Clear Sky Science · en
Molecular insights into the capsular polysaccharide transporter Wza-Wzc complex
How Bacteria Wear Invisible Armor
Bacteria that make us sick often protect themselves with a sugar-rich coat, or capsule, that helps them dodge our immune system and resist harsh conditions. This study uncovers, in atomic detail, how a key piece of machinery in common gut bacteria builds and exports that sugary armor. Understanding this process could point the way to new antibiotics and vaccines that strip pathogens of their protective shield instead of just trying to kill them outright.
The Sugar Shield Around Dangerous Germs
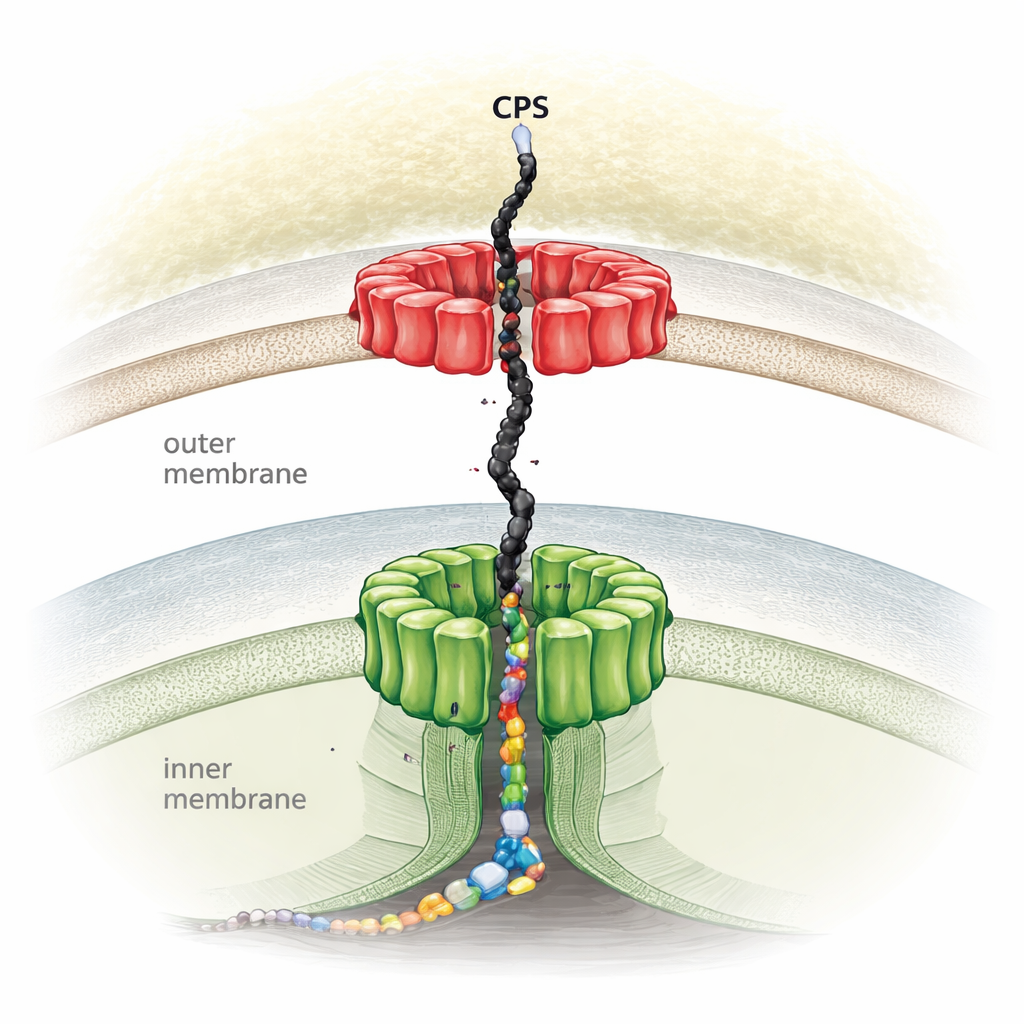
Many harmful bacteria surround themselves with capsular polysaccharides—long, branching chains of sugars that form a thick outer layer. This capsule helps them evade immune attack, resist antibiotics, and form resilient biofilms. The work focuses on Escherichia coli, a well-studied model organism that uses a widespread route called the Wzx/Wzy-dependent pathway to assemble these sugar chains. In this pathway, small repeat units of sugars are first built inside the cell, flipped across the inner membrane, linked together into long polymers, and finally pushed out to form the capsule. Although researchers knew the names of the main protein players, they lacked a complete picture of how they come together into a single machine that spans the entire bacterial envelope.
