Clear Sky Science · en
An isolable phosphaalumene(3) capable of small molecule activation via unique modes of reactivity
Breaking Tough Bonds with a New Chemical Tool
Chemists are always searching for ways to persuade stubborn molecules to react. Many everyday substances, from hydrogen gas to carbon dioxide, are impressively stable, which makes them hard to transform into useful products. This paper reports a newly designed molecule built from aluminum and phosphorus that can grab onto such reluctant partners and split open their bonds. Understanding and controlling this kind of bond activation could one day help turn waste gases into feedstocks, or offer milder, metal‑saving routes to make fine chemicals and materials.
A Designer Bond Between Aluminum and Phosphorus
At the heart of the study is a rare type of connection between two main‑group elements: aluminum and phosphorus. The researchers crafted a molecule, called “3” in the paper, in which these two atoms share a tight double bond. To keep this reactive pair from falling apart, they wrapped it in a bulky organic framework that shields it from accidental reactions. Using two different synthetic routes, they obtained this complex in isolable form and grew single crystals suitable for X‑ray diffraction, confirming that phosphorus is bonded to aluminum with a short, double‑bond‑like link while each atom is supported by surrounding nitrogen‑containing ligands. Calculations showed that electrons are unevenly shared between aluminum and phosphorus, making the bond polarized and primed for attack by other molecules.
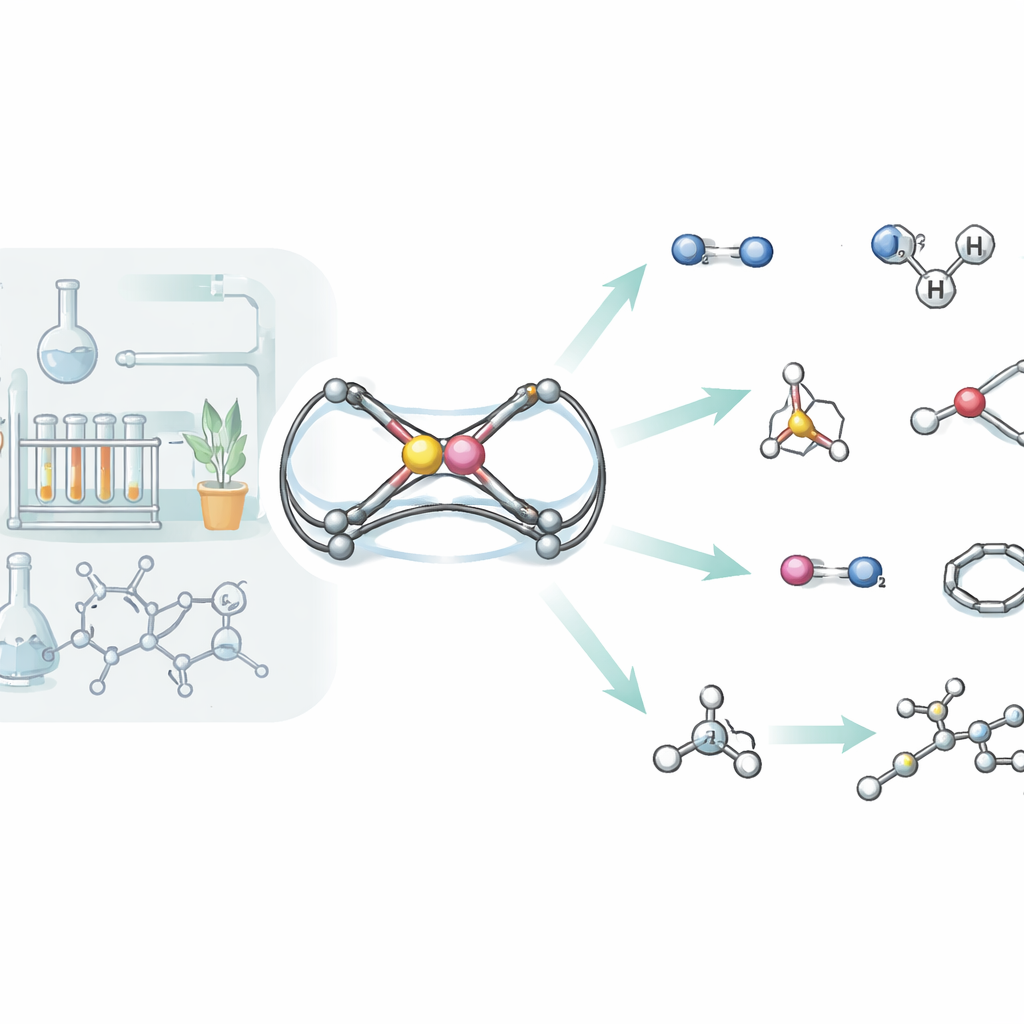
How the New Molecule Handles Hydrogen Gas
Hydrogen gas is famously difficult to split without using precious metals, yet the new aluminum‑phosphorus unit manages to do just that. When exposed to hydrogen under gentle conditions, the complex reacts in two distinct ways. In one pathway, the H–H bond simply adds across the aluminum–phosphorus double bond, turning it into two single bonds and attaching one hydrogen to each atom. In the other, the hydrogen molecule is pulled apart cooperatively by aluminum and a neighboring nitrogen atom, a behavior reminiscent of so‑called “frustrated Lewis pairs,” which activate hydrogen without traditional metal catalysts. Detailed computer simulations traced both routes, showing that their energy barriers are similar, explaining why both products are formed side by side.
