Clear Sky Science · en
Molecular mechanism of phosphate import by the bacterial PstSCAB transporter
Why tiny nutrients matter to us
Every living cell, from human neurons to soil bacteria, runs on phosphorus. This element helps store energy, build DNA, and keep cell membranes intact. Yet in the environment, usable phosphate—the form of phosphorus cells can actually import—is often scarce. Many disease-causing bacteria rely on an ultra-efficient molecular machine, called the PstSCAB transporter, to scavenge phosphate when it is in short supply. Understanding exactly how this machine works is not only a basic science question; it could open doors to new ways of disarming infections by starving bacteria of this vital nutrient.
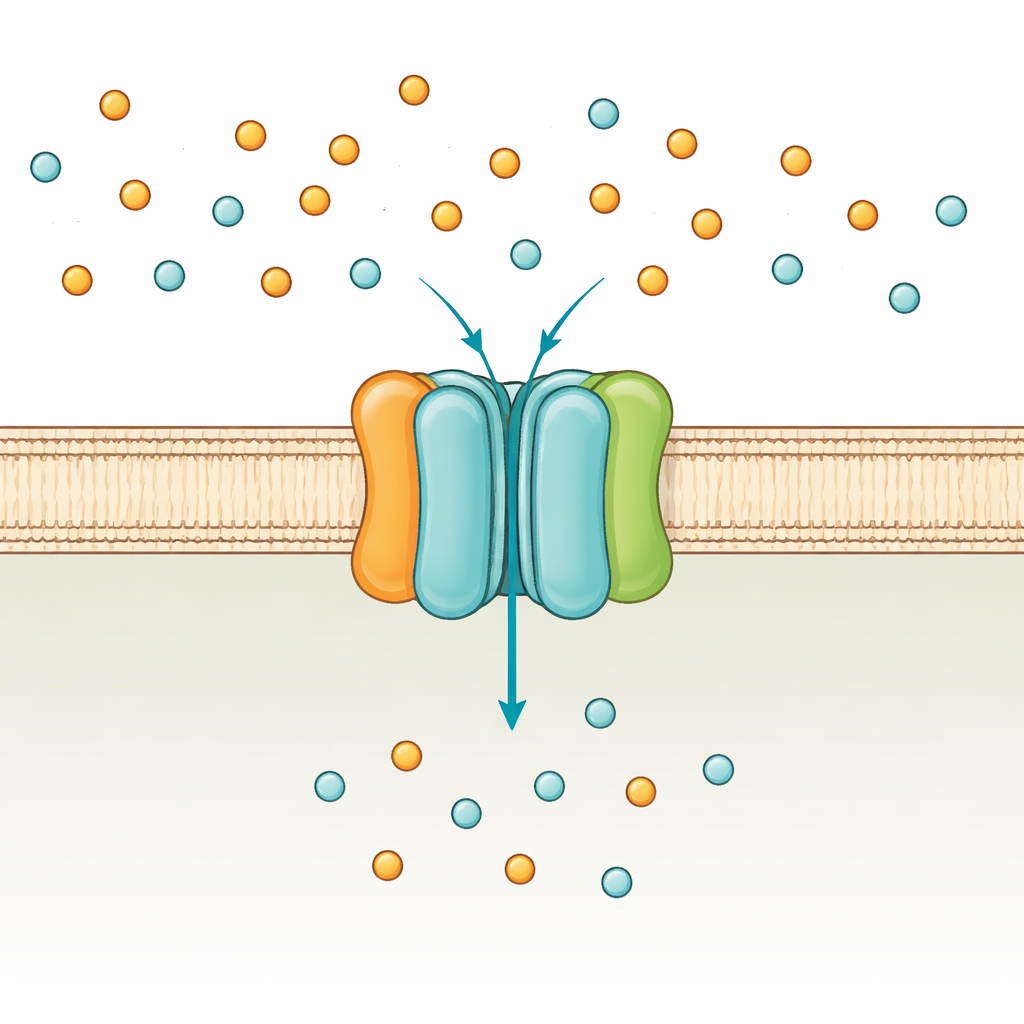
The cell’s phosphate gatekeeper
Bacteria such as Escherichia coli use two main routes to take up phosphate: a low-affinity system that works when phosphate is plentiful, and the high-affinity PstSCAB system that switches on under scarcity. PstSCAB sits in the cell membrane and is powered by ATP, the universal energy currency of the cell. It is built from five parts: two membrane-embedded subunits (PstA and PstC) that form the passageway, a soluble “catcher” protein outside the membrane (PstS) that grabs phosphate, and a pair of internal subunits (PstB) that burn ATP to drive transport. Because this system also helps control many genes linked to bacterial virulence, mutations in PstSCAB can weaken pathogens that cause urinary tract infections, sepsis in farm animals, and tuberculosis.
