Clear Sky Science · en
Kilowatt-scale alkali-cation-free CO2 electrolysis via accelerating mass transfer
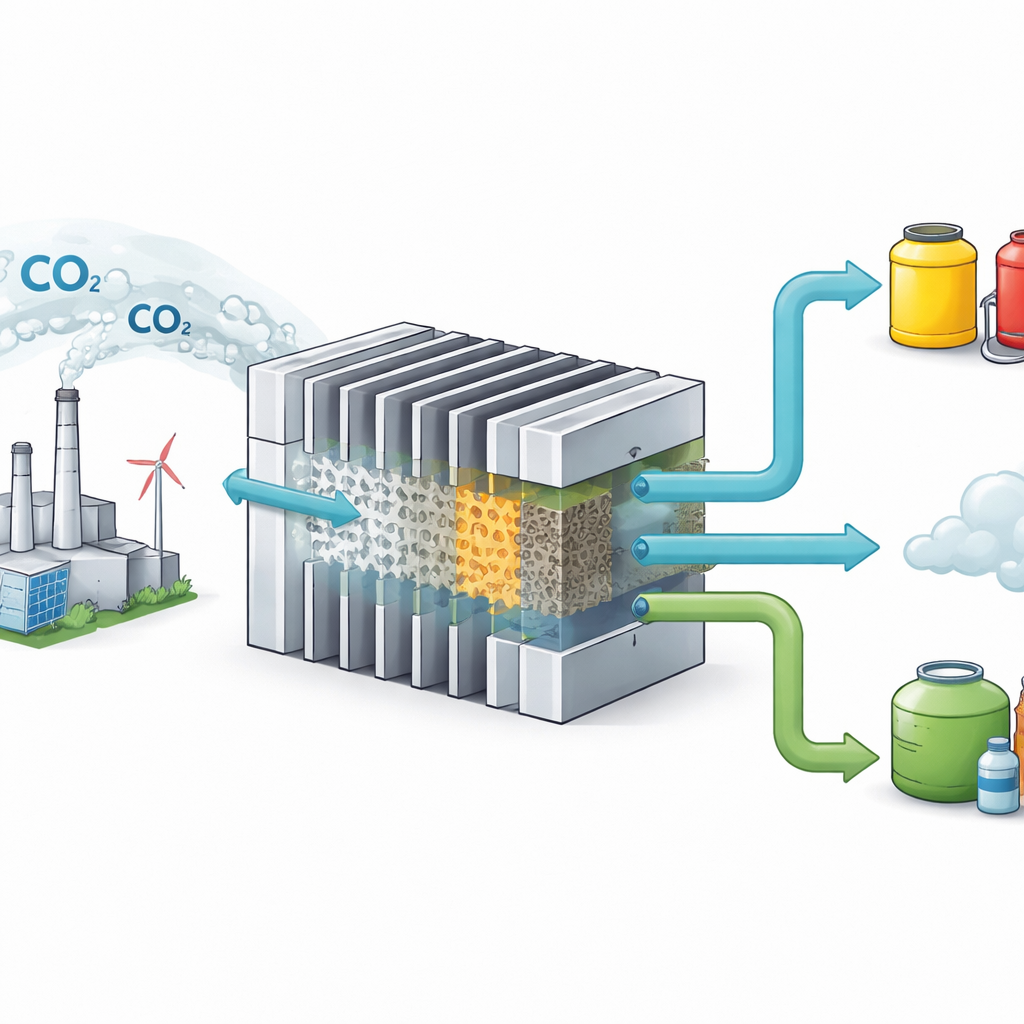
Turning a Climate Problem into Useful Products
Carbon dioxide (CO₂) from factories and power plants is a major driver of climate change, but it is also a cheap and abundant source of carbon. Scientists are developing devices that can “recycle” CO₂ into valuable fuels and chemicals using electricity from low‑carbon sources. This paper reports a key step toward running such CO₂‑to‑fuel machines at industrial scale, around the power level of a small neighborhood, while keeping them stable, efficient, and economically competitive.
