Clear Sky Science · en
Motor learning and dopamine-dependent striatal synaptic plasticity are controlled by astrocytic MEGF10
Why brain support cells matter for learning new moves
Learning to ride a bike or play the piano feels like a job for nerve cells, not their humble support staff. Yet this study reveals that star‑shaped brain cells called astrocytes quietly help us master new movements. By pruning and tuning connections between neurons in a key movement center of the brain, astrocytes—guided by the chemical messenger dopamine—shape how efficiently we learn and refine motor skills.
Cleaning up to learn better
Motor learning depends on the brain’s ability to strengthen some connections between neurons and weaken or remove others. The authors focused on the dorsolateral striatum, a region that helps turn practice into smooth, automatic actions. There, signals arrive from the motor cortex and are strongly influenced by dopamine, a messenger involved in movement, motivation, and diseases such as Parkinson’s. The team asked whether astrocytes actively remove unneeded connections, and whether this “clean‑up” is important for learning new motor tasks.
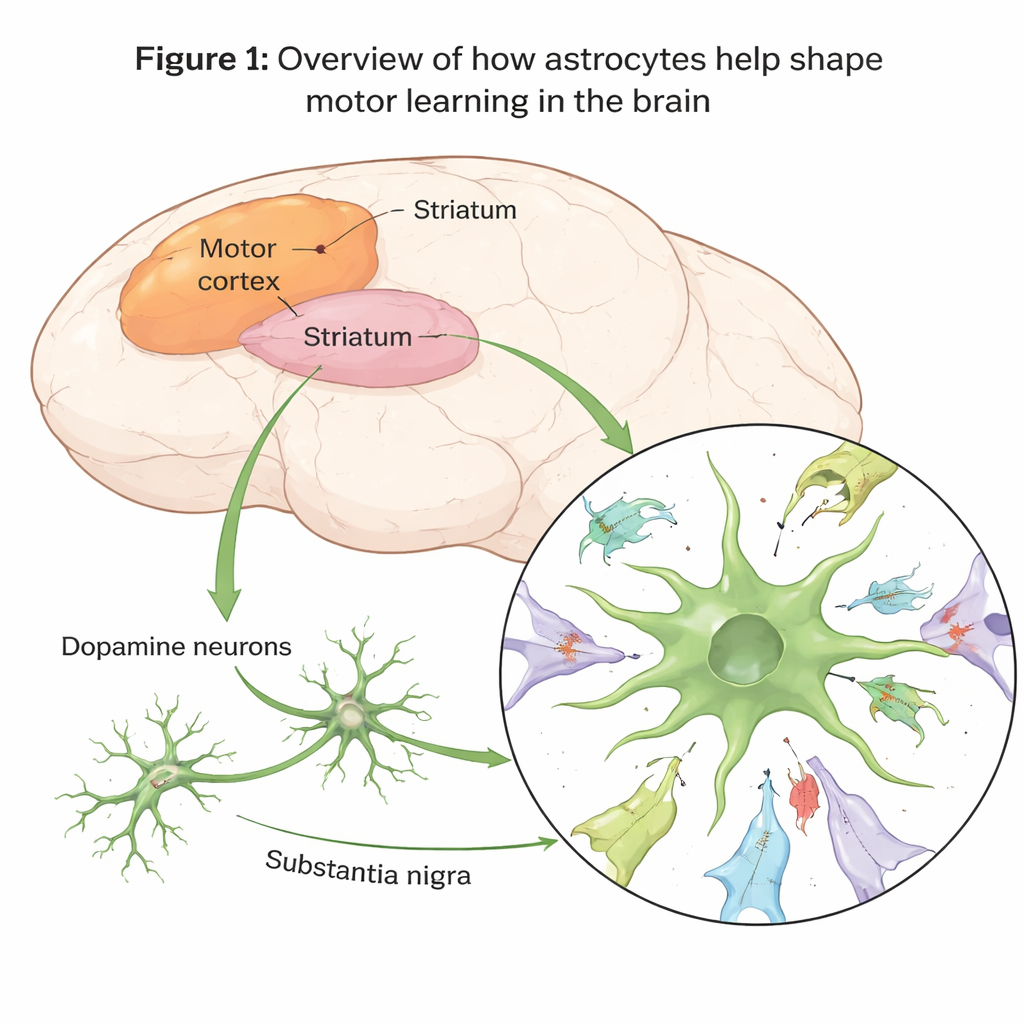
Astrocytes do the pruning, not the usual suspects
Using specially designed fluorescent tags in mice, the researchers could watch tiny pieces of synapses—contact points where neurons communicate—being swallowed by different glial cells. During days of motor training on tasks like a rotating rod or skilled forelimb reaching, astrocytes in the dorsolateral striatum increasingly engulfed both incoming cortical terminals and postsynaptic structures on the receiving neurons. In contrast, other glial cells that are often blamed or credited for pruning, such as microglia and certain precursor cells, showed little change. When the scientists disabled a particular astrocyte surface protein called MEGF10, which acts like a phagocytic receptor for “eat me” signals, synapse engulfment dropped sharply and the animals struggled to improve on the motor tasks.
Pruning supports stronger, flexible connections
Counterintuitively, blocking this astrocytic clean‑up did not leave the striatum bristling with extra, supercharged connections. Instead, the strength of communication between cortex and striatum fell, and two classic forms of synaptic flexibility—long‑term potentiation and long‑term depression—were blunted. After motor training, normal mice showed a clear boost in signal strength along this pathway, but mice lacking astrocytic MEGF10 gained far less. Additional experiments that temporarily blocked MEGF10 only during training similarly dulled both astrocytic pruning and learning. Together, the data suggest that removing weaker or poorly tuned synapses frees space and resources for stronger, more adaptable ones.
