Clear Sky Science · en
Dynamic chromium dopant promotes interfacial water activation on cobalt spinel oxide for efficient oxygen evolution in acid
Why this research matters for clean energy
Producing hydrogen fuel from water could power industry and transportation without carbon emissions, but today’s most efficient devices rely on rare and costly precious metals. This study explores how a carefully designed, low‑cost material based on cobalt and chromium can replace those metals in one of the toughest parts of water splitting: making oxygen in acidic conditions, as used in commercial‑style proton exchange membrane water electrolyzers (PEMWEs). 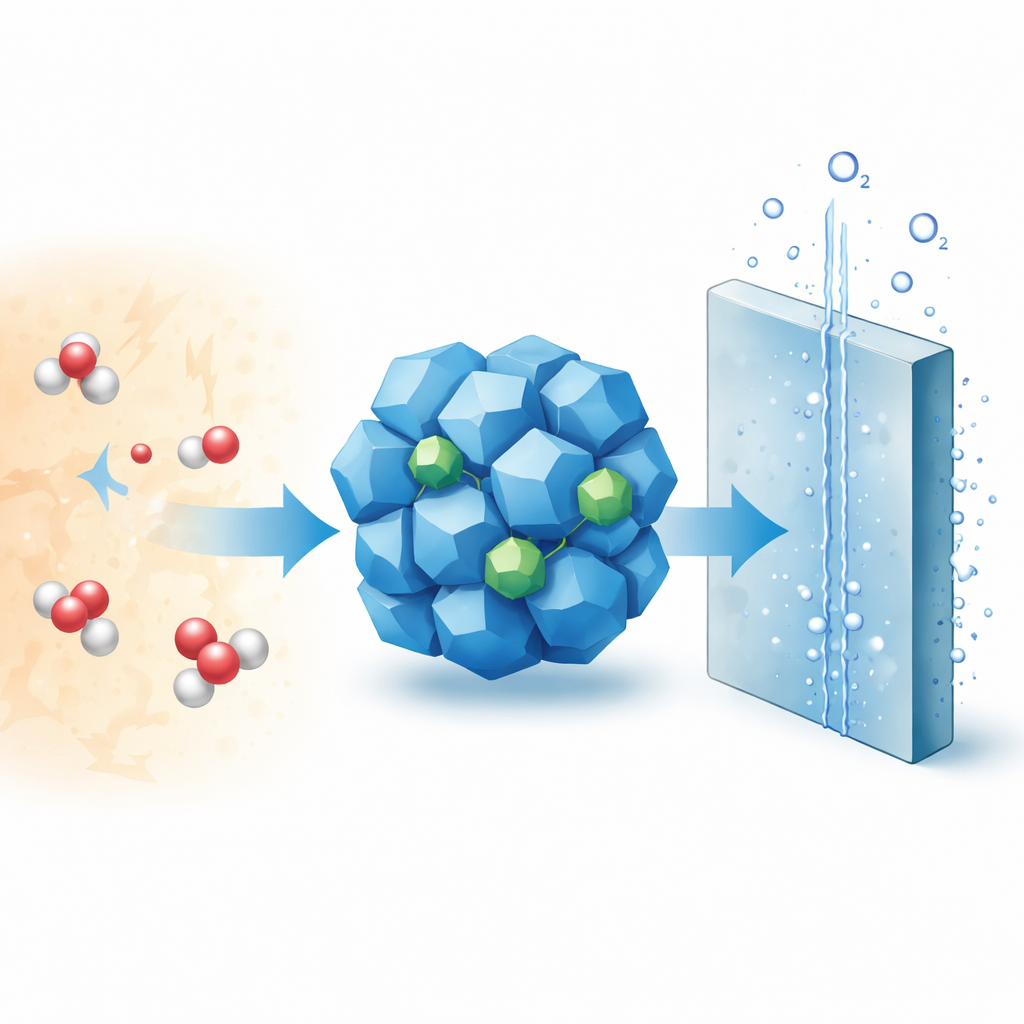
The challenge of making oxygen from water
Modern PEMWEs are attractive because they can rapidly convert electricity from solar or wind into hydrogen, even when power input fluctuates. However, on the oxygen‑producing side of these devices, the reaction is slow and harsh. It takes place in an acidic environment and involves moving both protons and electrons in a tightly coupled way. Today this step is usually handled by catalysts made from iridium and ruthenium oxides—metals that are scarce and expensive. Cobalt oxide (Co3O4) has emerged as a promising alternative, but in acid it tends to corrode: cobalt atoms dissolve into the liquid, the surface over‑oxidizes into unstable forms, and the catalyst gradually falls apart.
