Clear Sky Science · en
In situ structures of the portal-neck-tail complex of bacteriophage T4 inform a viral genome positioning mechanism
How a Virus Loads Its Genetic Spring
Bacteriophages—viruses that infect bacteria—are among nature’s most sophisticated nanomachines. One of the best studied, bacteriophage T4, must pack its long DNA genome into a tiny head and then fire it into a bacterium in a split second. This study reveals, almost atom by atom, how T4 carefully positions its DNA like a compressed spring inside its tail, ready to launch into a new host cell without losing so much as a single genetic “letter.” 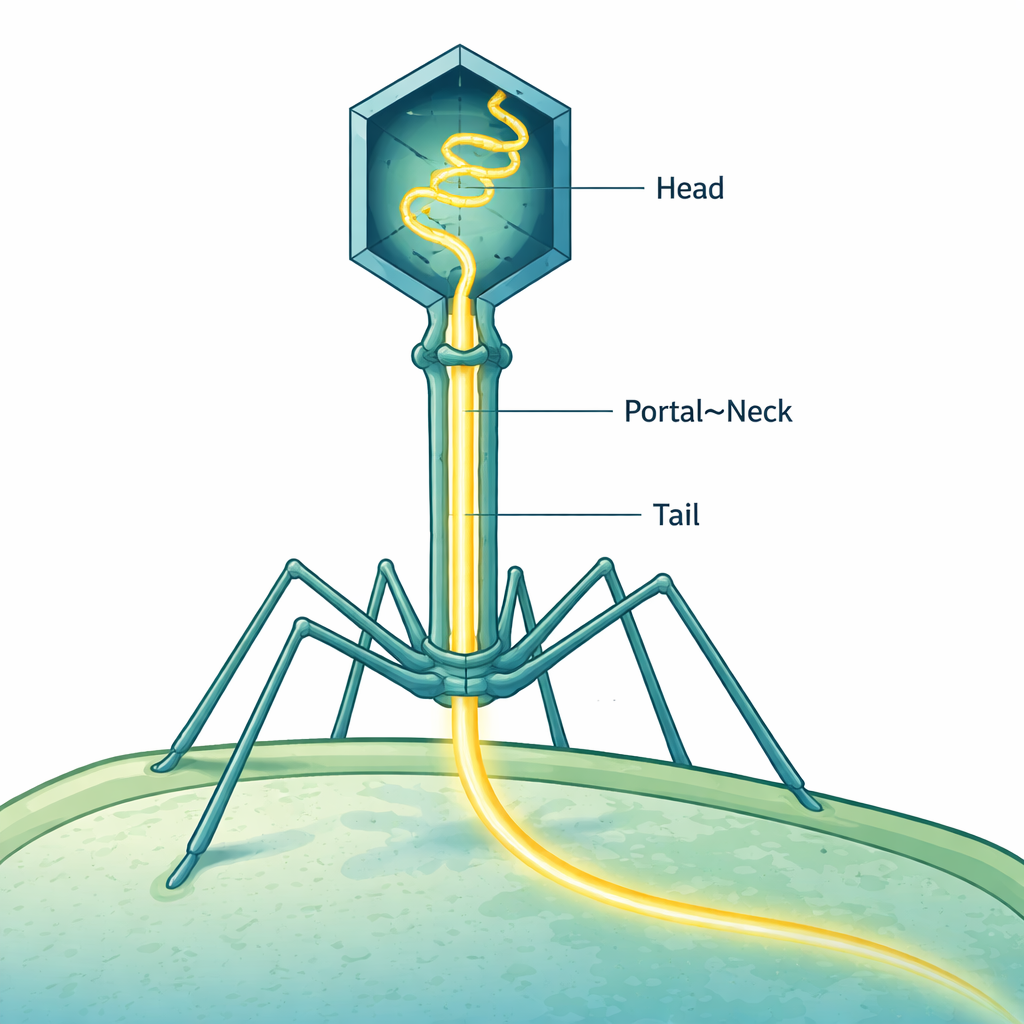
A High-Pressure Viral Head
Inside the phage’s protein shell, the DNA is crammed to near-crystal density, creating an internal pressure of roughly 25–35 atmospheres—similar to the pressure at deep ocean trenches. The authors used high-resolution cryo–electron microscopy to visualize what happens at the special “portal” through which DNA enters and exits the head. They found that when the head becomes full, the ring-shaped portal protein changes shape from a flying-saucer to a mushroom-like form and shifts downward relative to the shell. This pressure-driven shift appears to signal that enough DNA has been packaged, triggering the motor that was pumping DNA in to detach and exposing new docking sites for the next part of the viral machinery.
Building a Locking Neck Between Head and Tail
Once the DNA head is full, a neck structure assembles between head and tail, acting as both a connector and a valve. Two neck proteins, called gp13 and gp14, form rings beneath the portal. Gp13 swings part of its structure upward to clasp the portal and also reaches out to the surrounding shell, tying the head and neck tightly together and providing attachment points for decorative fibers. Gp14, sitting below, originally forms a double “genome gate” with a host protein called Hfq, effectively plugging the exit channel so the high-pressure DNA cannot leak out prematurely. In this sealed state, the DNA stops near the neck and is held in place while the rest of the virus finishes assembling.
