Clear Sky Science · en
Combined bacteriophage and antibiotic therapy for refractory peritoneal dialysis-related peritonitis caused by Klebsiella pneumoniae
Why this infection story matters
People whose kidneys have failed often rely on peritoneal dialysis, a home-based treatment that gently cleans the blood using the lining of the abdomen. A serious complication of this therapy is peritonitis—an infection inside the belly—that can be painful, hard to treat, and even life-threatening. As some bacteria become harder to defeat with standard drugs, doctors are searching for new ways to protect these vulnerable patients. This article describes how a team of clinicians and scientists used a virus that attacks bacteria, together with antibiotics, to successfully treat a stubborn infection in a man on peritoneal dialysis, and what they learned from closely tracking his response.
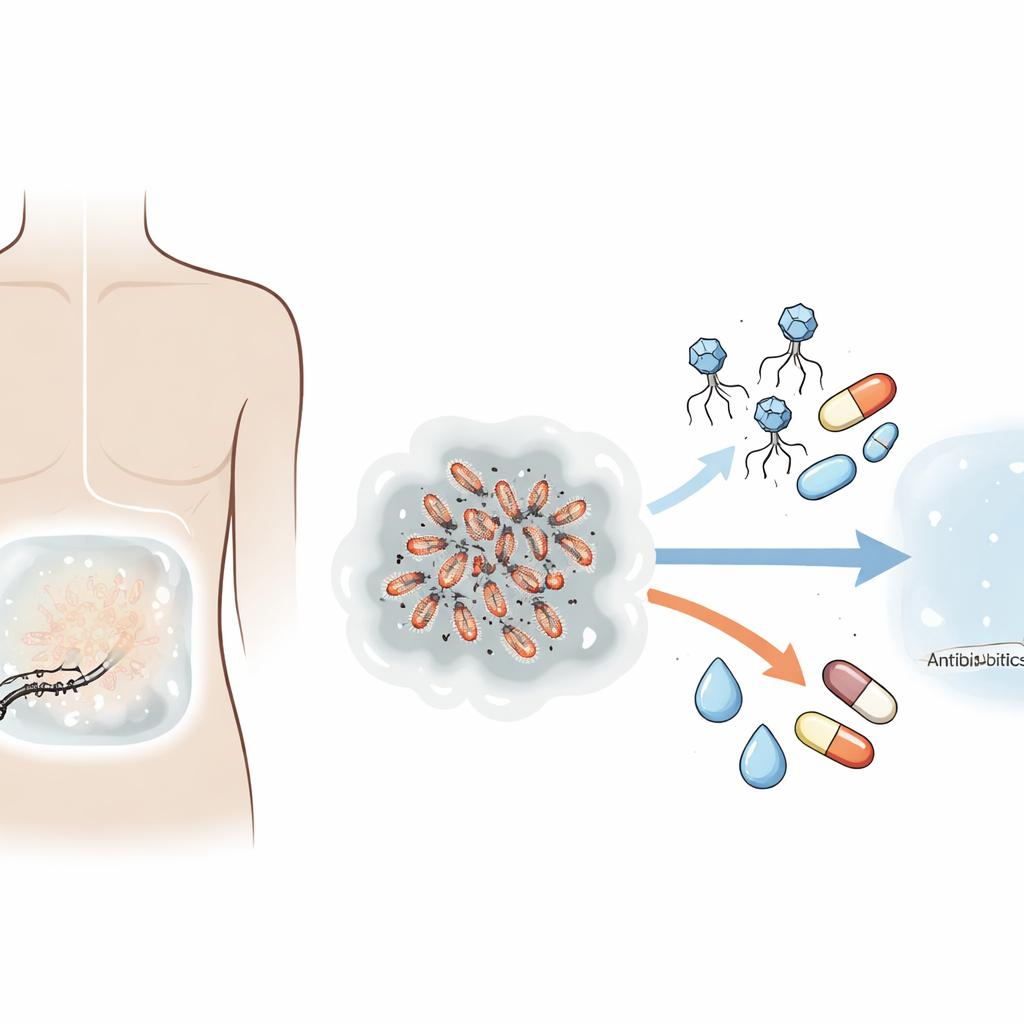
A hard-to-treat belly infection
The patient was a 71-year-old man with kidney failure managed by continuous ambulatory peritoneal dialysis. Over several years he had repeated bouts of peritonitis, first caused by common skin bacteria that can cling to the plastic catheter and form a slimy film, or biofilm. To break this cycle, doctors replaced his peritoneal dialysis catheter in a single procedure once the fluid drained clear, a strategy called simultaneous catheter replacement. Shortly afterward, however, he developed a new and more worrisome infection: the dialysis fluid again turned cloudy, and tests revealed Klebsiella pneumoniae, a Gram-negative bacterium linked to more severe illness, higher chances of losing the catheter, and greater risk of death in dialysis patients.
