Clear Sky Science · en
Structural basis of TACO1-mediated efficient mitochondrial translation
Keeping the Cell’s Power Plants Running Smoothly
Mitochondria are often called the power plants of our cells because they make the energy molecule ATP. To do this, they must build a small but vital set of proteins using their own specialized protein factories, called mitoribosomes. This study uncovers how a little-known helper protein, TACO1, fine‑tunes these factories so they can keep running smoothly, especially when they encounter particularly “awkward” stretches in the protein chain that tend to make the machinery stall.
Energy Factories with Their Own Hardware
Unlike most protein production, which happens in the watery interior of the cell, mitochondrial protein making takes place on mitoribosomes anchored to the inner membrane of the mitochondrion—the same membrane that hosts many energy‑producing enzymes. Using state‑of‑the‑art cryo‑electron microscopy on intact human mitochondria, the authors obtained near‑atomic snapshots of these mitoribosomes in their natural setting. They saw that all of the mitoribosomes they captured were attached to the inner membrane and positioned so that newly made protein chains could feed directly into a membrane insertase, a kind of molecular slot that threads fresh proteins into the membrane where they are needed for energy production. 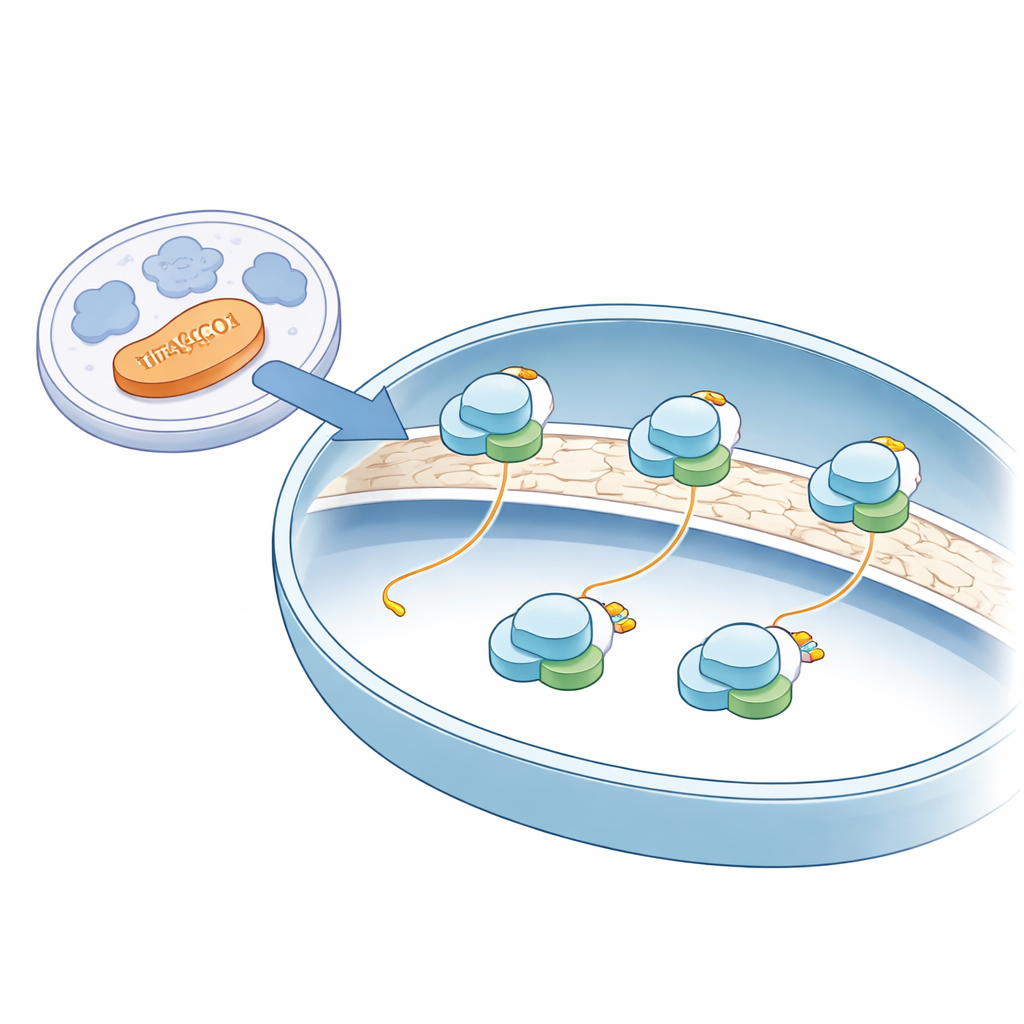
A Hidden Helper Comes into Focus
Amid the ribosomal components, RNA strands, and growing protein chains, the researchers noticed an extra patch of density that had never been seen in earlier, more artificial preparations. By carefully refining their maps and comparing shapes with predicted protein structures, they identified this mystery component as TACO1, a protein already linked to human mitochondrial disease but whose physical role had remained obscure. The images revealed that TACO1 nestles beside the part of the ribosome where the next amino acid–carrying RNA (the A‑site tRNA) arrives, making contacts with both ribosomal RNA and several ribosomal proteins. This positioning allows TACO1 to act almost like a brace, supporting the incoming tRNA as the ribosome forms a new bond to lengthen the protein chain.
