Clear Sky Science · en
An integrative structural biology approach reveals the dynamic organization of the R2SP quaternary chaperone complex
How Cells Build Complex Molecular Machines
Inside every cell, many proteins do not work alone—they must snap together into elaborate machines before they can do their jobs. Getting this assembly step right is vital for healthy breathing, movement, and even fertility. This paper explores one such assembly helper, a molecular "construction crew" called the R2SP complex, and shows how it is organized and how it works. Understanding R2SP matters because when its components fail, tiny hair‑like structures on cells called cilia do not move properly, leading to chronic respiratory problems and other diseases.
The Cellular Construction Crews R2TP and R2SP
Cells rely on teams of helper proteins, known as chaperone complexes, to guide other proteins into the correct multi-part shapes. One long‑studied team, called R2TP, is found in many tissues and helps assemble large machines such as RNA‑making factories and DNA‑damage sensors. R2SP is a close cousin of R2TP: both are built around the same ring‑shaped motor made of two partner proteins, RUVBL1 and RUVBL2, which burn the energy molecule ATP. What differs are the accessory adapters that plug clients into this motor. R2TP uses adapters called RPAP3 and PIH1D1, while R2SP uses SPAG1 and PIH1D2. These small differences in parts give the two complexes different client lists and link R2SP in particular to the construction of the moving cilia that sweep mucus and fluids across tissues.
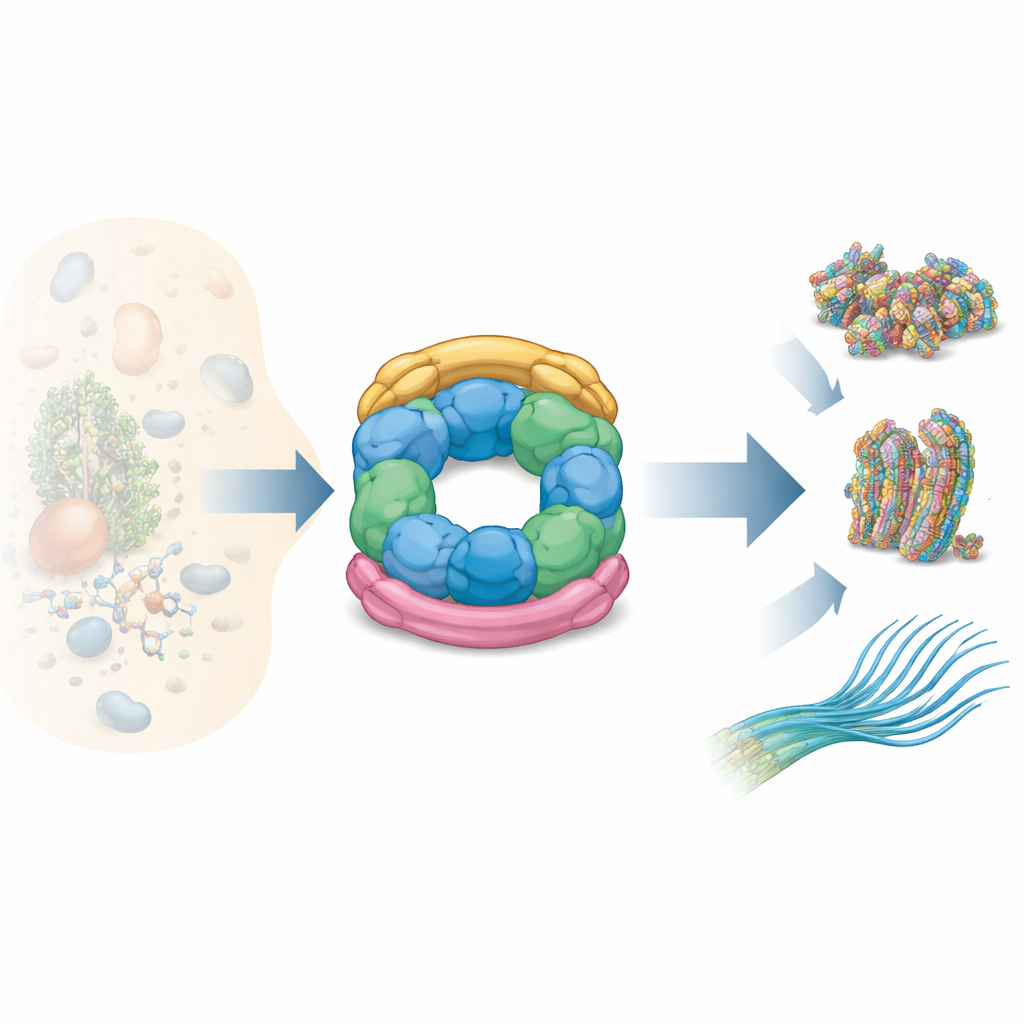
How the R2SP Parts Fit Together
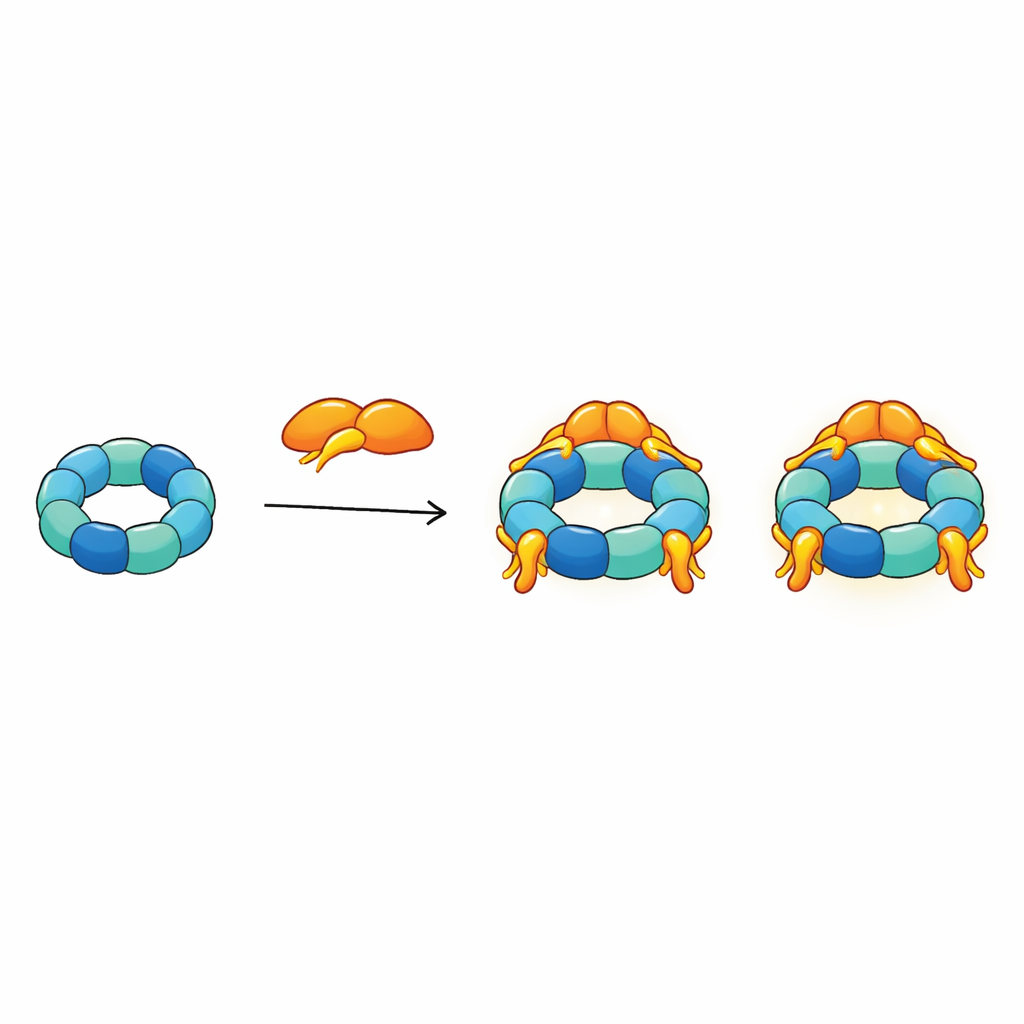
The authors combined several structural techniques—nuclear magnetic resonance, cryo‑electron microscopy, and cross‑linking mass spectrometry—with biochemical tests to map how R2SP is built. They showed that the tail end of SPAG1 grips the RUVBL1/RUVBL2 ring in a way that resembles how RPAP3 binds in R2TP, but with key tweaks in shape and contact points. A second adapter, PIH1D2, nestles under the ring and also reaches up to touch flexible side domains of the motor. These side domains act like hinged arms that help transmit movements from the ATP‑burning core to bound clients. The data reveal that SPAG1 and PIH1D2 do not attach independently: they cooperate, forming a coupled unit that clamps the ring from above and below, stabilizing a distinctive three‑dimensional architecture.
A Dynamic Ring That Switches Gears