Dopamine steers which synapses go and which stay
Dopamine input from a midbrain region called the substantia nigra turned out to be a powerful regulator of this astrocytic pruning. When the researchers artificially boosted activity in dopamine neurons, astrocytes became more active in engulfing presynaptic terminals, an effect that largely vanished without MEGF10. Yet dopamine’s influence on the receiving side of the synapse was more nuanced. The main striatal neurons fall into two groups: D1 cells, which are typically activated by dopamine, and D2 cells, which tend to be suppressed by it. Elevated dopamine reduced astrocytic removal of D1 neuron postsynapses but increased removal of D2 neuron postsynapses. Over time, this selective pruning matched changes in tiny dendritic spines: D1 neurons gained more stable, stubby spines, while D2 neurons lost thin, likely weaker spines—a pattern that again depended on MEGF10‑driven astrocyte activity.
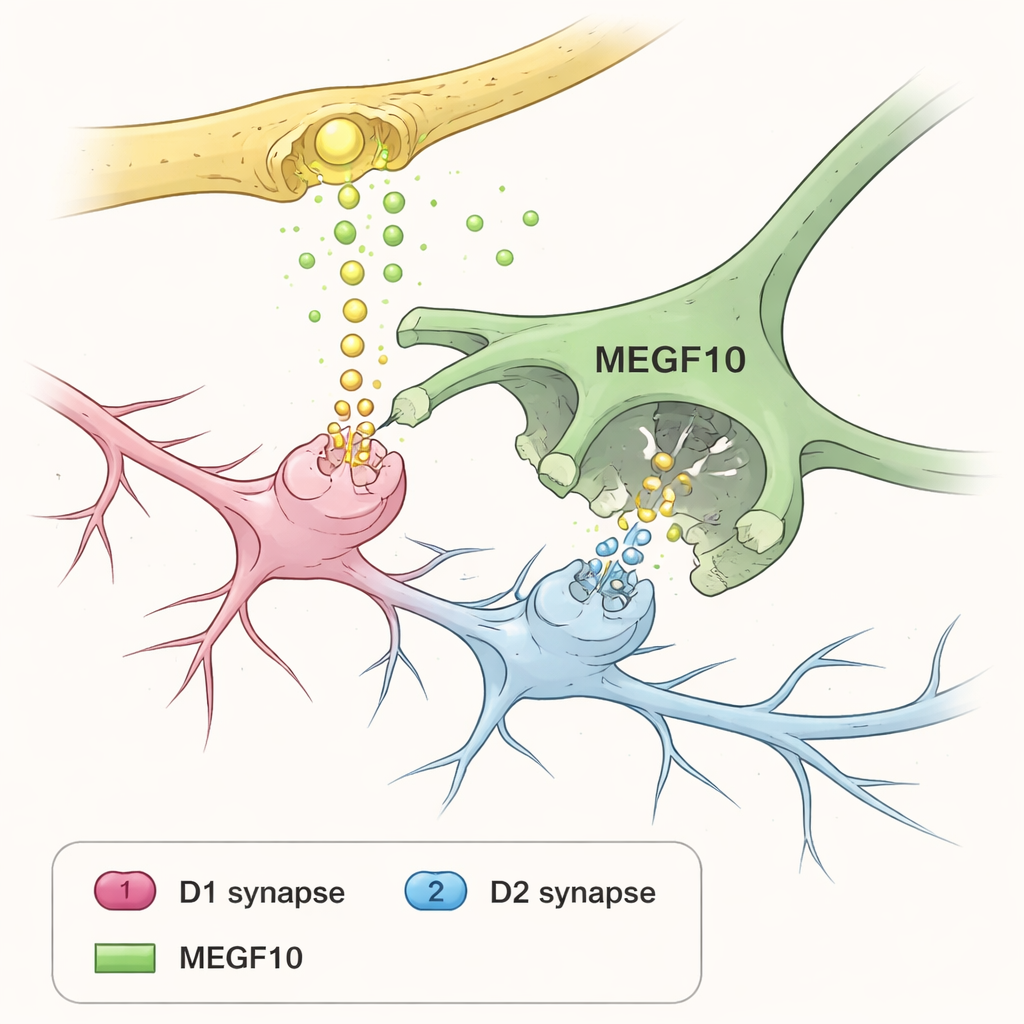
How this shapes movement and disease
For a non‑specialist, the take‑home message is that learning smooth, practiced movements requires more than neurons talking to one another. Astrocytes must constantly inspect and cull specific connections, and they do so under the guidance of dopamine and a key “eat me” receptor, MEGF10. Without this targeted clean‑up, the circuitry that underlies motor skills becomes weaker and less flexible, and animals learn new movements more slowly. Because dopamine loss in disorders like Parkinson’s disease severely disrupts these same pathways, the work raises the possibility that faulty astrocytic pruning contributes to motor symptoms—and that future therapies might one day target not just neurons, but their vigilant astrocyte partners.
Citation: Choi, YJ., Lee, Y.L., Kim, Y. et al. Motor learning and dopamine-dependent striatal synaptic plasticity are controlled by astrocytic MEGF10. Nat Commun 17, 1351 (2026). https://doi.org/10.1038/s41467-026-69129-1
Keywords: motor learning, astrocytes, dopamine, synaptic plasticity, striatal circuits