Clear Sky Science · en
MG53 mediates skeletal muscle-liver cross-talk and enhances alcohol metabolism in alcoholic liver disease
Muscles Helping the Liver
Excessive drinking can slowly poison the liver, leading to fatty buildup, scarring, and ultimately liver failure. At the same time, many heavy drinkers lose muscle mass and strength, a condition called muscle wasting. This study reveals that our muscles are not just bystanders in this story: they send out a protective protein that travels to the liver and helps it break down alcohol more safely. Understanding this hidden teamwork between muscle and liver may open new ways to treat alcoholic liver disease, a condition for which effective therapies are still scarce.
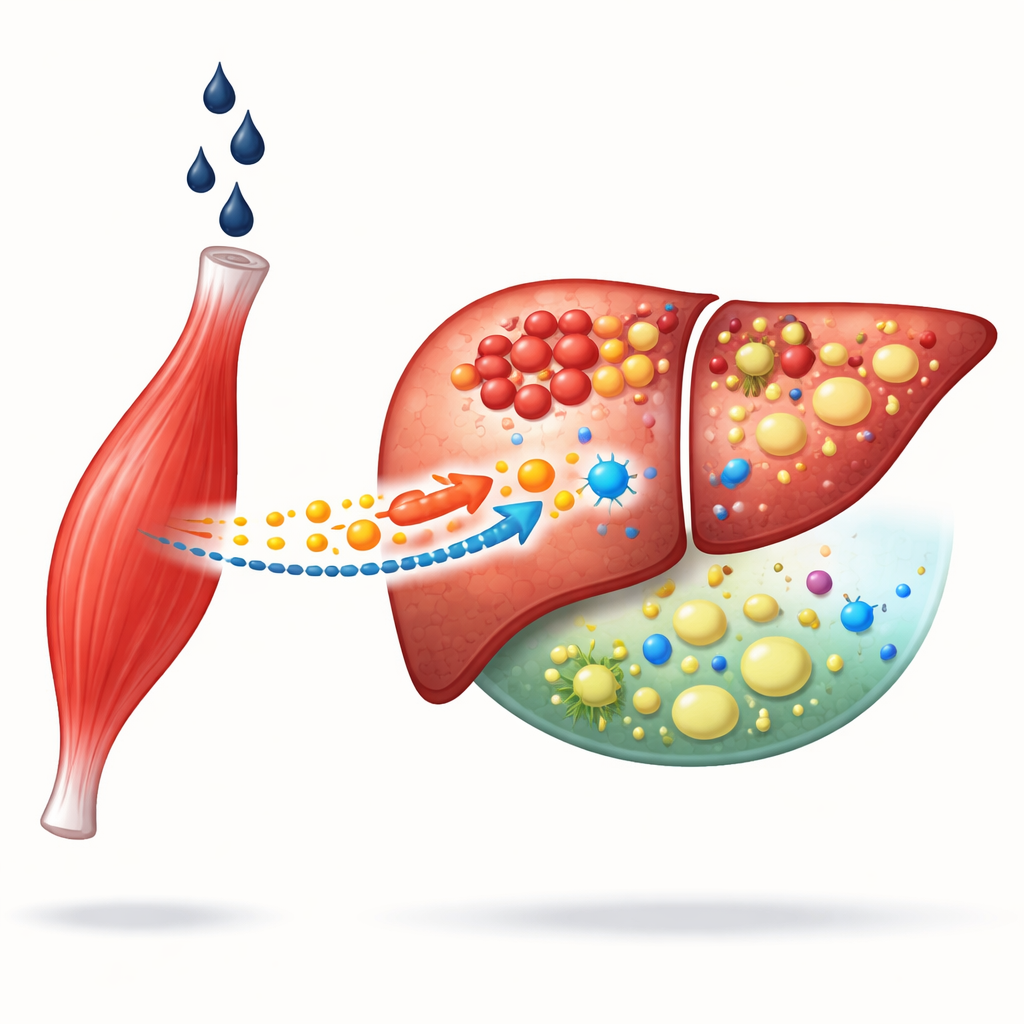
When Drinking Hurts Both Muscle and Liver
Alcoholic liver disease develops over years of heavy drinking and is closely linked to muscle loss. Patients with weaker muscles tend to have worse liver damage and higher risk of complications. The authors began by analyzing national health survey data and human liver protein databases. They found that people with low muscle mass were roughly twice as likely to have alcohol‑related liver problems, especially men. Among many circulating proteins, one stood out as a strong candidate connecting muscle health to liver outcomes: MG53, a protein made mainly by skeletal muscle that is known as a kind of “molecular bandage” for repairing damaged cell membranes.
