Clear Sky Science · en
Repurposing nuclear receptors for ligand-responsive liquid condensate formation and gene regulation
Turning Hormones into On‑Demand Cell Switches
Our cells constantly listen to signals like hormones and vitamins and translate them into actions, for example turning genes on or off. This study shows how scientists can tap into that natural language to build synthetic “switches” that respond to real body signals and even form tiny liquid droplets inside cells to supercharge gene activity. Such hormone‑controlled switches could one day help create smarter gene therapies, precise biosensors, or living drugs that are tightly coupled to a patient’s own physiology.
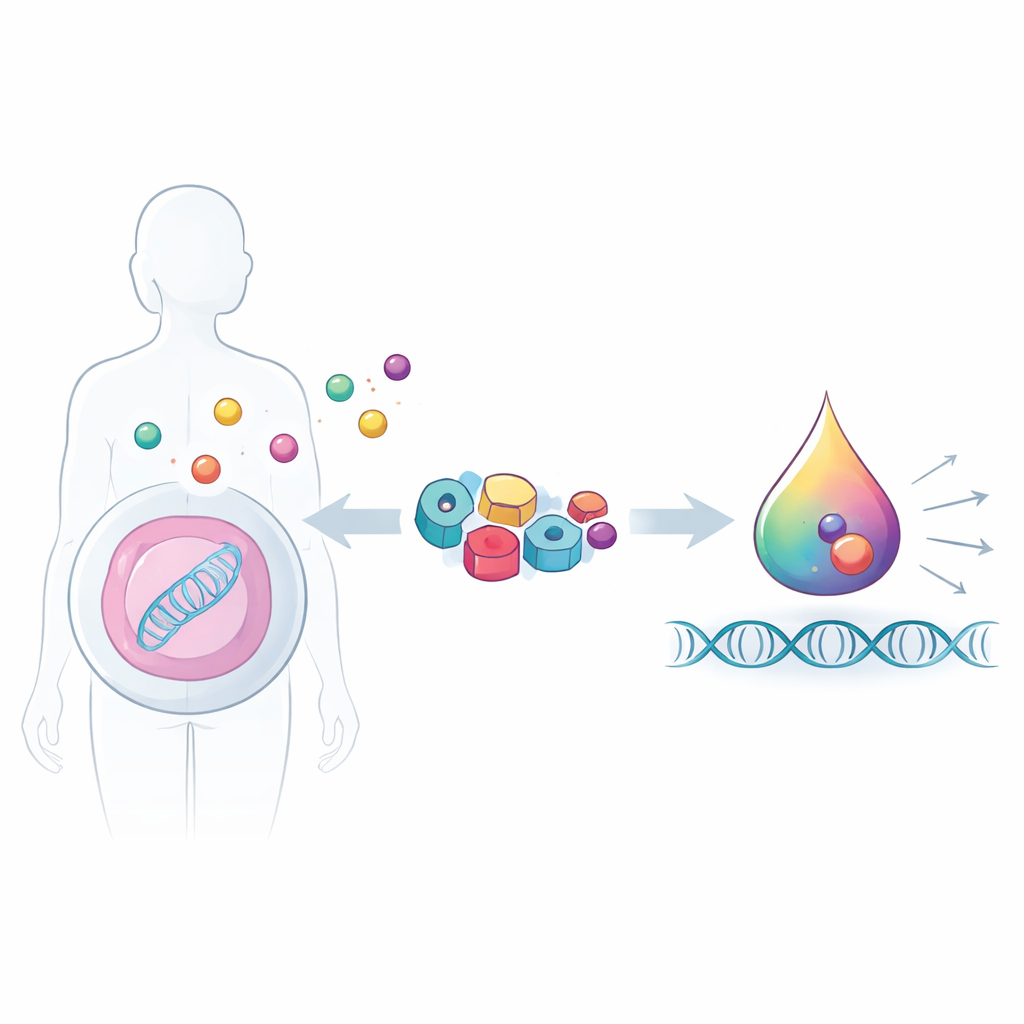
Using the Cell’s Own Signal Readers
Hormones and related molecules are read by a family of proteins called nuclear receptors, which normally sit in cells and control genes in response to signals like thyroid hormone, vitamin D, estrogen, and cortisol. Each receptor has a pocket that binds its specific hormone and a surface that grabs helper proteins, called coactivators, when the hormone is present. Instead of designing entirely artificial parts, the researchers reused these natural receptors and a small piece of a human coactivator protein (a short TIF2 peptide) as modular building blocks. This choice means their synthetic switches speak the same biochemical language as the body, making it easier to plug into existing signaling pathways.
Building Chemical On/Off Switches
The team first showed that hormone binding could bring two engineered protein fragments together on demand. They fused the ligand‑binding part of several nuclear receptors to one half of a split reporter enzyme and fused the TIF2 peptide to the other half. In human cells, adding the matching hormone caused the two halves to meet, restoring the reporter’s glow, while removing or blocking the hormone broke the connection. By pairing receptors with both their activating drugs (agonists) and blocking drugs (antagonists), they built two‑input switches that could be turned on by one compound and switched off by another, sometimes repeatedly and within minutes. They also used this strategy to move enzymes to specific places inside cells, changing local chemical levels on cue.
