Clear Sky Science · en
Hierarchical assembly of a Ti24 metal-organic polyhedron via kinetic trapping of intermediates
Building Tiny Cages for Big Jobs
Chemists are learning how to build miniature cages out of metal atoms and organic pieces—structures so small that thousands could fit across the width of a human hair. These hollow cages can trap gas molecules, act as tiny reaction flasks, or help separate valuable chemical mixtures. This article reports a new and unusually complex titanium-based cage and, crucially, shows how to steer its step-by-step construction, offering a roadmap for designing future "molecular machines" with tailor‑made functions.
Why Titanium Cages Are So Hard to Make
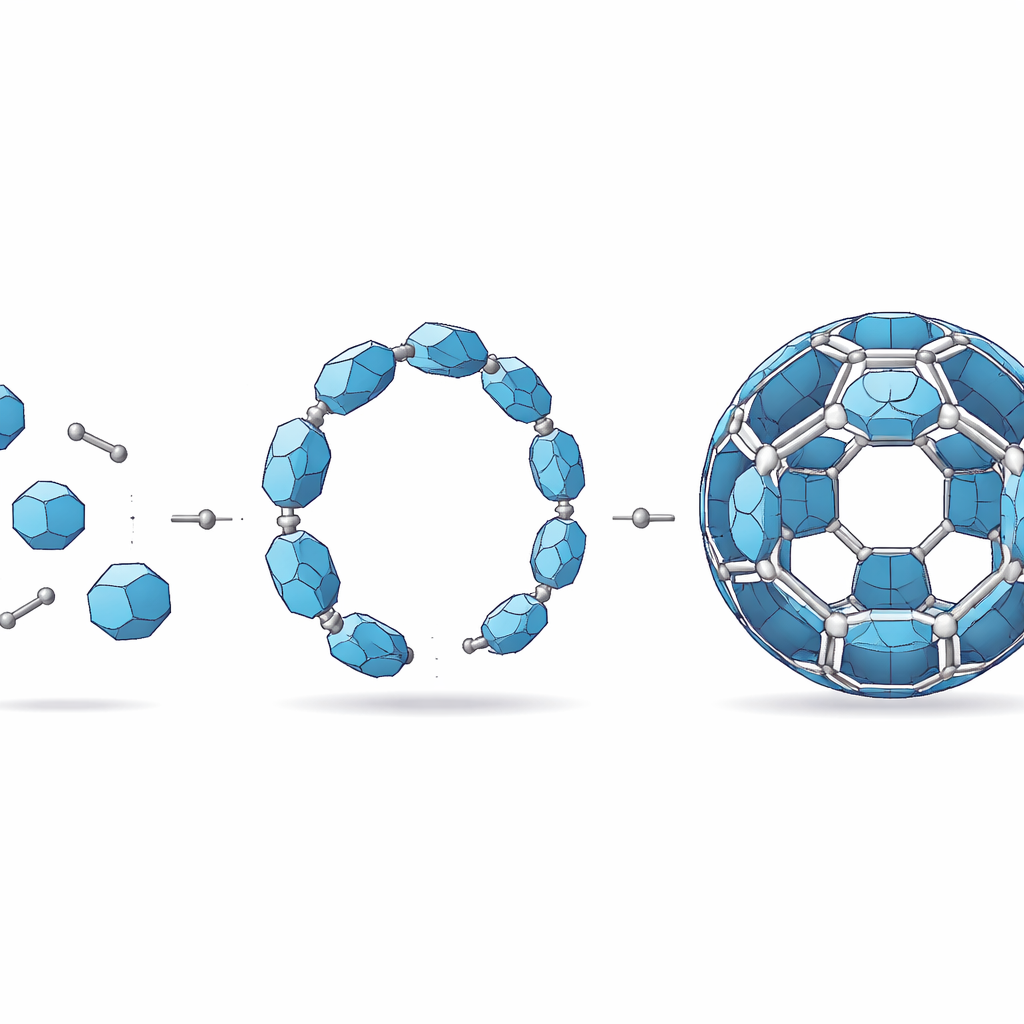
Metal–organic polyhedra are hollow, cage-like molecules assembled from metal atoms and carbon-based linkers. Many metals readily form such cages, but titanium is notoriously difficult to control: it reacts eagerly with oxygen and water, tending to form extended solids instead of neatly defined molecules. As a result, only a small handful of titanium cages were known, and they were relatively simple and small. The new work breaks this barrier by creating a titanium cage that contains 24 titanium atoms arranged in a truncated octahedron—imagine a soccer ball with its corners sliced off—representing the highest level of atomic complexity in this family so far.
