Clear Sky Science · en
An mRNA-delivered consensus allergen induces a neutralizing IgG response against food and pollen allergens
Why Many Allergies Might Be Treated at Once
For people who react not just to one food or pollen, but to many related ones, everyday life can feel like walking through a minefield. A bite of peach, a handful of nuts, or a spring breeze full of pollen can all trigger the same kind of dangerous response. This study explores a new way to calm such cross-allergies using a single, engineered protein delivered by mRNA—the same basic technology used in some COVID-19 vaccines—to train the immune system to block a whole family of related allergens at once.
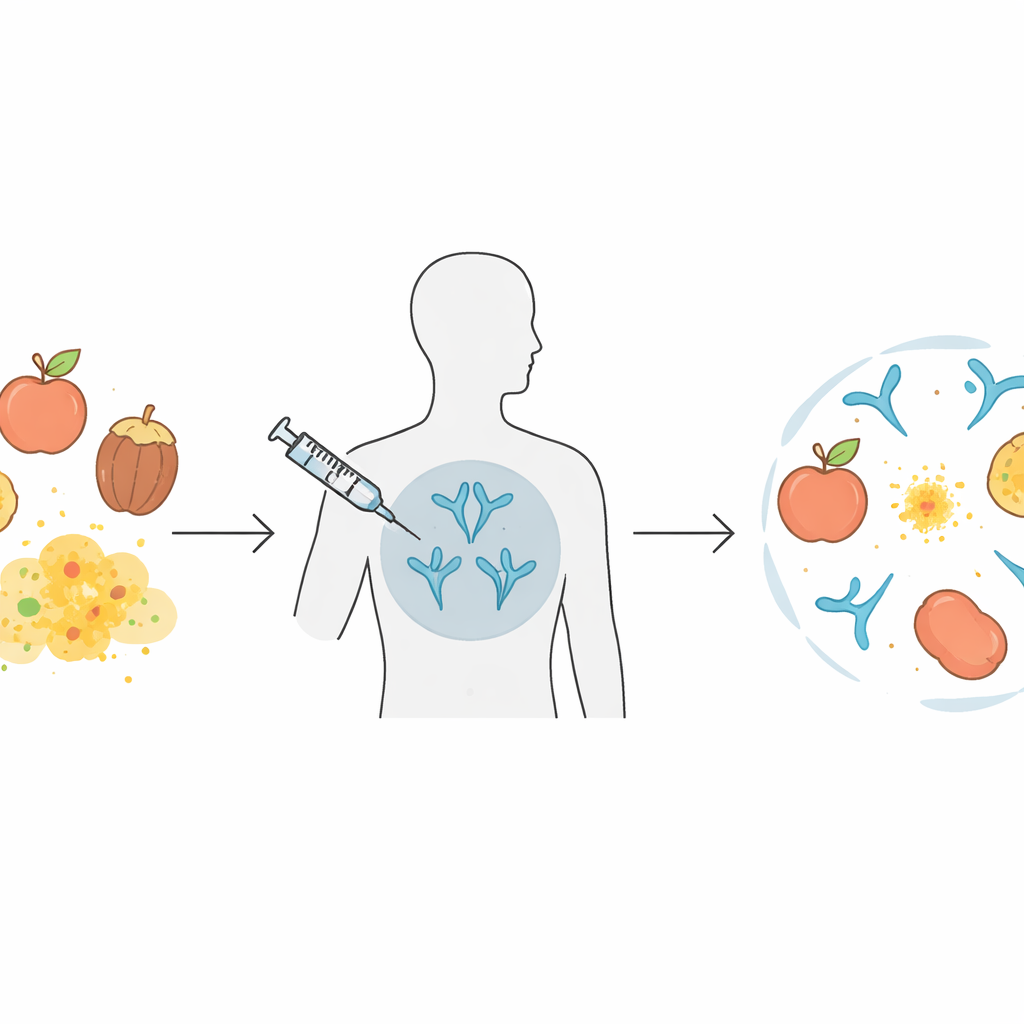
How One Allergy Can Turn Into Many
Many people with food allergies are not just allergic to one thing. They may react to several fruits, nuts, vegetables, and even pollens because these sources share similar proteins. In this work, the focus is on a family of plant proteins called non-specific lipid transfer proteins (nsLTPs), common in foods like peaches, apples, nuts, and in pollen from certain trees and weeds. In Mediterranean countries, so-called "LTP syndrome" can make patients react to a broad list of foods, sometimes with life-threatening anaphylaxis. Current allergy immunotherapies usually target extracts from a single source and may not cover all the cross-reactive cousins, which means long treatments and incomplete protection.
