Clear Sky Science · en
hnRNPM cooperates with BCAS2 to modulate alternative splicing during oocyte development
Why egg cell quality matters
Every human life begins with a single egg cell, yet scientists are still uncovering how these unusually large cells prepare for the moment of fertilization. Long before sperm ever arrive, growing egg cells stockpile thousands of RNA messages that will guide their maturation and the first days of embryo development. This study reveals how a little-known protein, working together with a partner, carefully edits those messages in mouse eggs—and what happens when that editing system fails.
Gatekeepers of genetic messages
Egg cells grow in the ovary for weeks while their DNA largely stays quiet. During this time, they accumulate a vast library of maternal RNA messages copied from the genome earlier. These messages must be trimmed and stitched in different ways, a process called alternative splicing, to produce the right mix of protein variants at just the right stages. The authors focused on a protein called hnRNPM, part of a large family that binds RNA, and on its partner BCAS2, both of which are abundant in the female reproductive system. Earlier work hinted that these proteins bind many RNAs in egg cells, but their precise role in shaping fertility remained unclear.
What goes wrong without this editor
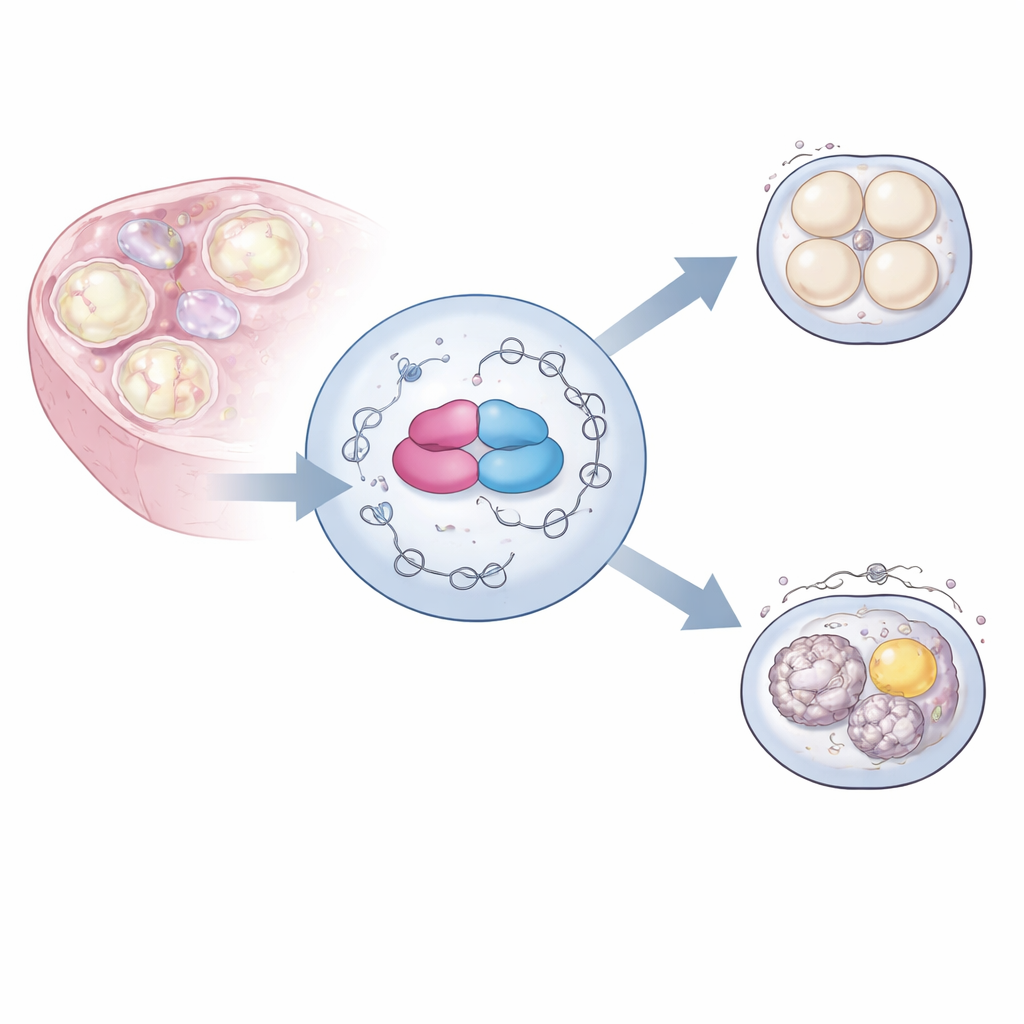
To probe hnRNPM’s function, the team used genetic engineering to remove the Hnrnpm gene specifically from mouse egg precursors. The ovaries of these females looked normal and produced similar numbers of eggs compared with controls. Yet when these eggs were fertilized in the lab, they almost never developed beyond the first divisions, rendering the females completely infertile. Closer inspection of the immature eggs revealed dark clumps in the normally clear cytoplasm. Electron microscopy showed that a fine internal scaffold called the cytoplasmic lattice was largely missing, and key organelles such as mitochondria and fat droplets were abnormally clustered, rather than evenly spread. These changes signaled a deep problem with the egg’s internal organization.
Broken cell division machinery
The same mutant eggs also struggled to complete meiosis, the special cell division that halves the chromosome number. When researchers matured the eggs in culture, they could initiate meiosis but usually stalled before completing it. Under the microscope, the structures that pull chromosomes apart—the spindles—were twisted, multipolar, or poorly anchored. A protein called pericentrin, which helps organize the spindle poles, appeared late and in the wrong places. Together, these defects meant the eggs could not line up and separate chromosomes reliably, a likely cause of the complete developmental failure after fertilization.