Revealing a Full-Length Molecular Tunnel
Using high-resolution cryo-electron microscopy, the authors solved the full three-dimensional structure of the Wza-Wzc complex—the core export machinery for capsular sugars in E. coli K12. They found that eight copies of each protein assemble into a tall, continuous channel that runs from the inner membrane to the outer membrane, bridging the watery space between them. Wza sits in the outer membrane as a rigid ring-shaped exit port, while Wzc forms a flexible tower anchored in the inner membrane. Together they create a roughly 250–360 angstrom-long tunnel wide enough to guide a large, flexible sugar polymer from where it is made at the inner membrane all the way to the cell surface without letting it diffuse away or leak. Mutating key contact points between Wza and Wzc completely stopped capsule production, confirming that this joint complex is essential for export.
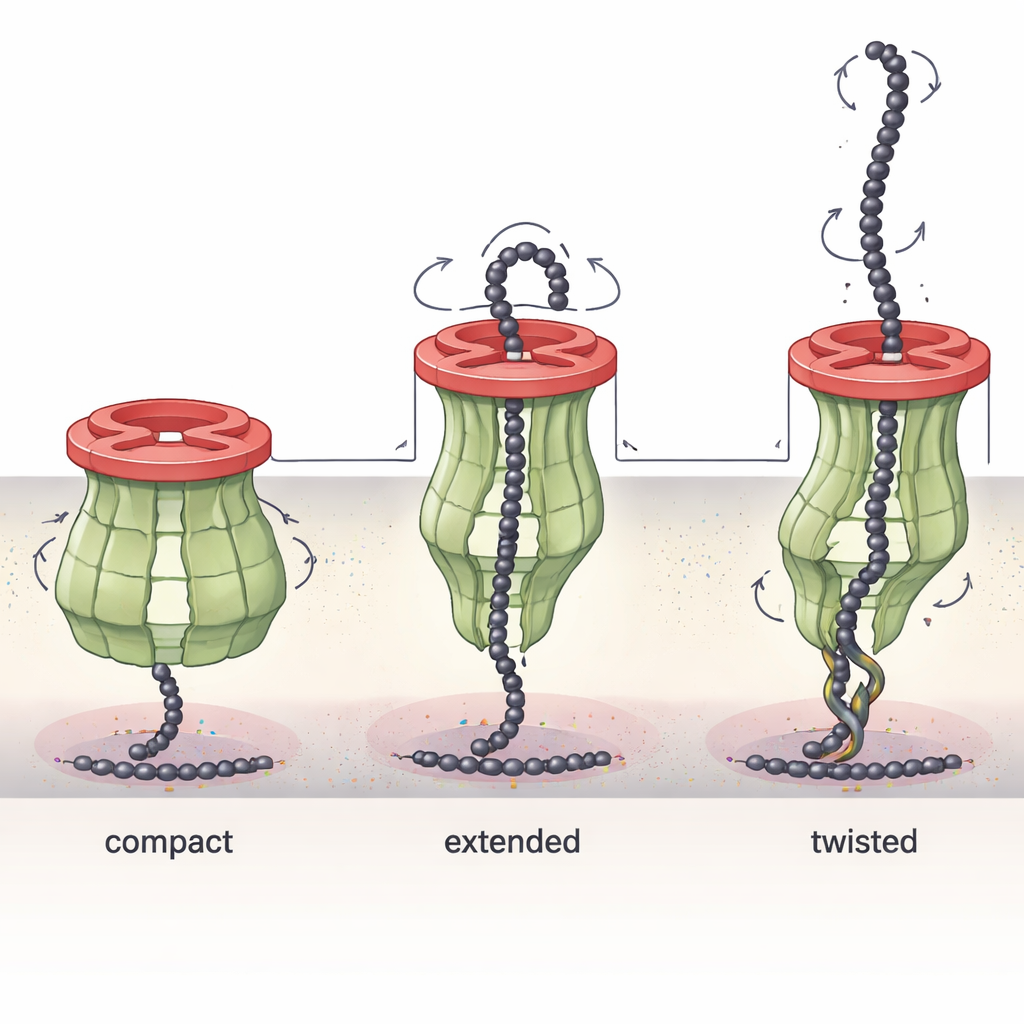
A Shape-Shifting Engine That Pulls Sugars Out
The Wzc part of the complex turned out to be remarkably dynamic. By capturing several structural “snapshots” under different chemical conditions, the researchers showed that Wzc’s long periplasmic arms can bend, twist, and extend like mechanical levers. In one state the channel is compact and tightly sealed; in others, Wzc has rotated and stretched, subtly changing the tunnel’s width and shape. In certain conformations the arms partly block the channel or contact Wza less tightly, and in extreme cases a single Wzc ring can engage two Wza rings at once. These observations support a model in which Wzc acts a bit like a molecular winch: its twisting and lengthening motions help pull the growing sugar chain away from the inner membrane and feed it into the outer-membrane pore, while also coordinating assembly and disassembly of the whole machine.
A Sugar-Sensing Landing Pad