When antibiotics are not enough
Laboratory testing showed that this Klebsiella strain remained sensitive to a few powerful antibiotics, including amikacin and imipenem. Doctors delivered these drugs directly into the abdominal cavity for two weeks, along with other supportive medicines and a pill to guard against fungal infections. Despite this, the fluid stayed cloudy and white blood cells—immune cells that surge during infection—remained very high. Bacteria continued to grow in cultures, meeting the definition of refractory peritonitis, where guidelines usually recommend removing the catheter and switching the patient to hemodialysis. Because traveling regularly for hemodialysis would have been extremely difficult, the team explored an experimental alternative: bacteriophage therapy targeted specifically to his Klebsiella.
Harnessing viruses that hunt bacteria
Bacteriophages, or phages, are viruses that infect and kill bacteria but do not attack human cells. From a large phage library, the researchers selected two phages that recognized the patient’s own Klebsiella strain. Genetic sequencing confirmed that these phages were purely lytic—built to destroy, not quietly coexist with, their bacterial hosts—and that they lacked genes linked to toxins or antibiotic resistance. In the lab, one phage alone rapidly killed the bacteria but allowed resistant survivors to emerge within hours. The second phage could mop up these escapees. Used together as a cocktail, they suppressed bacterial growth for a full day or longer. When combined with low doses of imipenem, the phage cocktail cleared bacteria more strongly than either treatment alone, revealing a clear synergy between the virus mixture and the antibiotic.
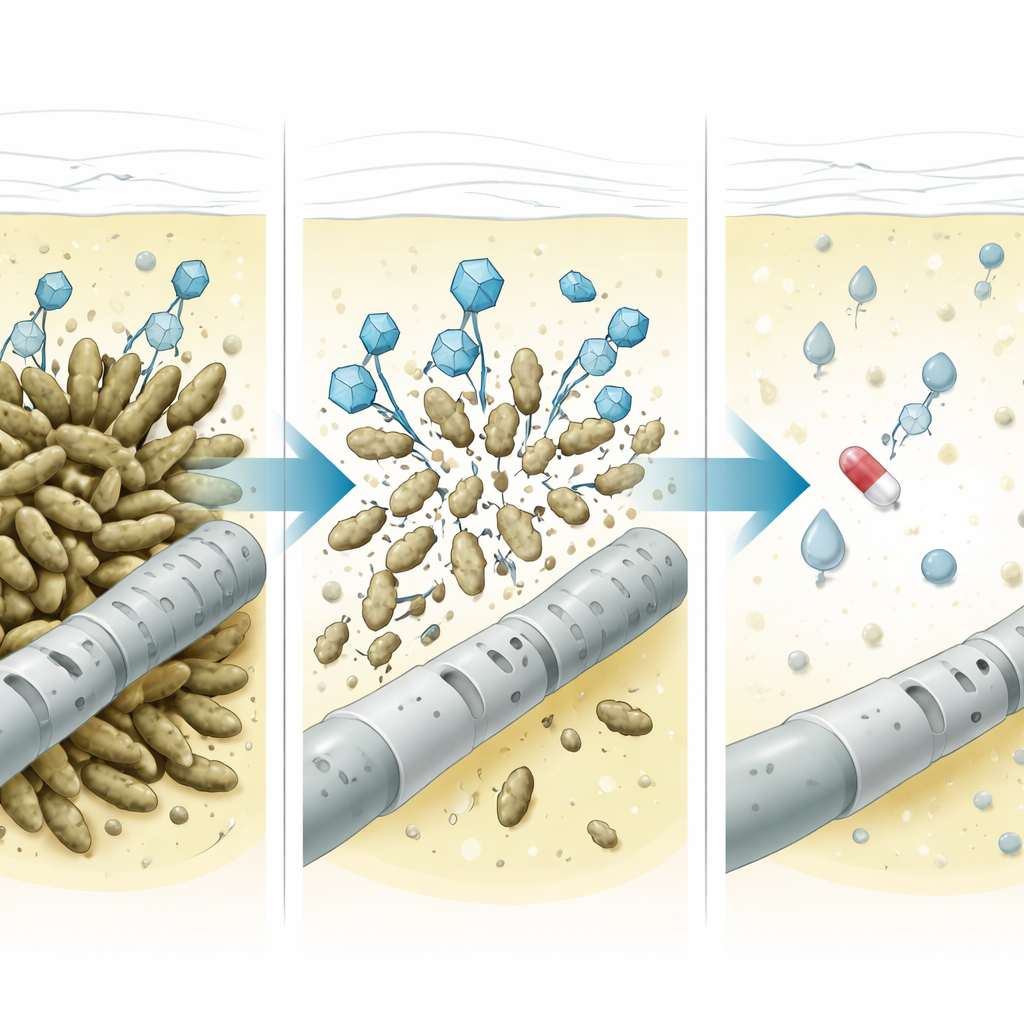
What happened inside the abdomen
Doctors infused the phage cocktail into the peritoneal cavity twice daily for three days while continuing intraperitoneal imipenem and oral fluconazole. They then carefully followed several signals in the drainage fluid. White blood cell counts initially fluctuated but soon plummeted, and Klebsiella stopped growing in culture. Interleukin-6, a protein that reflects local inflammation, started very high, spiked briefly during phage treatment—likely as bacteria burst and released their contents—and then steadily declined to low levels. Lipopolysaccharide, a component of the outer coat