Clear Sky Science · en
A multivalent adaptor mechanism drives the nuclear import of proteasomes
How Cells Keep Their Control Rooms Clean
Inside every cell, the nucleus acts like a command center, packed with DNA and the proteins that control which genes are turned on or off. To keep this control room in good working order, cells rely on powerful molecular "shredders" called proteasomes that break down worn-out or misbehaving proteins. This paper reveals how cells manage the surprisingly tricky task of hauling these bulky proteasomes through the narrow gateways into the nucleus at just the right time.
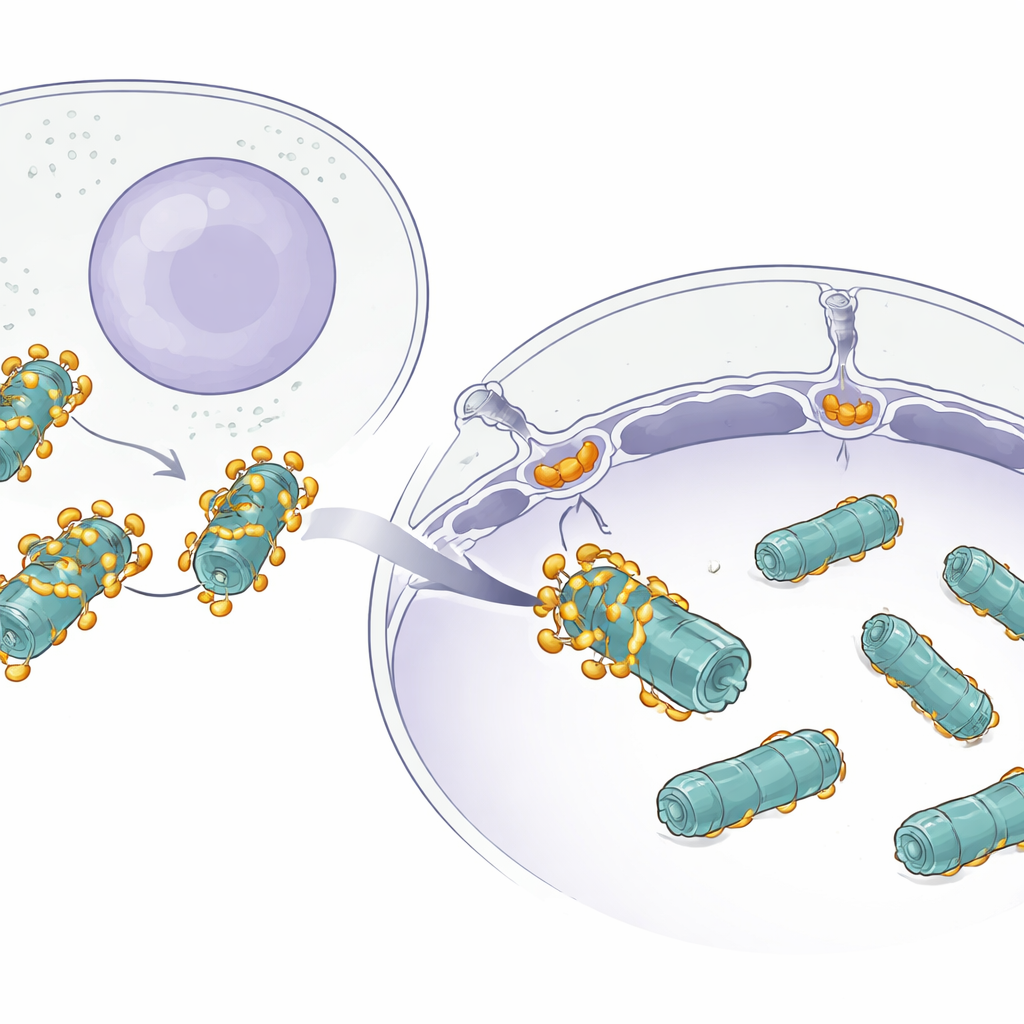
The Challenge of Moving Big Machines
Many proteins quietly shuttle between the main body of the cell and the nucleus through giant doorways in the nuclear envelope known as nuclear pores. Small proteins can slip through easily, but large machines like proteasomes are different: they are massive, barrel-shaped complexes built from many parts. While scientists already knew that small cargoes use helper proteins called importins to get into the nucleus, it was unclear how something as large as a proteasome squeezes through efficiently without getting stuck or lost.
A Shape-Shifting Helper Comes Into Focus
The team focused on a small, floppy protein called AKIRIN2, which earlier work had shown is absolutely required for proteasomes to enter the nucleus. Cells that divide without AKIRIN2 cannot bring proteasomes into the nucleus and quickly die because damaged nuclear proteins pile up. Using a combination of methods – including a huge mutational scan that altered nearly every amino acid in AKIRIN2, high-resolution cryo-electron microscopy to see the complex in 3D, and careful biochemical tests – the researchers mapped which parts of AKIRIN2 matter and what they do. They discovered a previously unappreciated “wing-like” segment and a coiled region that lets AKIRIN2 form dimers (pairs), both of which play key roles in gripping proteasomes and other partners.
Building a Multi-Handed Transport Handle
The structural images revealed that multiple copies of AKIRIN2 cluster on one side of the proteasome like many hands grabbing the same object. Each AKIRIN2 pair binds to specific spots on the proteasome’s outer surface while also engaging different importins. One importin, IPO9, holds onto AKIRIN2 in an unusual way, wrapping around it and changing shape as the complex assembles. Other importins, working in pairs of their own, recognize short nuclear-entry signals carried by AKIRIN2. Because several AKIRIN2 molecules sit on the same proteasome, they collectively display many of these signals at once. This multivalent “handle” allows a crowd of importins to latch on simultaneously, giving the huge proteasome enough pulling power to move through the nuclear pore.