A smart tweak: adding chromium atoms
The authors show that introducing a small amount of chromium into cobalt spinel oxide reshapes both the internal electronic structure of the solid and the thin layer of water at its surface. They synthesize tiny, uniform nanoparticles of chromium‑doped cobalt oxide (Cr‑Co3O4) and confirm, using diffraction and electron microscopy, that the material keeps the original spinel structure. Advanced spectroscopic techniques reveal that chromium atoms slip into specific tetrahedral positions in the lattice and are dispersed as single atoms rather than forming separate chromium oxide particles. This positioning creates a local chromium‑oxygen‑cobalt environment that slightly lowers the average charge on cobalt atoms, making them less prone to over‑oxidation.
Better performance in harsh acidic conditions
When tested in sulfuric acid, the chromium‑doped catalyst needs much less extra voltage than undoped cobalt oxide to reach the same current, indicating that it accelerates oxygen production. It also outperforms commercial ruthenium and iridium oxides at higher current densities and, crucially, maintains its activity for at least 160 hours, while the other catalysts degrade. Electrical measurements show that charge moves more easily across the interface between the doped catalyst and the liquid. When built into a full PEM water‑splitting device, Cr‑Co3O4 used as the oxygen‑side catalyst, paired with a standard platinum hydrogen‑side catalyst, operates stably for more than 750 hours at industrially relevant current, demonstrating practical durability.
How chromium reshapes the water layer
To move beyond simple performance numbers, the researchers probe how the catalyst surface and nearby water molecules evolve during operation. In situ X‑ray and Raman measurements show that in pure cobalt oxide the cobalt atoms become strongly over‑oxidized at high voltages, a precursor to structural breakdown. In the doped material, by contrast, the cobalt oxidation state stays nearly constant while chromium gradually shifts, indicating that chromium acts as an electron “buffer” that protects cobalt. Surface‑sensitive measurements further reveal that under operating voltages chromium