Taking Apart a Gallery of Small Molecules
Hydrogen is only the beginning. The team systematically tested how their aluminum‑phosphorus scaffold reacts with a wide range of small molecules, including white phosphorus, isocyanides, carbon dioxide, nitrous oxide, trimethylsilyl azide, selenium dimers, silanes, amines, styrene, and simple alkynes. In many of these reactions, the original Al=P link is completely broken, with the two atoms ending up in larger cage‑ or ring‑like structures that incorporate fragments of the incoming molecule. For instance, white phosphorus (P4) is cracked open and rebuilt into an aluminum‑phosphorus cage, while carbon dioxide inserts twice to form a six‑membered ring tying aluminum and phosphorus together through two new carbon‑oxygen bridges. With silanes and amines, the molecule instead behaves more like a classical double bond: the incoming Si–H or N–H unit adds neatly across Al=P, again demonstrating the “π‑bond” character of this unusual linkage.
Why the Surrounding Framework Matters
One of the key lessons of the work is that the bulky, nitrogen‑rich framework around the Al=P unit is not just scaffolding; it steers how and where reactions occur. By comparing their results with earlier, more short‑lived aluminum‑phosphorus systems, the authors show that small changes in the supporting ligands flip the balance between different pathways, such as simple addition across the double bond versus more elaborate bond cleavage and ring formation. Sophisticated analyses of electron distribution revealed strong polarization of the Al=P bond and highlighted how neighboring nitrogen atoms can cooperate with aluminum, especially in hydrogen activation. Steric bulk—the physical crowding of large groups—also nudges incoming molecules toward one reactive site or another.