Why Moving Molecules Matters
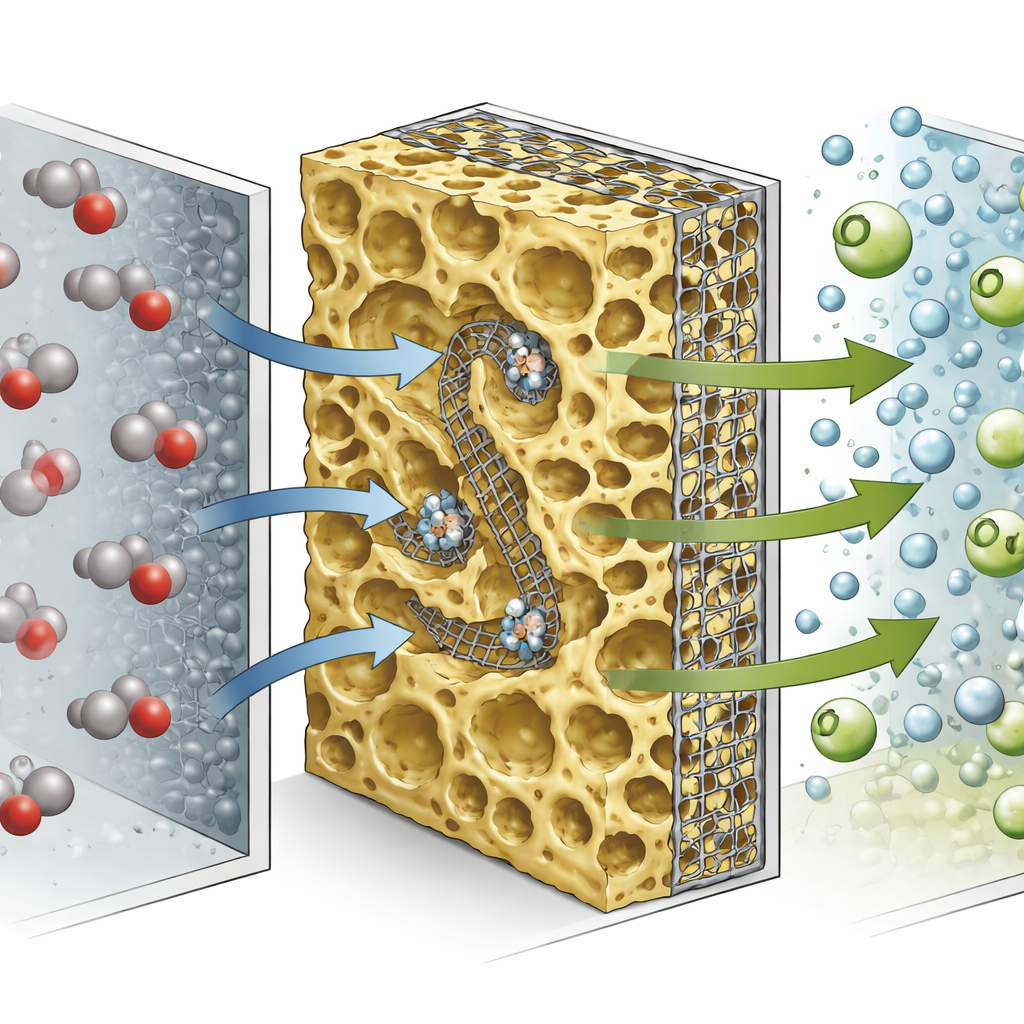
Modern CO₂‑to‑fuel devices push gas through thin, layered structures where CO₂ meets a catalyst and is turned into products such as carbon monoxide (CO) and ethylene (C₂H₄), a building block for plastics. For years, the main focus has been on inventing better catalysts. However, as the authors show, the larger bottleneck has become how quickly CO₂ and reaction products can move in and out of these layers—a challenge known as mass transfer. If CO₂ is fed too slowly, more of it is converted but the total output stays low. If it is fed quickly, the device makes plenty of product but wastes most of the CO₂. Conventional gas diffusion electrodes, built on thick carbon papers, trap gases in complex pathways and force a trade‑off between high selectivity for desired products and high CO₂ conversion.