Guiding Self‑Assembly Step by Step
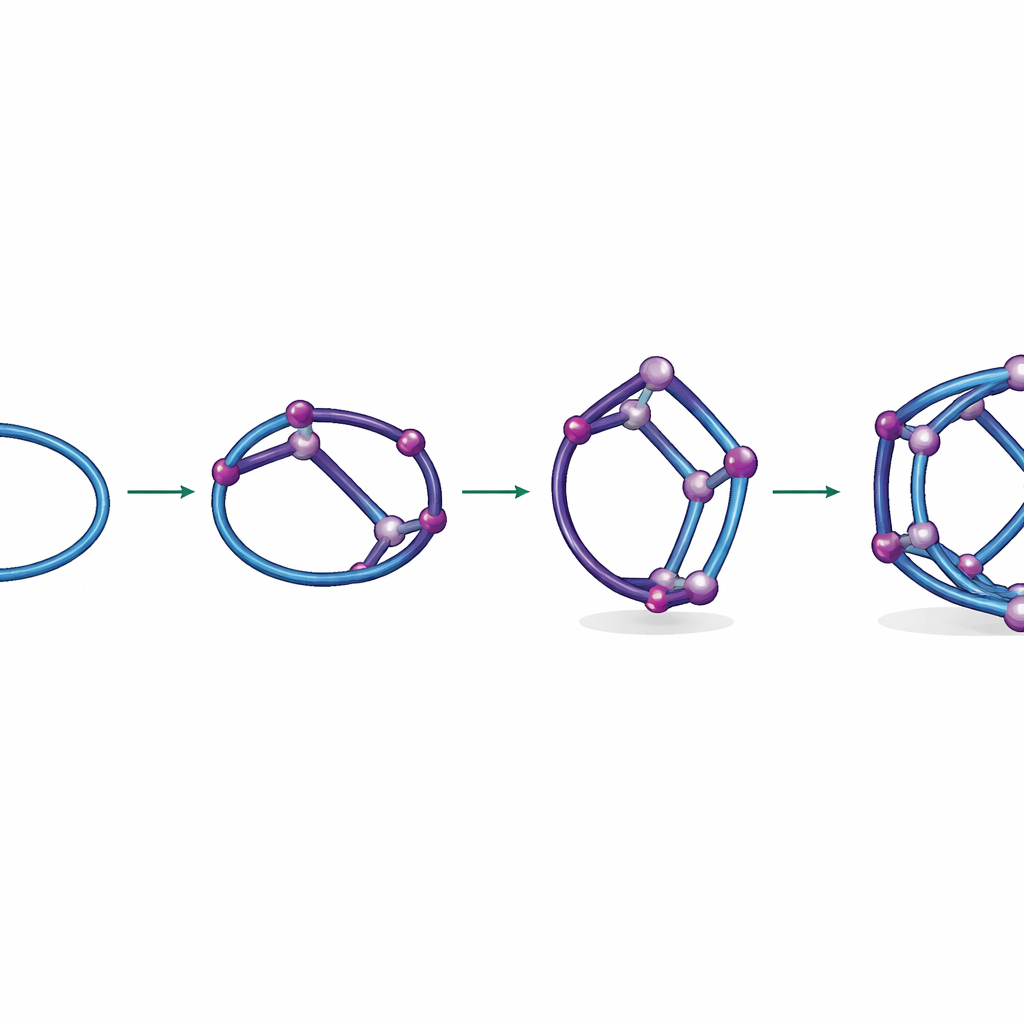
Left to itself, a mixture of titanium building blocks and a square-shaped organic acid gradually organizes into the final 24‑titanium cage, dubbed FIR‑151. But this process passes through short‑lived intermediate shapes that are usually invisible. The researchers designed a way to “pause” assembly and capture these fleeting forms. By adding nickel ions as helpers, they could temporarily lock in two key stages: first, a ring of 12 titanium atoms, and then a bent module where this ring is partially folded and threaded by the organic linker. These snapshots reveal that the final cage is built hierarchically, like snapping together pre‑curved panels rather than assembling every bond from scratch.
Using a Second Metal as a Traffic Controller
The key idea behind this control is a subtle difference in how tightly titanium and nickel bind to surrounding atoms. Titanium’s bonds change rapidly, allowing its structures to rearrange and explore many shapes, while nickel’s bonds are more reluctant to break. By sprinkling in nickel, the team created a kind of “kinetic trap”: nickel clamps onto partially formed titanium rings and modules, holding them long enough to be seen and crystallized, without permanently blocking the route to the final cage. This concept—using a second component with slower bond‑swapping to stabilize specific waypoints along a self‑assembly path—offers a general strategy for sculpting complex molecular architectures.
Tiny Pores with Useful Selectivity
Beyond the architectural feat, the new titanium cage behaves as a functional porous material. When packed together in the solid state, the cages form a regular array of tiny cavities and channels, giving rise to permanent microporosity and a relatively high internal surface area. The material can take up significant amounts of gases such as carbon dioxide and small hydrocarbons, and it distinguishes between closely related molecules like acetylene, ethylene, and ethane. These differences in uptake reflect how well each gas fits and interacts inside the cage’s pores, pointing to potential uses in gas purification or capture technologies.
Tuning the Cage After It Is Built
The team also showed that the outer “decorations” on the cage can be swapped without breaking its overall frame. By exchanging the original small ligands on the surface for bulkier or more aromatic ones, they altered properties such as how the cages pack, how water‑repellent the material becomes, and whether the cage carries groups that could be further polymerized into networks. This post‑assembly editing demonstrates that the titanium cage can serve as a versatile scaffold: its core shape remains intact while its outside can be chemically customized for different tasks.
From Molecular Puzzle to Design Principle
In accessible terms, the study turns a troublesome feature of titanium chemistry—its tendency to rearrange and react quickly—into an advantage. By pairing titanium with a more deliberate partner, nickel, the researchers could watch and guide how simple pieces climb a hierarchy of shapes to become a sophisticated, porous cage. The work delivers both a record‑setting titanium structure and a general lesson: by carefully balancing fast and slow bonding behavior, chemists can program how complex molecular objects assemble, opening new routes to materials that separate gases, host reactions, or store energy at the nanoscale.
Citation: Li, HZ., Yang, CY., Gu, C. et al. Hierarchical assembly of a Ti24 metal-organic polyhedron via kinetic trapping of intermediates. Nat Commun 17, 2302 (2026). https://doi.org/10.1038/s41467-026-69115-7
Keywords: metal-organic cages, titanium chemistry, self-assembly, porous materials, gas separation