Clear Sky Science · en
Acute peritonitis-induced adipose CD127+ ILC1s express PD-L1 and ameliorate inflammation in mice
Why belly fat can help fight deadly infection
When bacteria leak from a torn gut into the abdomen, the body can spiral into a life‑threatening condition called sepsis. This study reveals an unexpected ally in that battle: a special set of immune cells hiding in belly fat that migrate in during early infection and act as a built‑in brake on runaway inflammation. Understanding how these cells work could inspire new treatments that calm dangerous infections without shutting the immune system down completely.
A dangerous leak in the abdomen
Peritonitis occurs when the lining of the abdomen becomes inflamed, most often because bacteria spill out from a perforated intestine. If this early storm of inflammation is not controlled, patients can develop sepsis, which still kills roughly one in five affected people despite modern care. Doctors can treat the infection with surgery and antibiotics, but they have few tools to steer the body’s own immune reaction away from harmful overdrive. The fat that drapes over the intestines, called mesenteric adipose tissue, is known to swarm toward sites of gut damage, hinting that it plays an active role in this early immune response.
Hidden guardians in belly fat
In recent years, scientists have discovered “innate lymphoid cells,” fast‑acting immune sentinels that live in many tissues. One subtype, called group 1 ILCs, normally helps launch strong inflammatory reactions. In this mouse study, the researchers used a standard surgical model of peritonitis to ask how these cells behave in mesenteric fat. They found that, six hours after the injury, this fat lit up with inflammatory signals and a surge of ILC1 cells, while other related cell types barely changed. This timing matched the peak of illness in the animals, suggesting that ILC1s were closely tied to the early inflammatory phase in the abdomen.
Traveling cells that change their nature
Digging deeper, the team used advanced single‑cell profiling to split the ILC1 population into subgroups. Under normal conditions, most belly‑fat ILC1s lacked a molecule called CD127 on their surface. During acute peritonitis, however, a new group of CD127‑positive ILC1s appeared and expanded. These newcomers made less of the potent inflammatory signal interferon‑gamma than their CD127‑negative cousins, marking them as unusually