Clear Sky Science · en
Cryo-EM structures of bacteriophage T4 portal-neck assembly intermediates reveal a viral genome retention mechanism
How a Virus Keeps Its DNA Under Pressure
Bacteriophage T4 is a virus that infects E. coli bacteria and packs its DNA so tightly into its head that the pressure inside can be five to seven times higher than inside a champagne bottle. Yet the DNA does not leak out while the virus finishes building the rest of its body. This study reveals, in molecular detail, how T4 solves that problem using a tiny double gate that acts like an airlock for its genetic material.
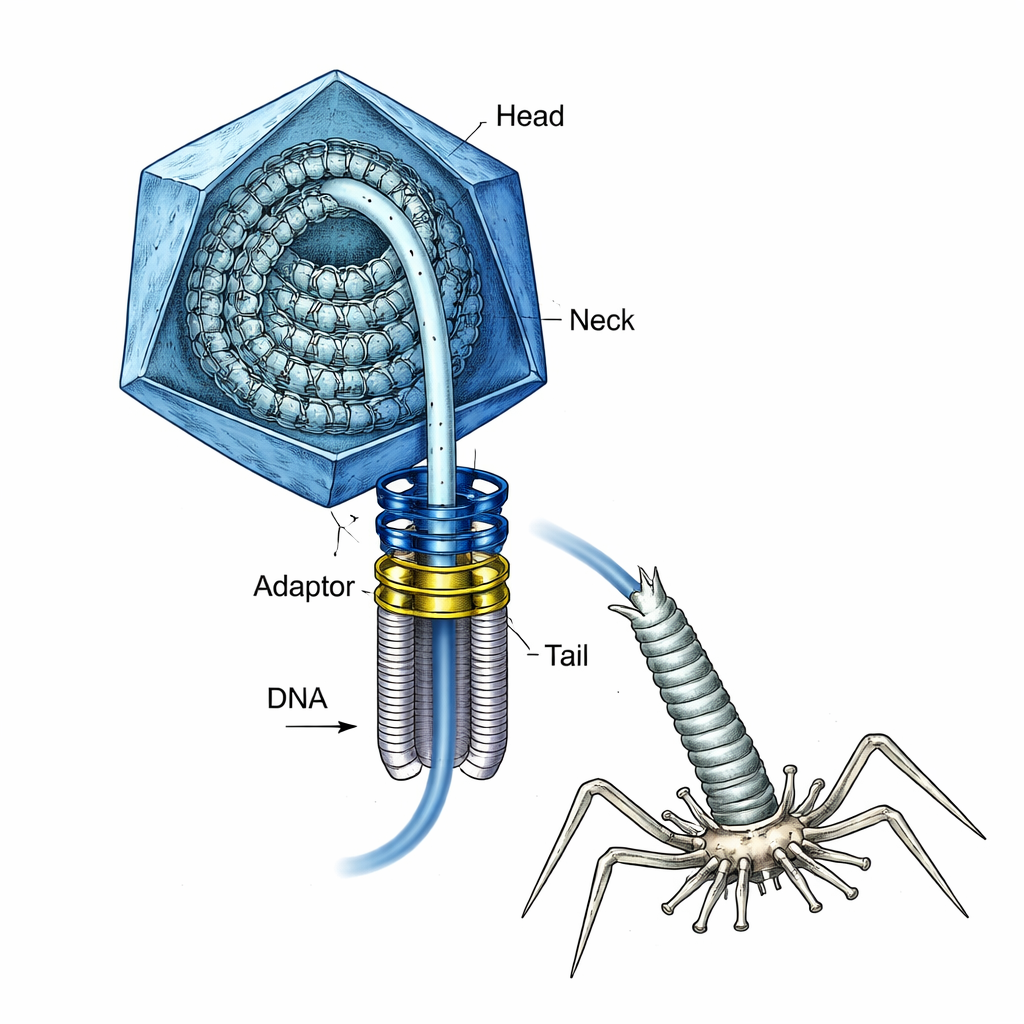
The Virus with a Loaded Spring Inside
T4 has long been a workhorse of molecular biology, and it is also a model for building future vaccines and gene delivery tools. During assembly, the virus first builds an empty protein shell, or head, and a special ring-shaped “portal” at one vertex. A powerful molecular motor then reels the DNA into the head through this portal until the head is “full.” At that point, the motor must come off, a neck and a tail must be attached, and later the DNA must be injected into a bacterium—all without allowing the pressurized DNA to squirt out prematurely. How the virus kept this highly stressed DNA safely contained during those transitions was not well understood.
Rebuilding the Neck in the Lab
The researchers recreated key pieces of T4 in bacteria and mixed them in controlled ways to watch how the neck—the connector between the head and tail—assembles. Two viral proteins, called gp13 and gp14, were produced separately. On their own they floated as single units, but together they snapped into stacked rings that form a central tunnel for DNA passage. To the scientists’ surprise, a third protein kept appearing in these preparations: Hfq, a well-known bacterial protein that usually helps control RNA and gene activity in E. coli. Mass spectrometry confirmed that Hfq was binding specifically to gp14, hinting that the virus borrows this host protein as part of its neck.