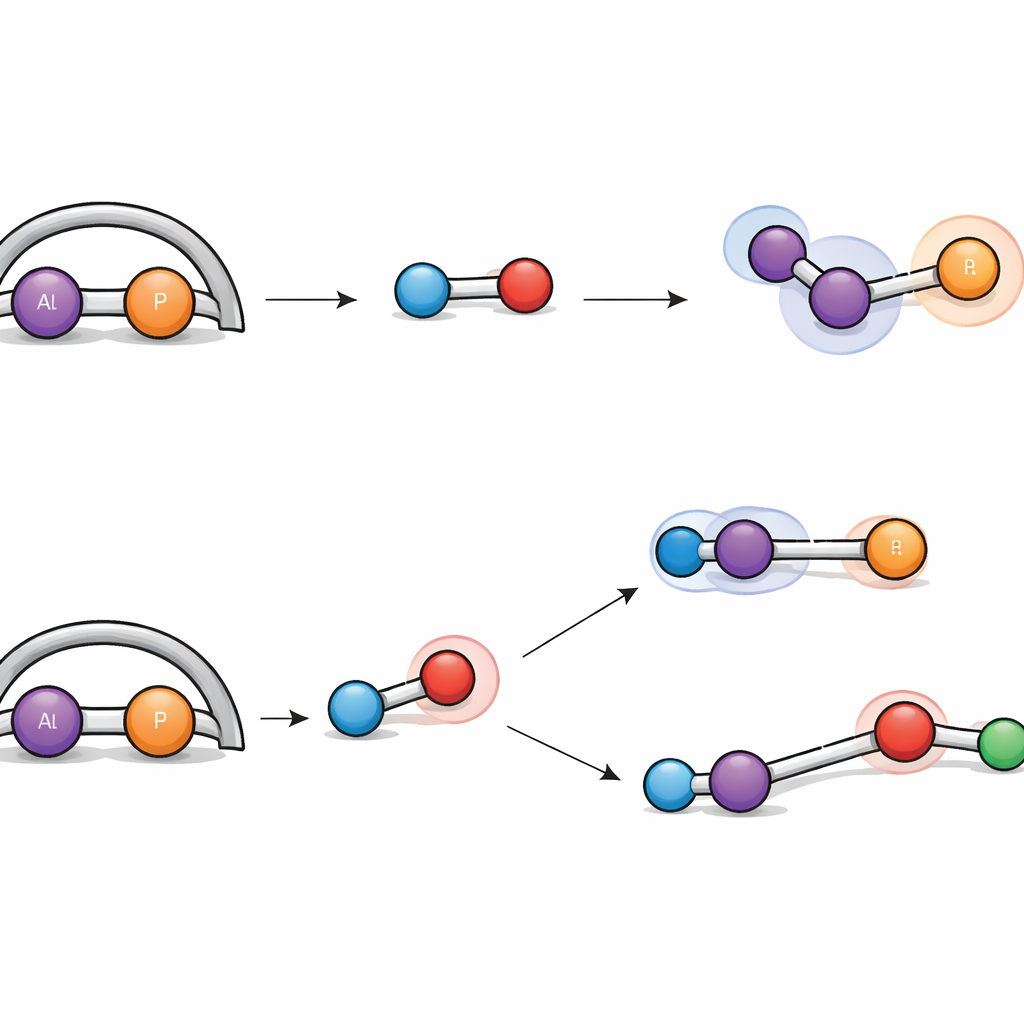
What This Means for Future Chemistry
To a non‑specialist, the alphabet soup of reagents in this study may seem remote, but the underlying message is clear: by carefully tuning both bonding and shape around main‑group elements, chemists can build nimble tools that rival or complement traditional metal catalysts. This isolable aluminum‑phosphorus complex not only survives in a bottle but also tackles a broad set of tough small molecules under mild conditions, splitting hydrogen, rearranging white phosphorus, and capturing carbon dioxide. The work shows that subtle changes in a molecule’s “handle” can profoundly redirect its reactivity, offering a blueprint for designing future catalysts that help convert simple, stable molecules into more valuable products with precision and efficiency.
Citation: Cha, Y., Yang, Z., Zhuang, X. et al. An isolable phosphaalumene(3) capable of small molecule activation via unique modes of reactivity. Nat Commun 17, 2390 (2026). https://doi.org/10.1038/s41467-026-69118-4
Keywords: small molecule activation, main group chemistry, aluminum phosphorus double bond, frustrated Lewis pair, bond activation mechanisms