Clear Sky Science · en
Late-stage N-Me selective alkenylation and alkynylation of drugs and unactivated tertiary amines using photoredox catalysis
Why changing old medicines still matters
Many of today’s medicines work well but leave room for improvement in their safety, side effects, or how precisely they hit their targets in the body. Chemists are increasingly interested in gently “tweaking” existing drugs late in the development process, rather than inventing completely new molecules from scratch. This study shows how beams of visible light can be used to clip new pieces onto a common type of drug-building block, creating improved antidepressant candidates and opening a faster route to refined treatments.
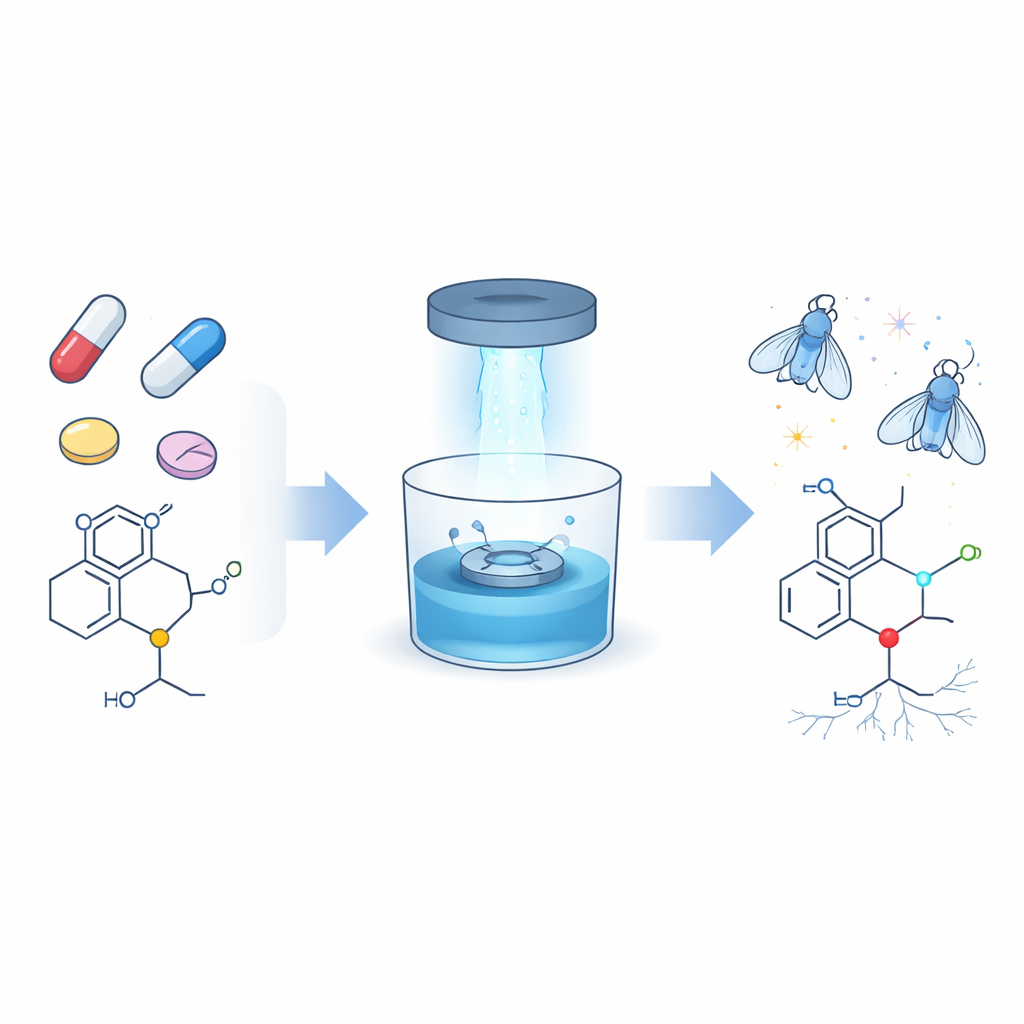
Upgrading a common feature in many drugs
A surprisingly large share of modern medicines—more than two out of every five—contain nitrogen-rich fragments called amines, and most of these are a subtype known as tertiary amines. They appear in antibiotics, cancer drugs, allergy pills, painkillers, and antidepressants. Until now, chemists have struggled to selectively modify just one tiny part of these groups, the N-methyl unit, when it sits among several nearly identical carbon–hydrogen bonds. The authors set out to develop a gentle, reliable way to attach new “handles,” specifically carbon–carbon double bonds (alkenes) and triple bonds (alkynes), to that N-methyl position without disturbing the rest of the drug molecule.
Using light to rewire one bond at a time
The team turned to photoredox catalysis, a technology that uses visible light and a light-sensitive catalyst to move single electrons around and trigger reactions under mild conditions. In their system, blue light excites an organic catalyst, which temporarily pulls an electron from the tertiary amine. This step creates a short-lived radical at the N-methyl site that can join to carefully chosen partner molecules carrying vinyl or alkynyl groups. By tuning the reaction conditions—choice of solvent, base, and additives—the researchers achieved high yields and excellent control, so that the new group lands almost exclusively on the N-methyl position, even when several similar sites are present. They demonstrated this on a wide panel of simple amines and then on real drugs, including antihistamines, heart medicines, anti-infectives, and several antidepressants.
From chemical tweak to better antidepressant action
To test whether these structural tweaks could matter medically, the authors focused on imipramine, a long-used tricyclic antidepressant. They designed and modeled more than twenty imipramine variants in which the new vinyl or alkynyl extension carried aromatic rings that could fit snugly into a hydrophobic pocket of the human serotonin transporter, a protein that controls levels of the mood-related messenger serotonin. Computer docking suggested that many of these modified molecules should bind as well as, or better than, the parent drug. The most promising candidates were then synthesized using the new light-driven method and tested in living systems: a fruit fly model of depression based on social isolation and, for the best two compounds, in mouse nerve cells that actively transport serotonin.