Beyond static pictures, the team asked how R2SP behaves. Using interaction measurements, they found that the RUVBL1/RUVBL2 motor can exist as either a single ring or a stacked double ring. When SPAG1 and PIH1D2 bind, they shift this balance strongly toward the single‑ring form and can decorate it with up to three adapter pairs. At the same time, the adapters markedly boost the motor’s ATP‑burning activity, and they change how quickly ATP‑like molecules bind and unbind from the active sites. Compared to the R2TP system, SPAG1 and PIH1D2 favor different steps in nucleotide release, suggesting that R2SP tunes the energy cycle of the core motor in its own way. This finely controlled energy use is thought to drive the stepwise assembly of client complexes needed for motile cilia.
A Flexible Platform for Building Cilia Components
By integrating all their structural restraints into a single model, the authors propose that R2SP acts like a flexible, many‑armed platform. The SPAG1 tail anchors firmly on the top of the ring, while its other regions and the two domains of PIH1D2 extend toward the more open, arm‑like side of the motor. This side is where client proteins and helper chaperones such as HSP70 and HSP90 most likely dock. Because up to three SPAG1–PIH1D2 units can attach to one ring and the flexible arms constantly move, the whole complex resembles an octopus whose tentacles can grab several client pieces at once, coordinate them, and pass them on to be built into larger ciliary structures.
What This Means for Health and Disease
For non‑specialists, the main message is that R2SP is a specialized version of a general cellular construction machine, rewired by swapping just two adapter proteins. This swap changes both how the central motor is controlled and how clients are positioned, explaining why R2SP focuses on building parts for motile cilia while R2TP serves other cellular factories. Faults in SPAG1 are already known to cause primary ciliary dyskinesia, a disorder marked by chronic infections and fertility problems. By revealing R2SP’s detailed layout and working principles, this study lays the groundwork for understanding how specific mutations disrupt cilia assembly and may eventually guide targeted treatments that restore or modulate this crucial molecular construction crew.
Citation: Santo, P.E., Chagot, ME., Gizardin-Fredon, H. et al. An integrative structural biology approach reveals the dynamic organization of the R2SP quaternary chaperone complex. Nat Commun 17, 2605 (2026). https://doi.org/10.1038/s41467-026-69157-x
Keywords: molecular chaperones, protein complex assembly, motile cilia, structural biology, RUVBL1 RUVBL2