Controlling Genes with Multiplied Contacts
To turn these switches into gene controllers, the researchers connected them to a CRISPR‑based DNA‑binding platform. A “dead” Cas9 protein was guided to chosen DNA sites, while the nuclear receptor carried a strong gene‑activating domain. When hormone was added, the receptor latched onto TIF2 segments attached to dCas9, bringing the activator to the target gene. A single TIF2 segment produced only modest effects, but stringing together many copies created a multivalent landing pad that could recruit many receptors at once. This dramatically boosted gene activation—up to hundreds of times above baseline—and the response remained sensitive within hormone ranges similar to those found in the body. Antagonist drugs could sharply dial this activity down again, demonstrating reversible and precise control.
Creating Liquid Droplets that Turbocharge Expression
As the number of TIF2 copies increased, the team noticed a sharp threshold in performance, hinting that the proteins might be condensing into droplets. Many natural gene regulators form liquid‑like “condensates” that concentrate the machinery needed for transcription. The researchers purposely designed scaffolds that bundle many TIF2 motifs together using coiled‑coil peptides, encouraging multiple receptors to pack into the same spot when hormone is present. In living cells, this produced bright, spherical droplets that behaved like liquids: they fused, recovered after photobleaching, and could be dissolved by chemicals that disrupt weak protein interactions. Importantly, droplets appeared only under the right multivalent and hormonal conditions, and antagonist drugs could make them disappear, proving that droplet formation was chemically tunable.
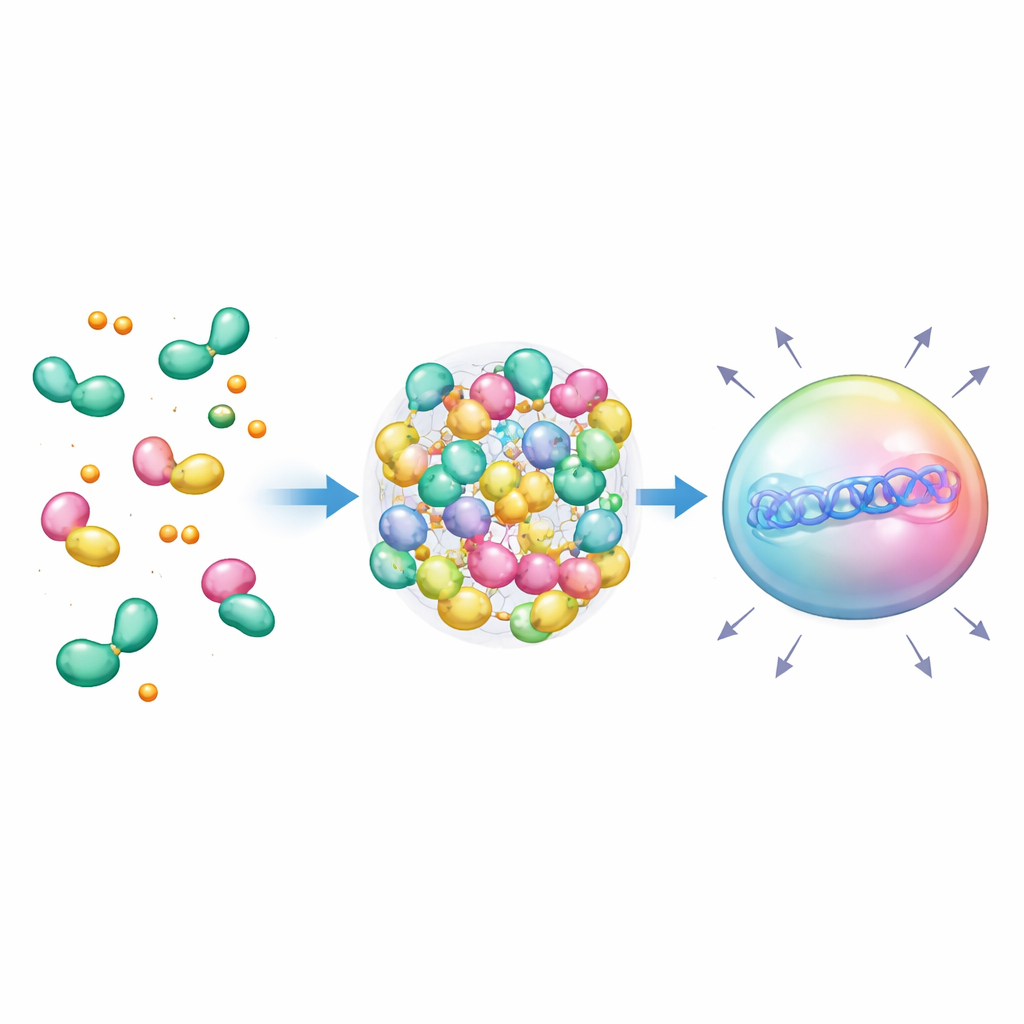
From Designer Droplets to Future Therapies
By fusing these hormone‑controlled droplets to DNA‑targeting CRISPR tools, the authors created nuclear condensates that sit directly over specific genes and massively amplify their activity—even when there is only a single DNA docking site. Because the components are human‑derived and respond to familiar signals like cortisol and estrogen, these systems could ultimately be wired into therapeutic circuits that respond automatically to a patient’s hormone levels. While careful testing will be needed to avoid interference with the body’s own receptors, this work illustrates a powerful concept: reusing natural hormone readers and liquid condensates as programmable switches to sense internal chemistry and drive tailored gene responses.
Citation: Rihtar, E., Fink, T., Ivanovski, F. et al. Repurposing nuclear receptors for ligand-responsive liquid condensate formation and gene regulation. Nat Commun 17, 2218 (2026). https://doi.org/10.1038/s41467-026-69099-4
Keywords: synthetic biology, nuclear receptors, hormone signaling, gene regulation, phase separation