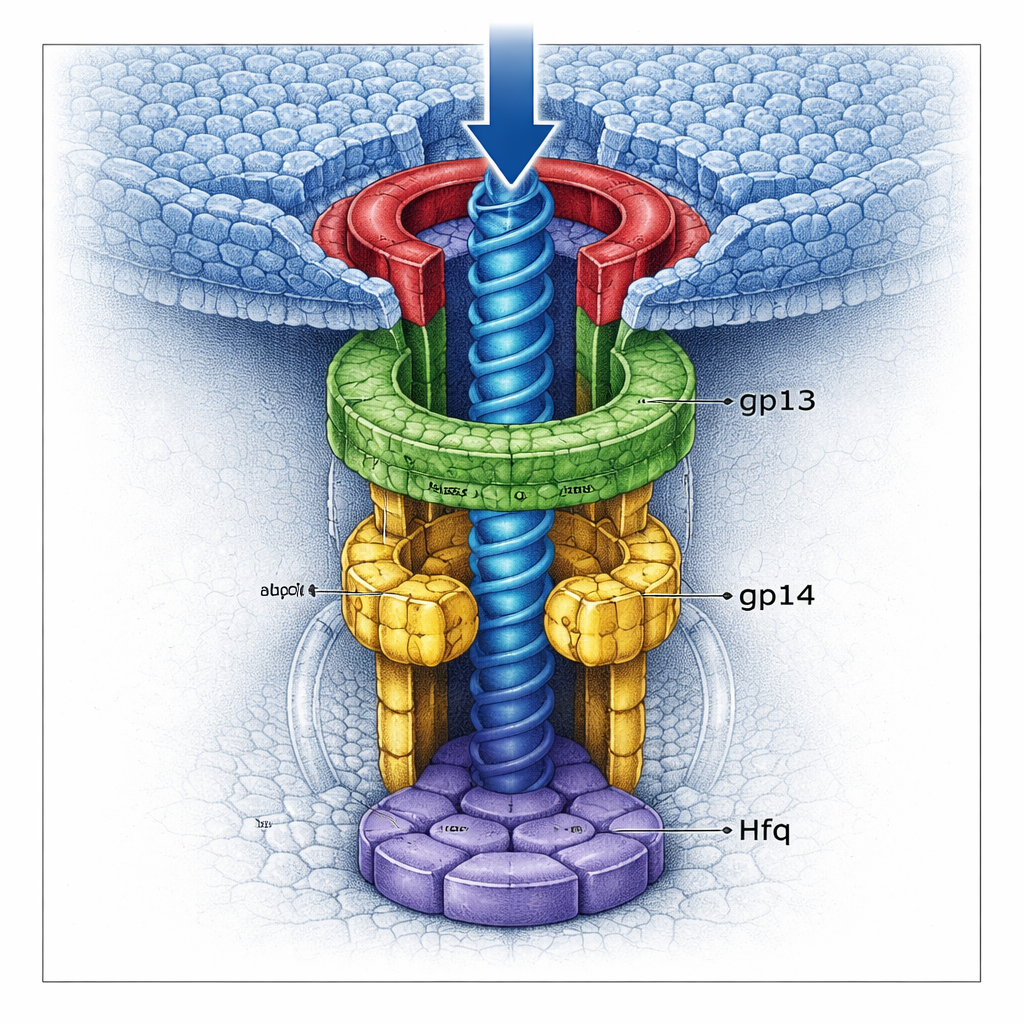
A Double Gate that Locks in the Genome
Using high-resolution cryo–electron microscopy, the team visualized several versions of the neck complex at near-atomic detail. They found that gp13 forms a broad ring that adapts to the portal, while gp14 sits beneath it as a hexamer, creating a narrower channel. Crucially, each gp14 subunit contributes a long loop that swings into the middle of the tunnel. Together, six such “stopper loops” form a tight gate that can catch the last segment of DNA as it tries to leave the head. Below gp14, the hijacked Hfq protein assembles into its own hexamer, plugging the bottom of the channel like a second door. When Hfq is present, the gp13–gp14 structure is more complete and rigid, and the combined gates constrict the tunnel even further, making leakage of DNA far less likely.
Timing, Error-Proofing, and a Borrowed Helper
The study also shows that this system is more than just a static plug; it is a carefully choreographed sequence of shape changes. Once the head is full, the internal pressure pushes the portal into a new conformation that exposes binding sites for gp13. A pre-assembled gp13–gp14–Hfq neck then docks onto the portal. Gp13 flexes, swings upward, and locks onto both the portal and the outer shell, weaving the neck firmly into the head. Throughout this stage, the gp14 stopper loops and the Hfq plug hold the DNA in place. Hfq plays a second role as a quality-control factor: by occupying key surfaces on gp14, it prevents gp14 from binding the portal in the wrong position, which would misassemble the neck. Only when the pre-built tail arrives does a tail-tip protein, gp15, form an even stronger bond with gp14, displacing Hfq and opening the way for controlled DNA release into the tail.
Why This Matters Beyond One Virus
In plain terms, T4 uses a two-door safety lock to hold back a powerful DNA “spring” while it finishes building the rest of the virus. The first door is built from its own neck protein, gp14, and the second door is borrowed from the host’s Hfq protein, which the virus uses temporarily and then discards. This double gate ensures that almost no DNA is lost and that the final virus is fully infectious. Because many large viruses pack their genomes under similar extreme pressures and share related architecture, this work suggests that comparable gating systems, and even the temporary hijacking of host proteins, may be widespread. Understanding these mechanisms could guide the design of safer viral vectors for medicine and point to new ways of blocking harmful viruses by targeting their genome “locks.”
Citation: Han, L., Mao, Q., Zhu, J. et al. Cryo-EM structures of bacteriophage T4 portal-neck assembly intermediates reveal a viral genome retention mechanism. Nat Commun 17, 1964 (2026). https://doi.org/10.1038/s41467-026-69107-7
Keywords: bacteriophage T4, viral assembly, cryo-electron microscopy, genome packaging, host–virus interaction