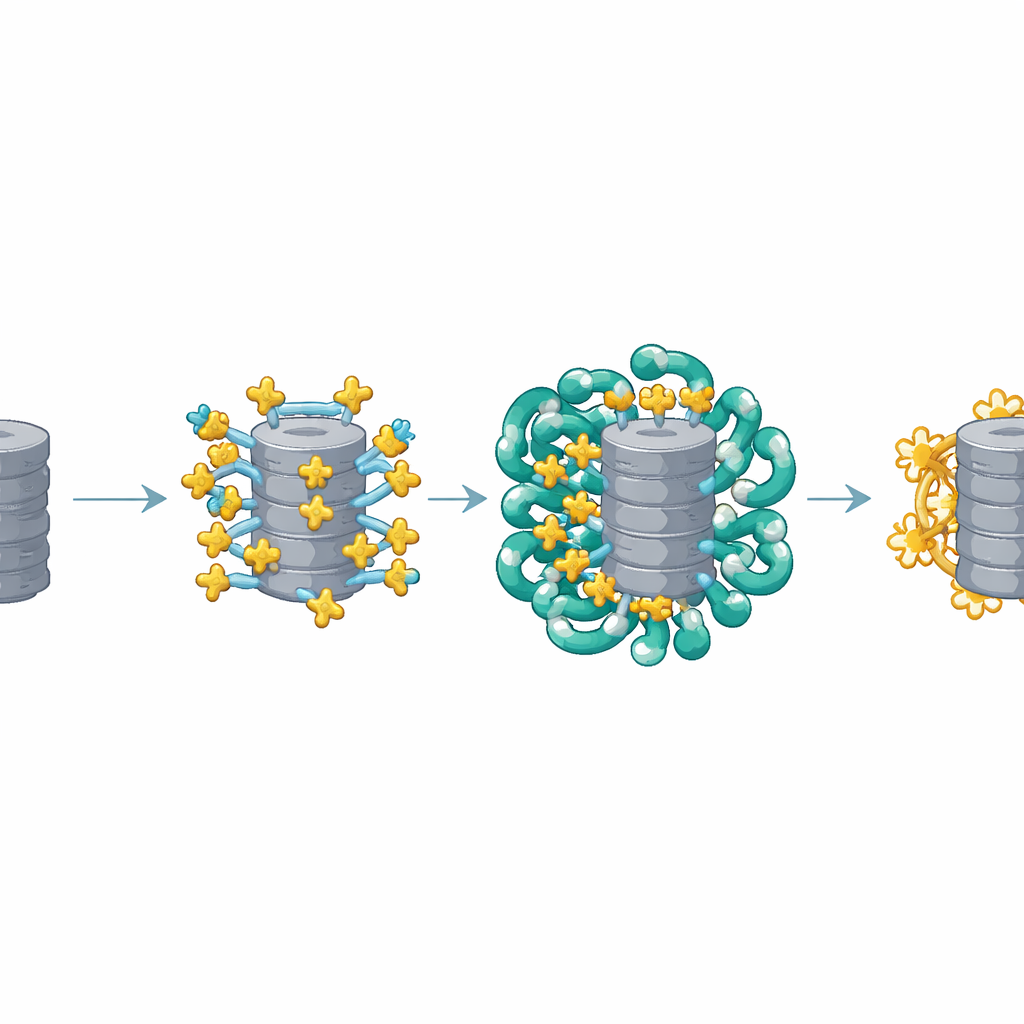
A Smart Assembly and Release Cycle
Reconstitution experiments in a simplified test-tube system showed that proteasomes only enter nuclei efficiently when both AKIRIN2 and importins are present together. The more AKIRIN2, the stronger and faster the import. Once inside the nucleus, the transport machinery must be cleared away so proteasomes can work. Here the cell uses a two-step release. A small switch protein called Ran, in its GTP-bound form, pries most importins off their cargo. AKIRIN2 and some IPO9 hang on more stubbornly, but the nucleus has a backup: nuclear proteasomes directly chew up AKIRIN2 itself, without the usual tagging with ubiquitin. Measurements across the cell cycle show that AKIRIN2 peaks during cell division, when new nuclei form, and then drops in early G1, consistent with this targeted cleanup.
Why This Matters Beyond One Protein
Together, these findings show that AKIRIN2 acts as a flexible scaffold that gathers many importins onto a single proteasome, turning a difficult transport problem into a coordinated group effort. By clustering nuclear-entry signals on an adaptor rather than on the proteasome itself, cells can control when and how strongly large machines move into the nucleus without redesigning those machines. Similar adaptor-based strategies appear to be used for other big protein assemblies, and even viruses exploit related tricks to invade the nucleus. Understanding this general transport logic deepens our picture of how cells maintain protein balance in their most sensitive compartment and may eventually point to new ways to intervene when protein breakdown or nuclear function goes awry.
Citation: Brunner, H.L., Kalis, R.W., Grundmann, L. et al. A multivalent adaptor mechanism drives the nuclear import of proteasomes. Nat Commun 17, 2359 (2026). https://doi.org/10.1038/s41467-026-69162-0
Keywords: nuclear transport, proteasome, AKIRIN2, importin, protein degradation