Another puzzle was how the machinery recognizes the correct sugar building blocks among the many carbohydrates in and around the cell. Wzc contains a "jellyroll" domain positioned just above the inner membrane whose role had been mysterious. Structural comparisons with known sugar-binding proteins, combined with biochemical tests on arrays of purified carbohydrates, revealed that this domain can recognize specific sugar motifs similar to those found in the E. coli capsule. Removing the jellyroll region sharply reduced, but did not fully abolish, capsule production, suggesting it acts as a landing pad that captures newly flipped sugar units and guides them into a polymerization platform formed by Wzc together with the enzyme Wzy, which stitches the units into a long chain.
Coordinating Growth, Export, and Reset
Finally, the study links these structural features to a biochemical control system based on phosphorylation—the reversible addition of phosphate groups to a tyrosine-rich tail on Wzc. When Wzc is heavily phosphorylated, it exists mostly as loose individual units. As phosphates are removed by a partner enzyme, the kinase domains of Wzc assemble into an octamer, rearranging its arms so they can engage Wza and surround the polymerase Wzy. As the capsule chain grows and is pulled through the tunnel, further twisting and stretching of Wzc likely helps release the finished polymer into Wza for export. Once secretion is complete, Wzc re-phosphorylates itself, prompting the complex to fall apart and resetting the system for another round of capsule production.
Why This Matters for Fighting Infection
In everyday terms, this work shows, almost bolt by bolt, how bacteria build a molecular pipeline that threads long sugar chains from inside the cell to the outside, where they form a protective cloak. By mapping the structure and motions of the Wza-Wzc complex, and pinpointing sugar-recognition and control elements, the study highlights several weak spots that future drugs or vaccines might target. Disrupting this export machine would not necessarily kill bacteria outright, but it could strip them of their armor, leaving them far more vulnerable to our immune defenses and to existing antibiotics.
Citation: Yuan, B., Sieben, C., Raj, P. et al. Molecular insights into the capsular polysaccharide transporter Wza-Wzc complex. Nat Commun 17, 1436 (2026). https://doi.org/10.1038/s41467-026-69136-2
Keywords: bacterial capsule, polysaccharide secretion, Wza-Wzc complex, cryo-EM structure, antimicrobial targets