Reading and rewriting RNA instructions
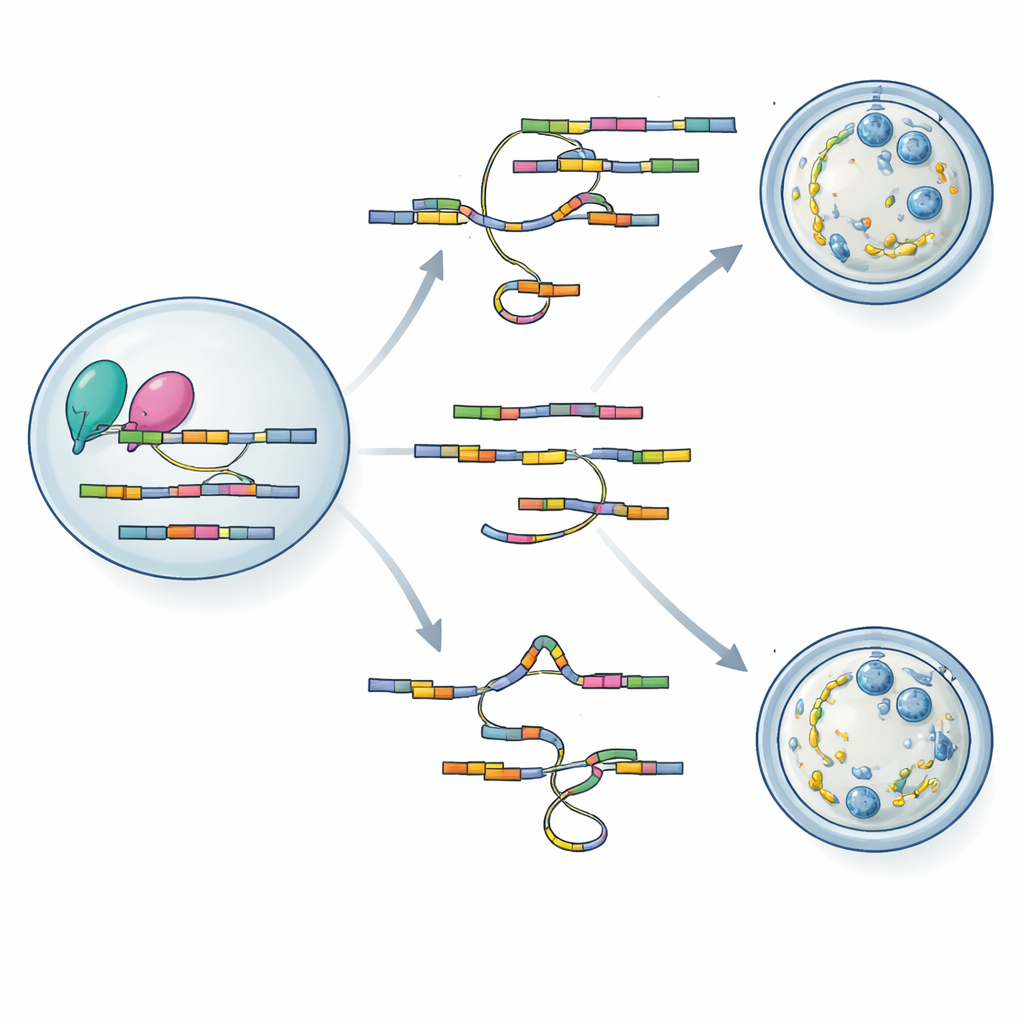
To understand how loss of hnRNPM leads to such sweeping defects, the team turned to two powerful sequencing tools adapted for tiny egg samples. Using SCAN-seq, which reads full-length RNA molecules from single oocytes, they discovered more than a thousand splicing changes in mutant eggs, including many previously unknown RNA variants. Genes involved in building the cytoplasmic lattice, controlling spindle behavior, and regulating the meiotic cell cycle were especially affected. A second method, LACE-seq, mapped exactly where hnRNPM binds on RNAs, revealing a preference for GU-rich stretches within exons. By overlaying the two datasets, the authors showed that hnRNPM sits directly on many of the messages whose splicing changes when the protein is missing, tying its binding activity to splicing accuracy.
A teamwork model for egg cell control
Protein interaction studies showed that hnRNPM physically associates with BCAS2 and with core components of the splicing machinery. The two partners bind many of the same RNA targets and often alter them in the same way when either protein is disrupted. Intriguingly, loss of hnRNPM reduced BCAS2 protein levels and weakened BCAS2’s grip on shared RNA targets, suggesting that hnRNPM not only edits messages but also helps stabilize its partner on those messages. The authors propose a model in which hnRNPM and BCAS2 form a cooperative complex that tailors key maternal RNAs during oocyte growth, ensuring proper assembly of the cytoplasmic lattice and reliable meiotic division.
What this means for fertility
In plain terms, this work shows that a molecular editing team—hnRNPM and BCAS2—acts behind the scenes to prepare egg cells for life after fertilization. When this team is missing, the egg’s internal scaffolding collapses, its chromosome-sorting machinery misfires, and even though the egg is produced in normal numbers, it cannot support embryo development. Because hnRNPM is highly similar in mice and humans, these findings point to a conserved quality-control system that could underlie certain forms of unexplained female infertility and offer new entry points for diagnosis or treatment.
Citation: Zhou, S., Liu, D., Gan, S. et al. hnRNPM cooperates with BCAS2 to modulate alternative splicing during oocyte development. Nat Commun 17, 2681 (2026). https://doi.org/10.1038/s41467-026-69176-8
Keywords: oocyte development, alternative splicing, female infertility, RNA-binding proteins, cytoplasmic lattice