csx29, that cuts a partner protein, Csx30. The authors realized that Csx30 could act as a customizable “hinge” between any two protein parts. They fused this hinge between the killer and safety-cap halves of human gasdermins, creating artificial executors that are only split apart when the CRISPR‑like sensor detects its chosen RNA inside a cell.
A Programmable Death Switch for Diseased Cells
This design, named DAMAGE (for Death Manipulation Gene), is built from five pieces: the Cas7‑11 sensor, the Csx29 cutter, a gasdermin–Csx30 fusion, a guide RNA, and the target RNA inside the cell. When the right RNA message appears, Cas7‑11 binds it, activates Csx29, and Csx29 slices the Csx30 hinge in the fusion protein. The gasdermin killer half is released, drills pores in the membrane, and the cell dies in an inflammatory burst. By simply changing the guide RNA, the team redirected DAMAGE to different cellular targets. They showed that it could recognize RNA from respiratory syncytial virus, high‑risk human papillomavirus in cervical cancer cells, and several cancer‑driving KRAS mutations that differ from normal RNA by only a single letter. In each case, cells carrying the suspect RNA were selectively eliminated, while nearby control cells remained intact. 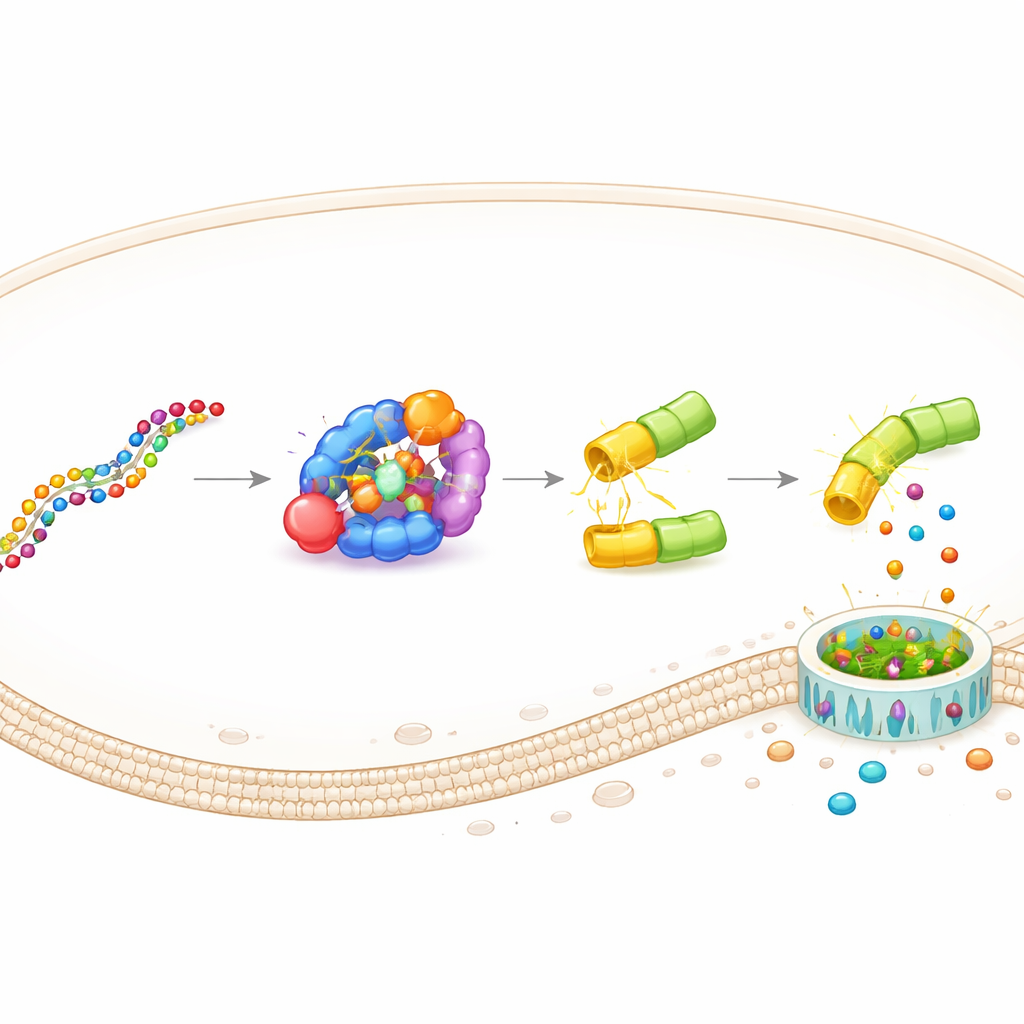
Homing in on Aging Cells
The researchers also asked whether DAMAGE could hunt down senescent cells—aged or stressed cells that stop dividing but accumulate in tissues and contribute to frailty and chronic disease. Such cells commonly crank up production of two gatekeeper proteins, p16 and p21. By programming guides against the RNAs for p16 and p21, the team created a version called DAMAGE‑Aging. This circuit killed cells with high levels of these messages while sparing cells in which p16 had been genetically removed, confirming that the system reads real‑time RNA abundance rather than fixed DNA changes. Drug treatments that raised p16 or p21 levels made cells more vulnerable to this targeted self‑destruct.
Packaging the System for Future Therapies
To move closer to practical use, the authors compressed the many DNA parts of DAMAGE into a streamlined construct, DAMAGE‑Plus, and showed it still functioned reliably. They then transcribed this construct into synthetic mRNA, packaged it in lipid nanoparticles similar to those used in COVID‑19 vaccines, and delivered it into cells in culture. The mRNA version successfully assembled the circuit and triggered targeted inflammatory death in cells carrying viral or cancer‑related RNAs. Although the overall system is still bulky and has only been tested in cell lines, these results hint that programmable, RNA‑guided cell killing might one day be deployed through injectable mRNA formulations.
A New Way to Read and React to Cellular Messages
In plain terms, DAMAGE is a molecular bouncer that checks each cell’s internal RNA “ID” and throws out only those that look dangerous—virus‑infected, mutation‑bearing, or senescent—by forcing them into an explosive form of self‑destruct. The work demonstrates that bacterial RNA sensors can be coupled to human cell‑death machinery with high precision, even distinguishing single‑letter differences in genetic messages. While much engineering remains before such a system could be safely used in animals or people, it outlines a powerful new strategy: reading the live RNA chatter inside cells and translating it directly into life‑or‑death decisions.
Citation: He, M., Wang, W., Zhou, H. et al. A synthetic system for RNA-responsive pyroptosis based on type III-E CRISPR nuclease-protease. Nat Commun 17, 2565 (2026). https://doi.org/10.1038/s41467-026-69179-5
Keywords: pyroptosis, CRISPR, RNA-targeted therapy, cancer immunotherapy, cellular senescence