Clear Sky Science · en
Weak localized electrons enhance electronic coherence for efficient photocatalytic uranium removal from nuclear wastewater
Cleaning Up Nuclear Wastewater with Sunlight
Nuclear power can generate huge amounts of low-carbon electricity, but it leaves behind wastewater laced with radioactive uranium. Safely removing this uranium is essential to protect drinking water, soils, and ecosystems. The study summarized here describes a new sunlight-driven material that can pull uranium out of wastewater far more efficiently than previous approaches, bringing real-world treatment systems closer to practical use.
Why Today’s Cleanup Methods Fall Short
Conventional methods for removing uranium from water rely mainly on adsorption: uranium ions weakly stick to the surface of a filtering material. These methods can be slow, often capture only part of the uranium, and typically require complicated steps to strip the uranium off the filter so it can be reused. In contrast, photocatalytic methods use light-activated materials to convert dissolved uranium into solid particles that fall out of the water on their own. This avoids the problem of desorbing uranium, but existing photocatalysts struggle because the electric charges they generate from light tend to quickly recombine instead of driving useful chemical reactions.
Turning Sunlight into a Uranium Trap
The researchers tackle this bottleneck using a class of porous, crystalline organic materials known as covalent organic frameworks, or COFs. These frameworks can be built from modular organic molecules arranged into highly ordered, nano-sized tunnels. When light hits the COFs, electrons are excited and can help convert oxygen in water into reactive forms like hydrogen peroxide and superoxide. These reactive oxygen species then react with dissolved uranium ions to form uranyl superoxide particles, which readily precipitate from the water, effectively locking the uranium away in a solid form.
Fine-Tuning the Material with Fluorine Atoms
The key innovation is the precise introduction of fluorine atoms into one part of the COF structure. The team constructed three related frameworks: one with no fluorine, one with a moderate level of fluorination (called TAPT-TPA-2F), and one with heavy fluorination. Fluorine is strongly electronegative, meaning it pulls on nearby electrons. By partially decorating the COF with fluorine, the researchers created “weakly localized” electrons that remain mobile enough to move directionally, but not so free that they scatter chaotically. This subtle tuning enhances what the authors call electronic coherence: electrons travel along preferred, ordered pathways from electron-donating regions of the COF to electron-accepting regions, instead of randomly roaming and recombining with holes.