Tail Docking Opens the Gate
The next step is to attach a pre-built tail, which consists of an inner tube surrounded by a contractile sheath and ends in a complex baseplate that recognizes the bacterial surface. At the top of this tail is a “tail terminator” ring, gp15, and just below it another ring, gp3, caps the inner tube. When this tail docks onto the neck, gp14 undergoes a dramatic rearrangement: its gate-forming loops rotate downward and clamp onto gp15, while an extended tail of gp14 wraps around gp15 to form a very large, charged interface. These movements eject the Hfq stopper and swing the gp14 loops out of the way, transforming the once-closed neck into a fully open channel that now lines up with the tail’s hollow tube.
DNA Captured by a Molecular Ruler
With the gate open, the DNA—still under high pressure—does not simply spill out. Instead, it travels about 17 nanometers down through the newly formed portal–neck–tail connector. At the junction with gp3 and the top of the tail tube, it encounters the “tape-measure protein” (TMP), a long coiled protein that originally acted as a ruler to set the tail’s length. The end of TMP has DNA-binding segments that grab the genome’s tip. Continued pressure from the packed head then pushes this DNA–TMP complex further down into the tail tube, compressing the TMP’s coiled-coil segments like a spring and moving the DNA tip to the bottom of the second ring of the tail tube. 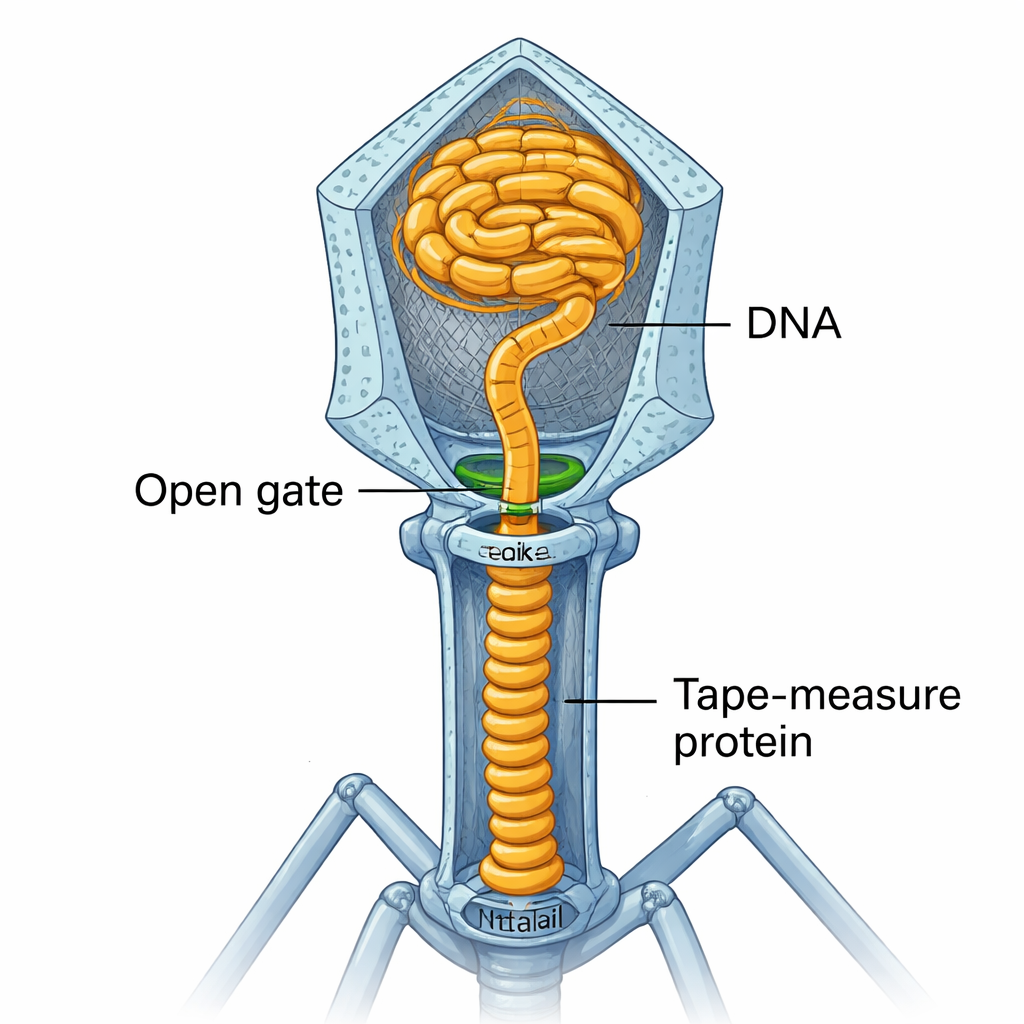
A Spring-Loaded Genome Ready to Fire
These structural snapshots reveal that after assembly, the phage genome is not simply stored in the head; it is deliberately positioned so that its leading end is suspended deep within an internal tunnel that runs from the head, through the neck, and into the tail. The DNA is held there by the compressed tape-measure protein and a “plug” at the baseplate, maintaining a metastable, spring-loaded state. When the baseplate senses and locks onto a bacterial receptor, it triggers changes that remove this plug and allow the compressed DNA–TMP complex to surge forward, guiding the genome smoothly into the host cell. In essence, the virus has evolved a pressure-powered loading and aiming system that ensures fast, complete, and reliable delivery of its genetic cargo.
Citation: Fokine, A., Zhu, J., Klose, T. et al. In situ structures of the portal-neck-tail complex of bacteriophage T4 inform a viral genome positioning mechanism. Nat Commun 17, 1965 (2026). https://doi.org/10.1038/s41467-026-69106-8
Keywords: bacteriophage T4, viral DNA packaging, cryo-electron microscopy, virus structure, phage infection mechanism