Preventing Traffic Jams on the Protein Assembly Line
Protein building moves in a cycle: an elongation factor (mtEF‑Tu) delivers a loaded tRNA, the ribosome checks and uses it, and the factor departs so the next step can proceed. The authors found that TACO1 binds in roughly the same region that mtEF‑Tu uses, and that the two factors cannot occupy the ribosome at the same time. In normal cells, many mitoribosomes appeared in a state where the new tRNA is fully in place and elongation is progressing. When the team examined mitochondria from cells lacking TACO1, they instead saw an accumulation of ribosomes stuck in an earlier state in which mtEF‑Tu is still attached and the new tRNA has not fully settled. Biochemical experiments confirmed that, without TACO1, mtEF‑Tu lingers longer on actively translating ribosomes, and the large and small subunits are more likely to drift apart—signs of stalled or failed translation. 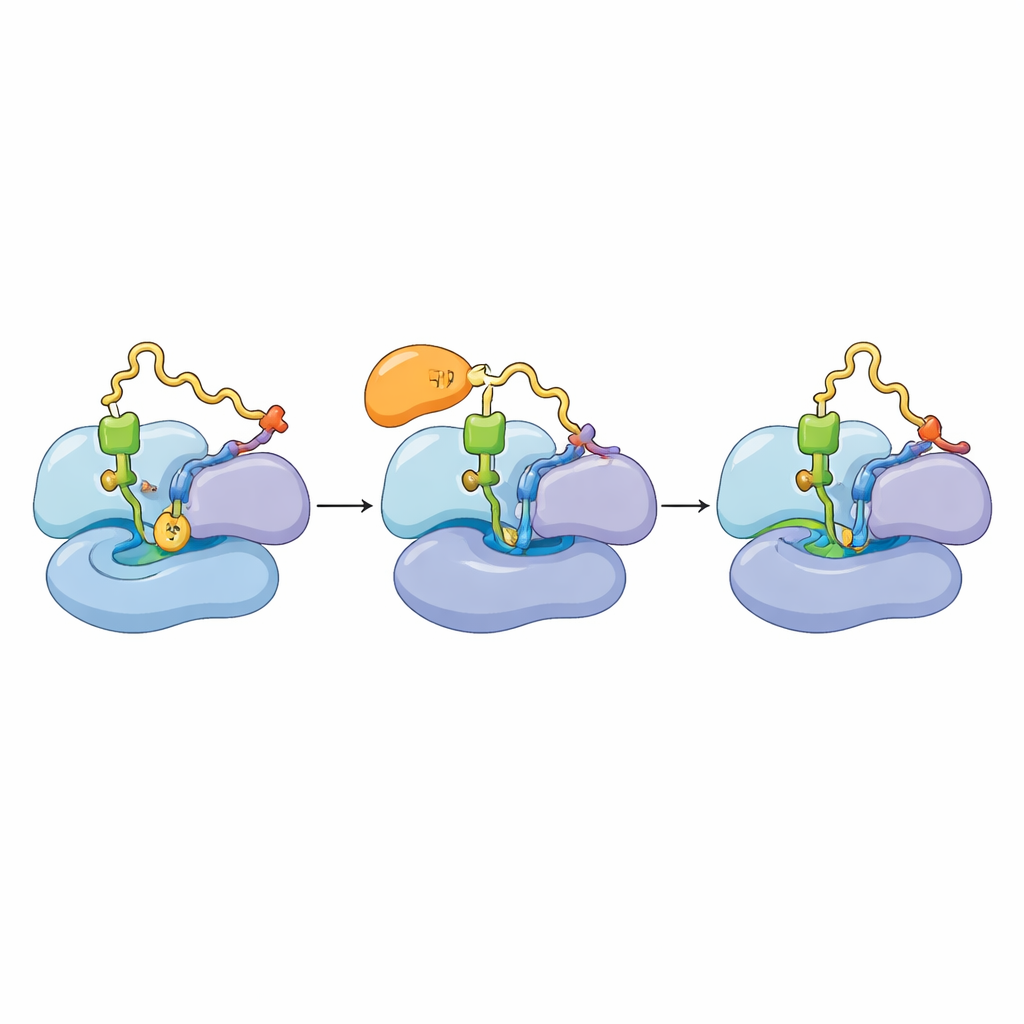
Why Certain Protein Stretches Are So Troublesome
One of TACO1’s most important jobs appears to be helping the ribosome work through runs of the amino acid proline, which naturally slow down bond formation because of proline’s rigid ring structure. Earlier work showed that TACO1 is crucial for producing cytochrome c oxidase subunit 1, a core energy‑chain component that contains a rare triple‑proline motif, and that its loss causes a breakdown of this energy complex and a human brain disorder called Leigh syndrome. In bacteria and in the cell’s main cytoplasmic system, different helper factors perform a similar anti‑stall function, but mitochondria lack those conventional factors. This new structural work shows that TACO1 has evolved as a mitochondria‑specific solution: by pushing aside mtEF‑Tu, stabilizing the incoming tRNA, and supporting bond formation, it helps the mitoribosome push past these difficult sequence patches instead of grinding to a halt.
An Ancient Strategy with Modern Health Implications
Beyond human cells, related proteins to TACO1 are found in many bacteria and other organisms, and some of these bacterial versions have now been shown to relieve ribosome stalling as well. This conservation suggests that using a TACO1‑like factor to rescue slow or stuck ribosomes is an ancient strategy for keeping protein synthesis efficient. For people, the work gives a concrete, visual explanation of why mutations in TACO1 can so severely damage energy production and the nervous system, and it hints that manipulating TACO1 or its relatives might one day offer ways to adjust mitochondrial translation—either to treat mitochondrial disease or to target harmful bacteria that depend on similar stalling‑relief systems.
Citation: Wang, S., Brischigliaro, M., Zhang, Y. et al. Structural basis of TACO1-mediated efficient mitochondrial translation. Nat Commun 17, 2521 (2026). https://doi.org/10.1038/s41467-026-69156-y
Keywords: mitochondrial translation, TACO1, mitoribosome, ribosome stalling, Leigh syndrome