Designing a “Consensus” Allergen
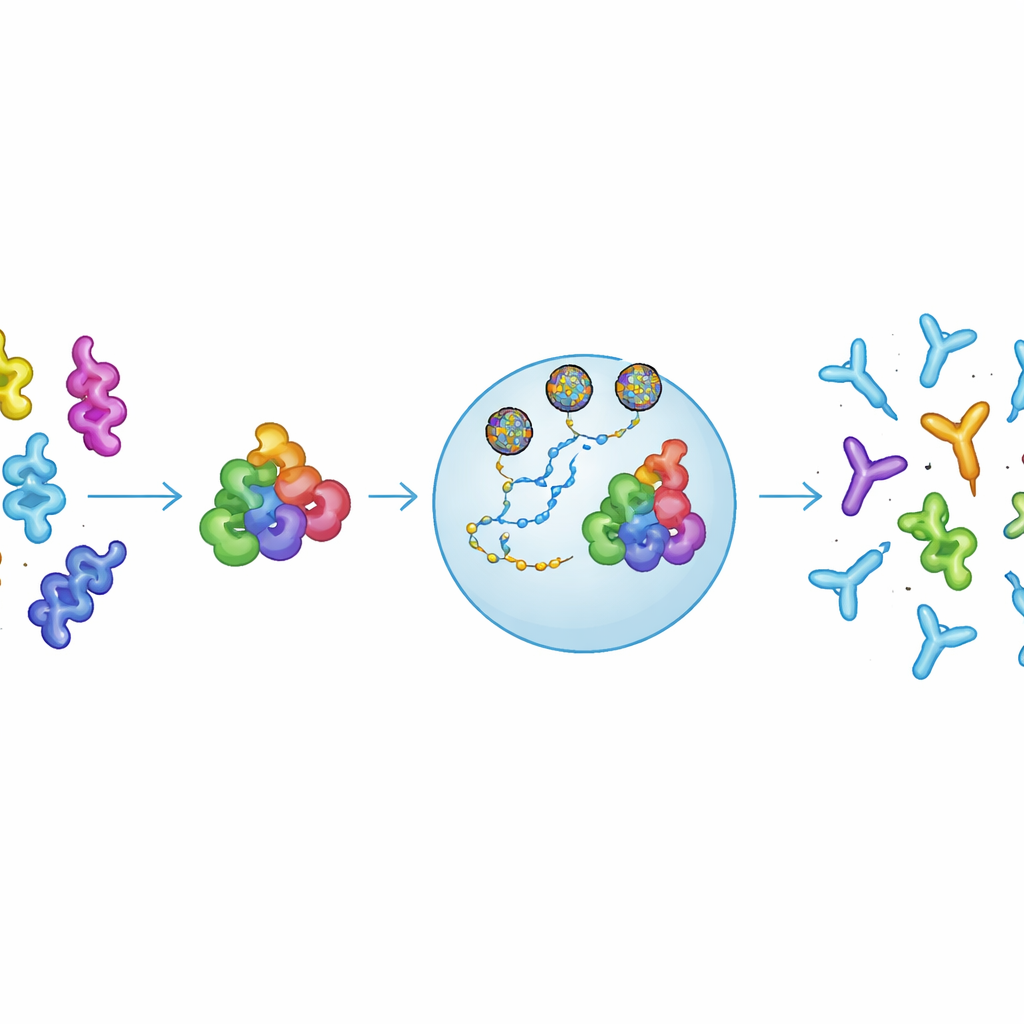
The researchers set out to design a single protein that mimics many different nsLTP allergens at once. They compared amino acid sequences from nsLTPs found in both foods and pollens and built a consensus version, called cnsLTP1, that does not copy any one natural protein but captures shared structural features across the family. Laboratory tests showed that this engineered protein folded like natural nsLTPs, stayed stable when heated and cooled, and, crucially, was recognized by IgE antibodies from patients already allergic to nsLTPs. That recognition indicates that cnsLTP1 presents the same key “allergy-triggering spots” as real-world allergens, which is essential if it is to retrain the immune system to respond differently.
Training the Immune System With mRNA and Protein Vaccines
The team then tested cnsLTP1 in mice using two different delivery methods: a traditional protein vaccine mixed with adjuvants, and an mRNA vaccine packaged in lipid nanoparticles, similar in composition to licensed mRNA vaccines. In naive mice, both approaches triggered strong antibody responses against cnsLTP1, particularly IgG subclasses known in mice to play a protective, allergy-blocking role. The mRNA-based vaccine tended to produce more of a certain IgG type and did so after fewer doses. Antibodies raised against cnsLTP1 bound not only to the engineered protein but also to a broad panel of different food and pollen nsLTPs, often more broadly than antibodies raised against single natural allergens like peach or pellitory nsLTPs. This means the consensus design successfully taught the immune system to recognize many related allergens.
Blocking Allergic Reactions in the Lab and Testing in Allergic Mice
To see whether these antibodies could actually interfere with allergic reactions, the researchers mixed mouse serum with allergens and then added human serum from patients with severe nsLTP allergies. They found that cnsLTP1-induced antibodies could block IgE from binding to multiple nsLTPs, and they reduced the degranulation of humanized basophil-like cells—a cell-based stand-in for allergic activation. In a more demanding test, the team used a mouse model already sensitized to peach nsLTP and prone to anaphylaxis. The mRNA vaccine was well tolerated and boosted IgG antibodies against several nsLTPs, but under the specific conditions tested it did not clearly reduce anaphylactic symptoms such as body temperature drop or markers of mast cell activation. This suggests that while the immune system was reshaped, the regimen was not yet strong or finely tuned enough to fully prevent severe reactions.
What This Could Mean for Future Allergy Care
The study provides a proof of concept that a single, engineered “consensus” allergen delivered by mRNA can trigger broad, cross-reactive, and functionally blocking IgG antibodies against a whole family of related food and pollen proteins. Although the current protocol did not yet protect allergic mice from anaphylaxis, the findings point to a flexible platform that might be improved by adjusting dose, adjuvants, or route of administration. In the long run, such consensus-based mRNA therapies could make it possible to treat clusters of related allergies—such as those driven by nsLTPs, other plant protein families, or even rapidly mutating pathogens—with shorter, more precise, and more broadly protective treatments.
Citation: Møiniche, M., Johansen, K.H., Parrón-Ballesteros, J. et al. An mRNA-delivered consensus allergen induces a neutralizing IgG response against food and pollen allergens. Nat Commun 17, 2402 (2026). https://doi.org/10.1038/s41467-026-69134-4
Keywords: food allergy, pollen allergy, mRNA immunotherapy, cross-reactivity, lipid transfer proteins