atoms increasingly bind hydroxyl groups (OH). These surface hydroxyls change the structure of the water layer touching the electrode: infrared spectroscopy shows that hydrogen‑bond networks become weaker and that the fraction of more mobile “free” water molecules rises. Because these water molecules dissociate more easily, the reaction that turns water into oxygen speeds up. 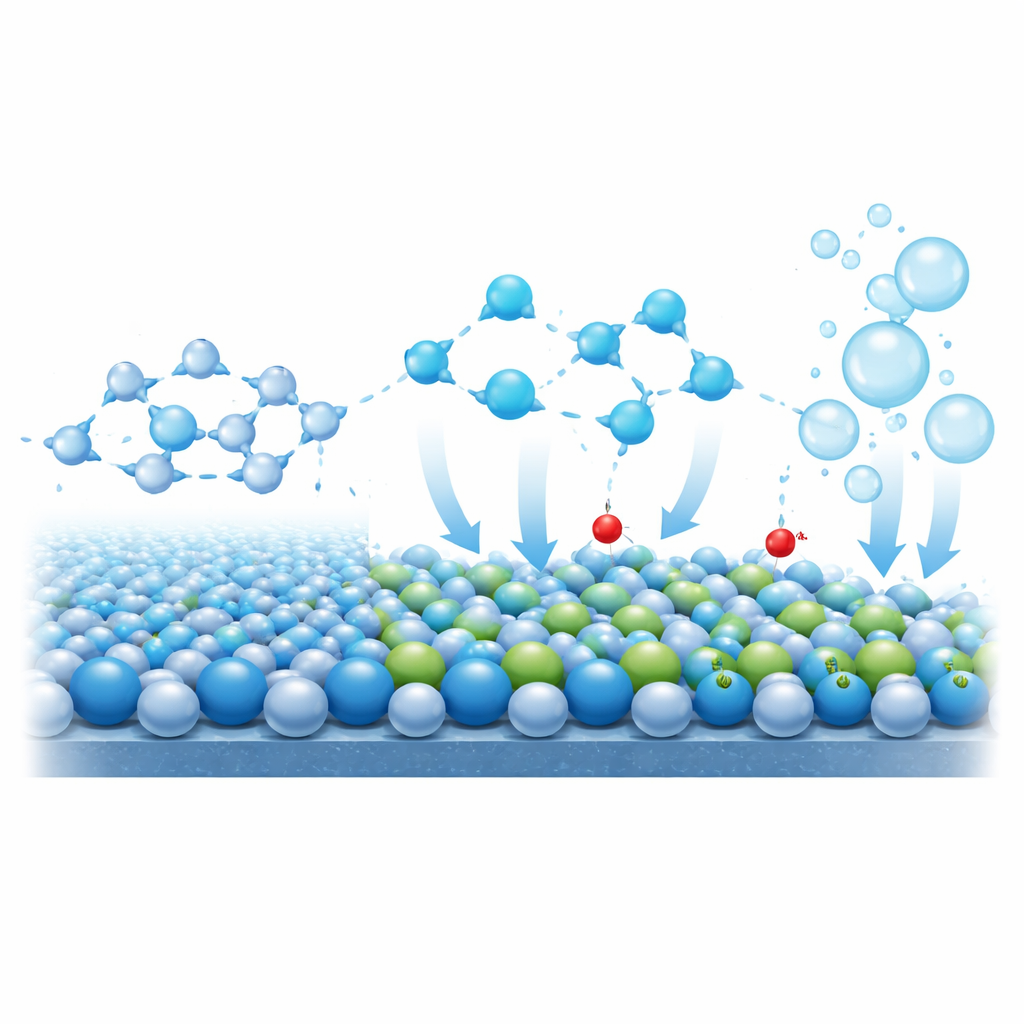
Connecting theory and experiment
Computer simulations back up this picture. Calculations show that chromium prefers the same lattice sites identified experimentally and tends to donate electron density toward nearby cobalt. The energy required to split a water molecule on the doped surface is lower than on pure cobalt oxide, especially when a hydroxyl is already present on the chromium site, mirroring the experimental finding that hydroxyl‑rich surfaces are more active. The simulations also indicate that pulling a cobalt atom out of the doped surface costs more energy, explaining the improved resistance to dissolution. Overall reaction‑energy diagrams confirm that the hardest step in the oxygen‑forming sequence becomes easier once chromium is introduced.
What this means for future water‑splitting devices
Together, these results show that a small amount of the right dopant can dramatically change how both electrons and water molecules behave at a catalyst’s surface. By using chromium to stabilize cobalt and to foster a layer of easily activated, weakly bound water, the team creates a robust, non‑precious‑metal catalyst that rivals noble‑metal oxides in demanding acidic conditions. For a lay reader, the key takeaway is that smart atomic‑scale engineering—a few chromium atoms in just the right places—can make affordable materials do the hard work of splitting water, bringing large‑scale green hydrogen production closer to reality.
Citation: Wu, L., Zhao, B., Huang, W. et al. Dynamic chromium dopant promotes interfacial water activation on cobalt spinel oxide for efficient oxygen evolution in acid. Nat Commun 17, 2598 (2026). https://doi.org/10.1038/s41467-026-69124-6
Keywords: green hydrogen, water electrolysis, oxygen evolution reaction, cobalt oxide catalyst, interfacial water