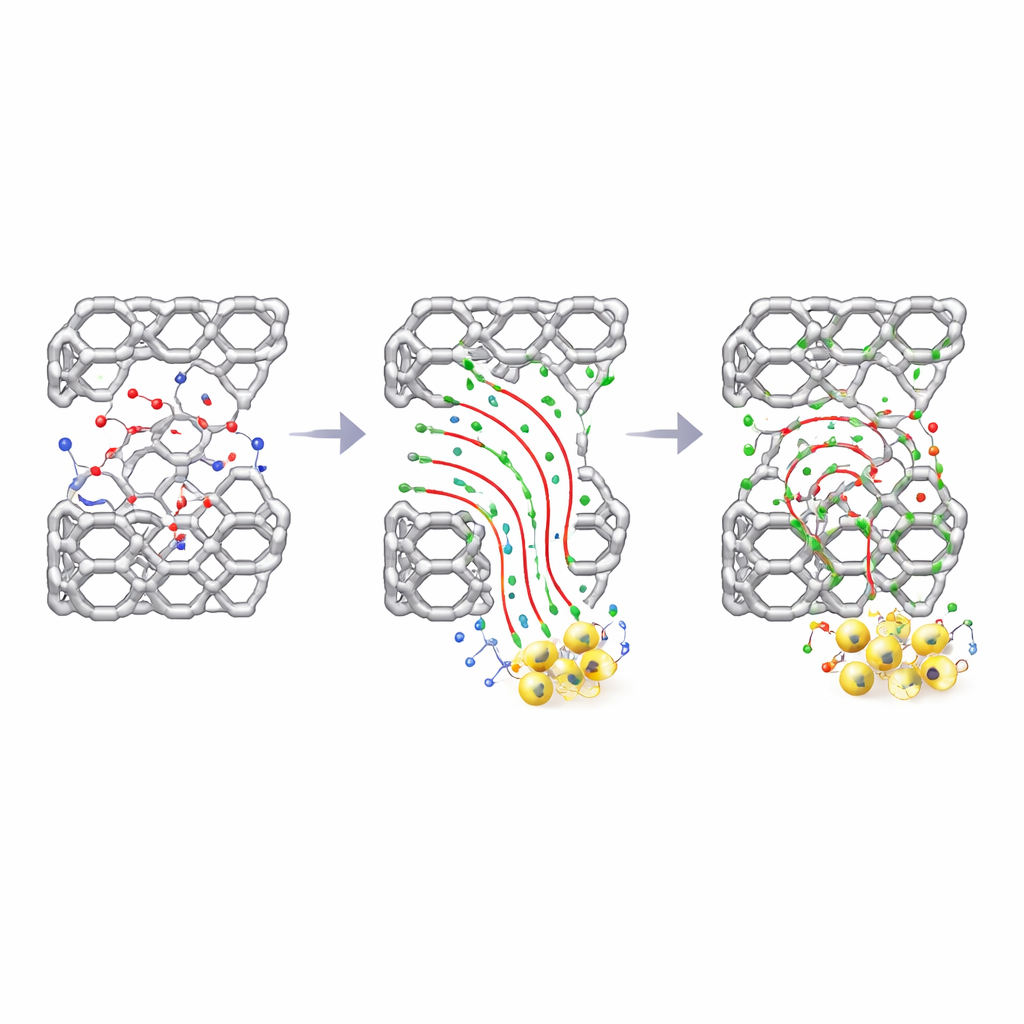
How Better Charge Flow Boosts Uranium Removal
A suite of advanced measurements and simulations shows how this design translates into performance. Ultrafast spectroscopy reveals that the partially fluorinated COF keeps excited charges separated for much longer times than either the non-fluorinated or heavily fluorinated versions. The moderate-fluorine material also shows faster photoresponse, higher carrier mobility, and lower exciton binding energy, all pointing to easier splitting and transport of photo-generated charges. As a result, it produces far more reactive oxygen species, and molecular simulations indicate that uranium ions are more strongly drawn into its pores. In laboratory tests, this optimized COF achieves a record solar-to-chemical conversion efficiency of 1.52% and removes essentially 100% of uranium over a wide pH range, outperforming both its sister materials and many previously reported photocatalysts.
From the Lab Bench to Flowing Wastewater
To move beyond beakers, the team built a compact flow-through reactor in which nuclear wastewater continuously passes over a thin coating of the fluorinated COF while being illuminated, including by natural sunlight. Even at very low uranium concentrations, this device removed 99% of uranium and processed the equivalent of hundreds of grams of uranium per square meter of catalyst area per day, surpassing earlier systems and meeting World Health Organization discharge limits. The material remained structurally intact and reusable over many cycles, suggesting it could operate reliably in real treatment plants.
What This Means for Safer Nuclear Power
In everyday terms, the study shows that by gently “steering” electrons inside a carefully designed porous material, it is possible to use sunlight much more effectively to strip uranium from contaminated water. Partial fluorination creates just the right balance: electrons are guided rather than trapped or wasted, enabling more reactive oxygen to form and more uranium to be locked into harmless solids. This approach could make nuclear wastewater treatment more efficient, compact, and scalable, helping nuclear power deliver low-carbon energy with a smaller environmental footprint.
Citation: Xu, Y., Zhao, R., Liu, Y. et al. Weak localized electrons enhance electronic coherence for efficient photocatalytic uranium removal from nuclear wastewater. Nat Commun 17, 3262 (2026). https://doi.org/10.1038/s41467-026-69178-6
Keywords: nuclear wastewater, uranium removal, photocatalysis, covalent organic frameworks, water treatment