A New Highway for Carbon Dioxide
To break this trade‑off, the researchers redesigned the heart of the device—the gas diffusion electrode—into what they call a high‑diffusion‑flux gas diffusion electrode (HDF‑GDE). Instead of a catalyst layer coated on a separate carbon backing, the new design is essentially all catalyst, reinforced by a thin stainless‑steel mesh in the middle. Large, well‑connected pores and added water‑repelling material let CO₂ gas reach the active sites directly, without squeezing through an inert substrate. Tests with a specially engineered silver catalyst that turns CO₂ mainly into CO showed that this new electrode can run at industrial‑level current densities while keeping product selectivity very high. In a compact cell fed only with pure water on one side and CO₂ on the other—no added alkaline salts—the device reached about 400 milliamps per square centimeter with roughly 90 percent of the electric current going to CO, far better than earlier alkali‑free systems.
Scaling Up to Kilowatt Power
Promising lab cells often fail when scaled up, so the team built a full stack of six membrane‑electrode units, each about the size of a paperback book, to test real‑world performance. Using the silver‑based HDF‑GDEs, the stack operated at around 1.3 kilowatts of electrical power for more than 1,000 hours, converting about 81 percent of the incoming CO₂ into CO at a steady gas flow comparable to what a small industrial unit might see. Over that time, it produced roughly 144 kilograms of CO. The same design was then adapted to a copper catalyst that favors ethylene. In this configuration, a similar kilowatt‑scale stack ran for over 1,000 hours and yielded about 17 kilograms of ethylene, increasing CO₂‑to‑ethylene conversion by about 15‑fold compared with older electrode structures.
Peeking Inside the Process
To understand why the new electrodes worked so well, the authors combined detailed imaging, real‑time laser spectroscopy, and computer simulations. They found that the open, mesh‑reinforced catalyst layer supports faster gas transport than conventional carbon‑paper designs at both the microscopic and device scales. More CO₂ and key reaction intermediates cover the catalyst surface, while hydrogen—an undesired side product in this context—is suppressed. Simulations reveal that, although the CO₂ concentration eventually drops along the flow path as it is consumed, the overall “traffic” of carbon species through the HDF‑GDE is much higher, which in turn boosts both current and CO₂ conversion without needing to throttle the gas feed.
From Lab Bench to Business Case
Finally, the team evaluated whether such systems could make financial sense. Using performance data from their kilowatt‑scale stacks, they built a techno‑economic model that includes equipment costs, electricity prices, and recycling of unreacted CO₂. For CO production, the calculated cost is around 0.48 US dollars per kilogram—already below current market prices—and could drop further if the devices last several years and use affordable low‑carbon electricity. Ethylene is not yet cost‑competitive, mainly because selectivity is still modest, but the analysis shows that combining technology improvements with climate policies such as carbon pricing could make CO₂‑to‑ethylene viable as well. Overall, the study demonstrates that redesigning how gases move through electrochemical reactors can unlock both technical and economic progress, bringing carbon‑neutral chemical manufacturing closer to large‑scale reality.
Citation: She, X., Xu, Z., Ma, Q. et al. Kilowatt-scale alkali-cation-free CO2 electrolysis via accelerating mass transfer. Nat Commun 17, 2641 (2026). https://doi.org/10.1038/s41467-026-69175-9
Keywords: CO2 electrolysis, gas diffusion electrode, carbon-neutral fuels, electrocatalysis, mass transfer