Capturing the machine in action
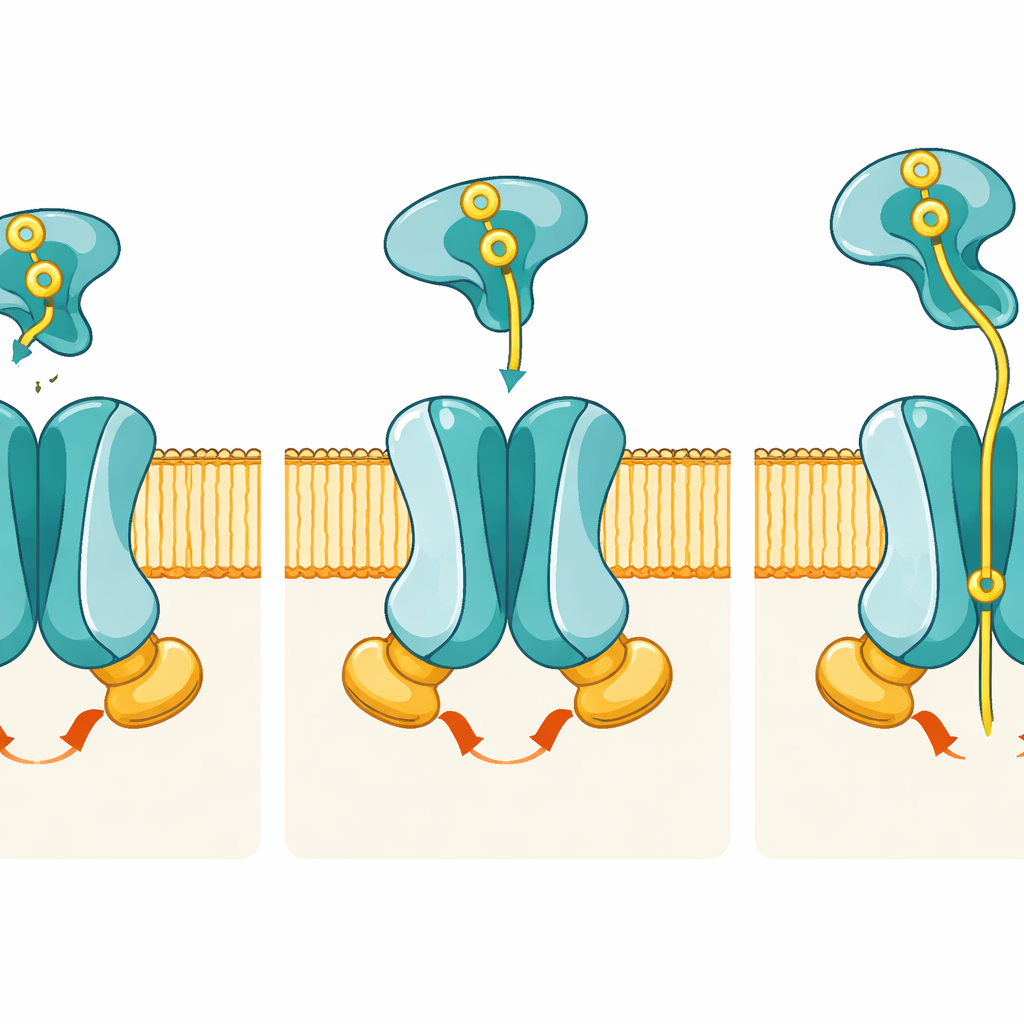
Until now, scientists had only partial views of PstSCAB, mostly of the catcher protein PstS alone. In this study, the researchers used high-resolution cryo-electron microscopy to visualize the entire transporter in several key steps of its working cycle. They reconstituted the bacterial proteins into artificial membrane “nanodiscs” and froze them so quickly that the molecules were trapped mid-motion. By carefully tweaking the proteins and adding or withholding ATP, they obtained structures of three main states: a resting inward-facing form without the catcher attached, a pretranslocation state where phosphate-loaded PstS docks onto the membrane complex, and an ATP-bound catalytic intermediate in which phosphate is held inside the transporter.
How shape changes move phosphate
The images reveal how coordinated shape changes move phosphate from outside to inside the cell. In the resting state, the passageway formed by PstA and PstC opens only toward the cell interior; the outer side is sealed by a set of “gate” residues, meaning phosphate cannot yet enter from outside. When phosphate-loaded PstS docks in the pretranslocation state, it nestles between flexible loops of PstA and PstC but, strikingly, the membrane subunits barely change shape. The real shift comes when PstS begins to open its two lobes to release phosphate and, in concert, the two PstB subunits swing together to bind ATP. This ATP binding clamps PstB into a dimer and pulls on connecting helices in PstA and PstC, flipping the entire pore into an outward-facing conformation that exposes a specific phosphate-binding pocket to the outside.
A custom pocket for phosphate
In the outward-facing, ATP-bound structure, the authors find a density in the middle of the membrane region that matches a phosphate ion. This ion is cradled by positively charged amino acids—particularly two arginines, one from PstA and one from PstC—that act like molecular fingertips gripping the negatively charged phosphate. Computer simulations show that phosphate remains stably bound in this pocket over microseconds, and genetic experiments confirm its importance: when these key residues are mutated, the transporter still burns ATP but imports phosphate very poorly. This separation of “where energy is spent” from “where cargo is recognized” highlights how precisely tuned the machine is for phosphate.
From structural snapshots to medical promise
Taken together, the structures and supporting biochemical tests outline a full cycle: PstS captures phosphate outside, docks to the inward-facing transporter, then—coupled to ATP binding and breakdown by PstB—triggers a flip that passes phosphate into the membrane pocket and finally into the cell interior. After ATP is hydrolyzed, the machine relaxes back to the resting state, ready for another round. For a lay observer, the key message is that we now have an atomic-level blueprint for how many bacteria survive in low-phosphate environments and regulate their virulence. This detailed map can guide the design of drugs, antibodies, or peptides that jam the transporter or block its catcher protein, potentially turning an essential nutrient gate into a vulnerable therapeutic target.
Citation: Xiao, H., Li, S., Qi, R. et al. Molecular mechanism of phosphate import by the bacterial PstSCAB transporter. Nat Commun 17, 2294 (2026). https://doi.org/10.1038/s41467-026-69153-1
Keywords: phosphate transport, bacterial nutrient uptake, ABC transporter, PstSCAB, antibacterial targets