restrained. The scientists showed that many of these cells did not simply divide in place: instead, ILC1s from the spleen traveled through the bloodstream into mesenteric fat, guided in part by a chemical “homing” signal called CXCL10 and its partner receptor CXCR3. Once in the fat, they shifted their surface markers, gaining CD127 and adopting this quieter, more regulatory personality.
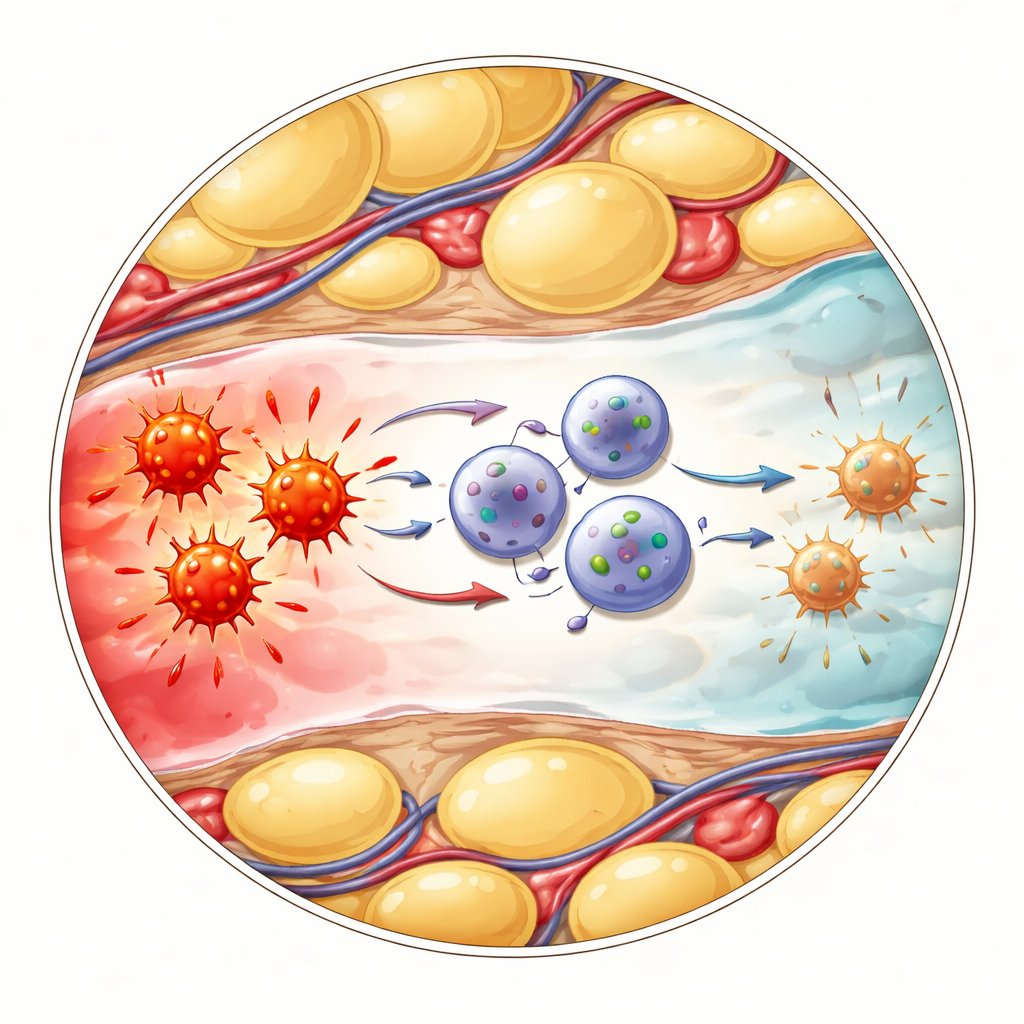
How calming cells talk to fiery cells
The next question was how these altered ILC1s actually cooled inflammation. The researchers focused on another immune group called gamma‑delta T cells, which respond quickly to danger and can release high amounts of the alarm molecule TNF. Single‑cell analyses suggested that CD127‑positive ILC1s and gamma‑delta T cells interact through a well‑known “brake” pathway involving the surface proteins PD‑L1 and PD‑1. In the inflamed belly fat, the newly arrived ILC1s strongly increased PD‑L1, while nearby gamma‑delta T cells displayed high levels of PD‑1. When the team blocked PD‑1 with antibodies, gamma‑delta T cells pumped out more TNF, confirming that this contact normally restrains their inflammatory output. Likewise, mice genetically engineered to lack ILC1s suffered worse illness, higher inflammatory markers, and more TNF from gamma‑delta T cells—effects that could be eased by directly blocking TNF.
What this means for future treatments
Putting these findings together, the authors propose a “spleen‑to‑belly‑fat” axis: soon after bacteria leak into the abdomen, ILC1s leave the spleen, home to mesenteric fat, and there transform into CD127‑ and PD‑L1‑rich cells that dampen TNF production by gamma‑delta T cells. Rather than fanning the flames, these specialized ILC1s act as local peacekeepers, preventing inflammation from overshooting and possibly helping stop peritonitis from progressing to deadly sepsis. In the long run, drugs that boost or mimic this regulatory circuit in abdominal fat could offer a new way to treat severe infections—tuning the immune response so it remains strong enough to fight germs, but not so strong that it endangers the patient.
Citation: Nagata, R., Akama, Y., Goncalves, P. et al. Acute peritonitis-induced adipose CD127+ ILC1s express PD-L1 and ameliorate inflammation in mice. Nat Commun 17, 2391 (2026). https://doi.org/10.1038/s41467-026-69100-0
Keywords: peritonitis, innate lymphoid cells, mesenteric adipose tissue, immune regulation, sepsis