How flies and cells revealed stronger drugs
In isolated male fruit flies, depression-like behavior shows up as elevated aggression. Feeding imipramine reduces this aggression, mirroring its antidepressant action in humans. Remarkably, several of the imipramine derivatives produced by the new method reduced aggression just as well, and four candidates worked even better than imipramine itself. In cultured mouse hippocampal cells, two lead compounds more strongly blocked serotonin reuptake than imipramine, as confirmed both by direct chemical measurement of serotonin and by a fluorescent dye that lights up when taken into cells. These tests indicate that subtle late-stage changes at the N-methyl site can genuinely enhance antidepressant potency rather than merely reshaping the molecule on paper.
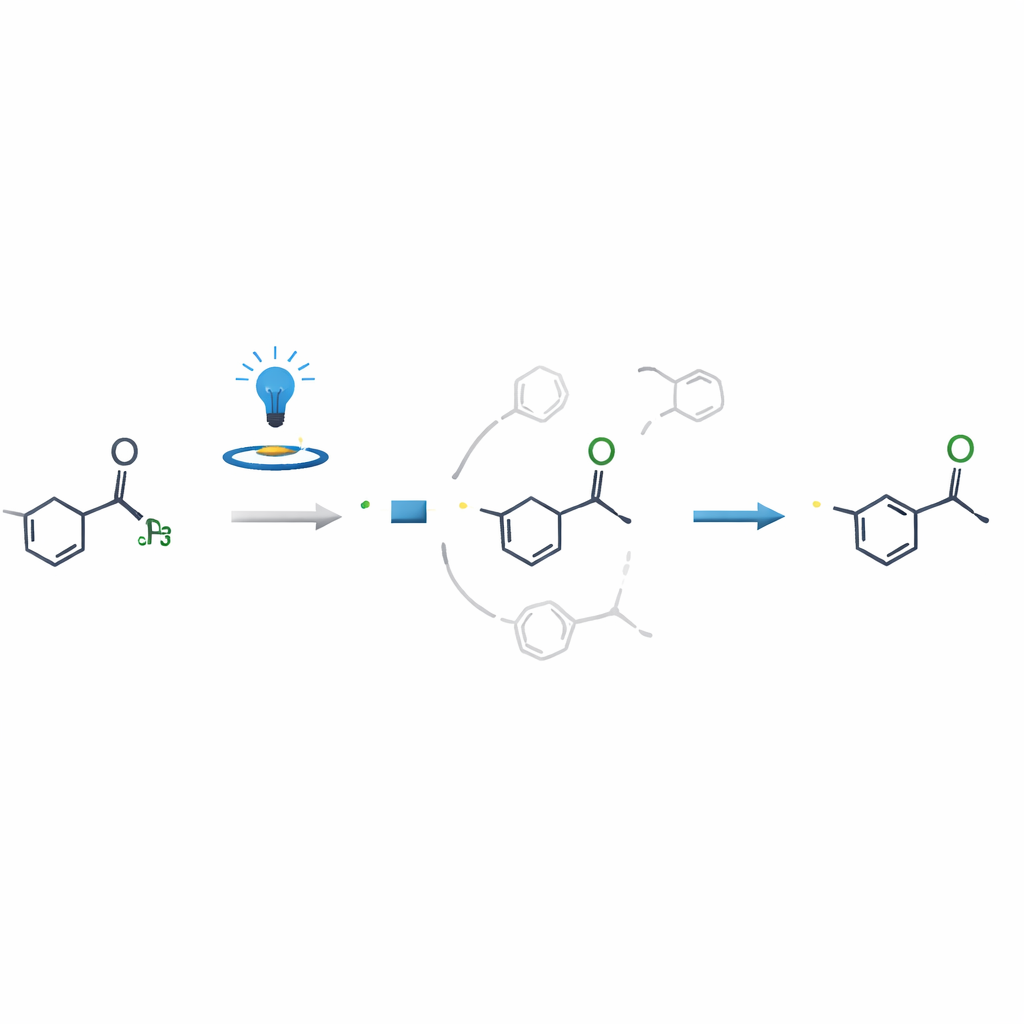
Why the reaction is so selective
A key scientific question was why the reaction modifies the N-methyl group so cleanly when other nearby positions look just as reactive. Through control experiments, electron spin measurements, and detailed computer calculations, the authors showed that the crucial decision point is not when the radical is first formed, but when that radical adds to the vinyl or alkynyl partner. The pathway leading from the N-methyl radical to the final product is both energetically easier and much faster than competing routes, effectively steering the reaction to a single outcome. This insight helps explain the high selectivity and could guide similar strategies on other complex molecules.
What this work means for future medicines
Overall, the study introduces a practical way to surgically upgrade existing drugs that contain tertiary amines, adding compact chemical handles that can tune how they interact with biological targets or link to other components. By showing that such late-stage modifications can turn a well-established antidepressant into even more effective variants in simple animal and cell models, the work points toward a powerful blend of modern synthetic chemistry, computation, and biology. As the approach is extended to other medicines, it may speed the refinement of drug candidates and help refresh older therapies with improved performance and safety.
Citation: Paul, S., Mahato, S., Mahapatra, S.K. et al. Late-stage N-Me selective alkenylation and alkynylation of drugs and unactivated tertiary amines using photoredox catalysis. Nat Commun 17, 2695 (2026). https://doi.org/10.1038/s41467-026-69131-7
Keywords: photoredox catalysis, late-stage functionalization, tertiary amines, antidepressant drug design, serotonin transporter