A Traveling Guardian from Muscle to Liver
Under normal conditions, liver cells do not make their own MG53 and rely on what arrives from the bloodstream. In people and mice with alcoholic liver disease, the researchers saw MG53 levels dropping in the blood but rising inside the liver, suggesting that the protein is being pulled out of circulation into injured liver tissue. Mice genetically engineered to lack MG53 were much more vulnerable to alcohol‑induced damage: their livers became fattier, more inflamed, more scarred, and their survival rates plummeted. When the missing protein was replaced with a laboratory‑made version, many of these problems improved, indicating that MG53 is a crucial part of the body’s defense against chronic alcohol injury.
How MG53 Supercharges Alcohol Breakdown
Breaking down alcohol safely requires two key liver enzymes, alcohol dehydrogenase (ADH) and aldehyde dehydrogenase 2 (ALDH2). These machines convert alcohol into a highly reactive intermediate and then into a less harmful substance that the body can excrete. This process depends on zinc, a trace metal that is often depleted in heavy drinkers. The study shows that MG53 physically binds to both enzymes and helps deliver zinc to them, acting like a metallochaperone—a carrier that loads the enzymes with the metal they need to work efficiently. In mice lacking MG53, liver zinc levels fell further, alcohol and its toxic byproducts accumulated in the blood, and oxidative stress surged. Adding back recombinant MG53 restored zinc levels, boosted enzyme activity, and reduced toxic alcohol intermediates.
Building a Smarter Drug Delivery Package
To turn these insights into a potential treatment, the team engineered tiny bio‑derived nanoparticles that package MG53 together with zinc. These MG53@Zn particles are built on a human serum albumin shell and are sized to accumulate in the liver. In cell cultures exposed to very high alcohol levels, the particles entered liver cells, reduced oxidative stress and fat buildup, and preserved cell survival better than MG53 alone. In mice fed a chronic‑plus‑binge alcohol diet, repeated dosing with MG53@Zn nanoparticles improved survival, restored liver zinc stores, lowered markers of liver injury and inflammation, reduced scarring, and cleared alcohol and its toxic intermediate more efficiently. Importantly, the nanoparticles appeared safe in healthy mice over a month‑long treatment period.
What This Means for Future Care
For a layperson, the key message is that muscle and liver work together more intimately than previously appreciated. A protein released from muscle can travel to the liver, carry zinc to alcohol‑processing enzymes, and help the organ cope with the stress of heavy drinking. By designing a nanoparticle that delivers both this protein and zinc directly to the liver, the researchers outline a promising strategy to treat alcoholic liver disease. While these results are so far limited to animal models and male subjects, they point toward future therapies that support the body’s own repair systems rather than simply treating symptoms after severe damage has already occurred.
Citation: Shu, T., Zeng, X., Wang, J. et al. MG53 mediates skeletal muscle-liver cross-talk and enhances alcohol metabolism in alcoholic liver disease. Nat Commun 17, 2722 (2026). https://doi.org/10.1038/s41467-026-69132-6
Keywords: alcoholic liver disease, MG53, muscle–liver crosstalk, zinc metabolism, nanoparticle therapy