of Gram-negative bacteria and a strong trigger of immune reactions, showed a similar pattern: high before therapy, a small bump during bacterial breakdown, then a fall to undetectable levels. The team also looked for neutralizing antibodies—immune molecules that might block the phages—and found no meaningful increase in the peritoneal fluid. Phage levels themselves dropped by about ten-thousand-fold within a day and became undetectable within 48 hours, consistent with the viruses doing their job and then disappearing.
Clues from other patients
To put this story in context, the authors reviewed all published cases of human infections caused by Klebsiella pneumoniae that were treated with phages. They found 14 patients worldwide, with infections in the urinary tract, lungs, bones, joints, and brain coverings. In 13 of these, symptoms improved or the infection cleared, and only one case failed to respond. Most patients received phages alongside antibiotics, though a few were treated with phages alone. Across these reports, serious side effects were rare, supporting the idea that carefully prepared phage therapies can be safe when monitored closely.
What this could mean for future care
After three weeks of combined phage and antibiotic treatment, the patient’s abdominal infection resolved, his inflammation markers normalized, and he was discharged without any ongoing antibiotics. At a one-month follow-up, there was no sign of recurrence, and he was able to remain on peritoneal dialysis. This single case cannot prove that phage therapy will work for every dialysis patient with refractory peritonitis, and the authors emphasize the need for larger studies, better pathogen detection tools, and deeper exploration of how phages interact with the immune system. Still, their experience shows that personalized phage cocktails, thoughtfully paired with effective antibiotics, may offer a valuable rescue option when standard treatments are failing and catheter removal would be devastating for a patient’s quality of life.
Citation: Yang, X., Wu, N., Jiang, X. et al. Combined bacteriophage and antibiotic therapy for refractory peritoneal dialysis-related peritonitis caused by Klebsiella pneumoniae. Nat Commun 17, 2468 (2026). https://doi.org/10.1038/s41467-026-69154-0
Keywords: phage therapy, peritoneal dialysis, Klebsiella pneumoniae, antibiotic resistance, peritonitis