Clear Sky Science · en
Binding properties of sulfur to enable solvent-free fabrication of high-performance polymer-free sulfur-carbon positive electrodes
Why This New Battery Recipe Matters
Lithium‑ion batteries power our phones, laptops, and electric cars, but making them is energy‑hungry, expensive, and relies on toxic solvents. This study explores a different kind of battery chemistry—lithium–sulfur—that promises much higher energy at lower cost, using sulfur, an abundant industrial by‑product. The researchers discovered a way to build the sulfur‑based electrode without any liquid solvents or polymer glues, instead letting sulfur itself act like the “glue.” Their approach could make future batteries cheaper, cleaner to manufacture, and longer‑lasting.
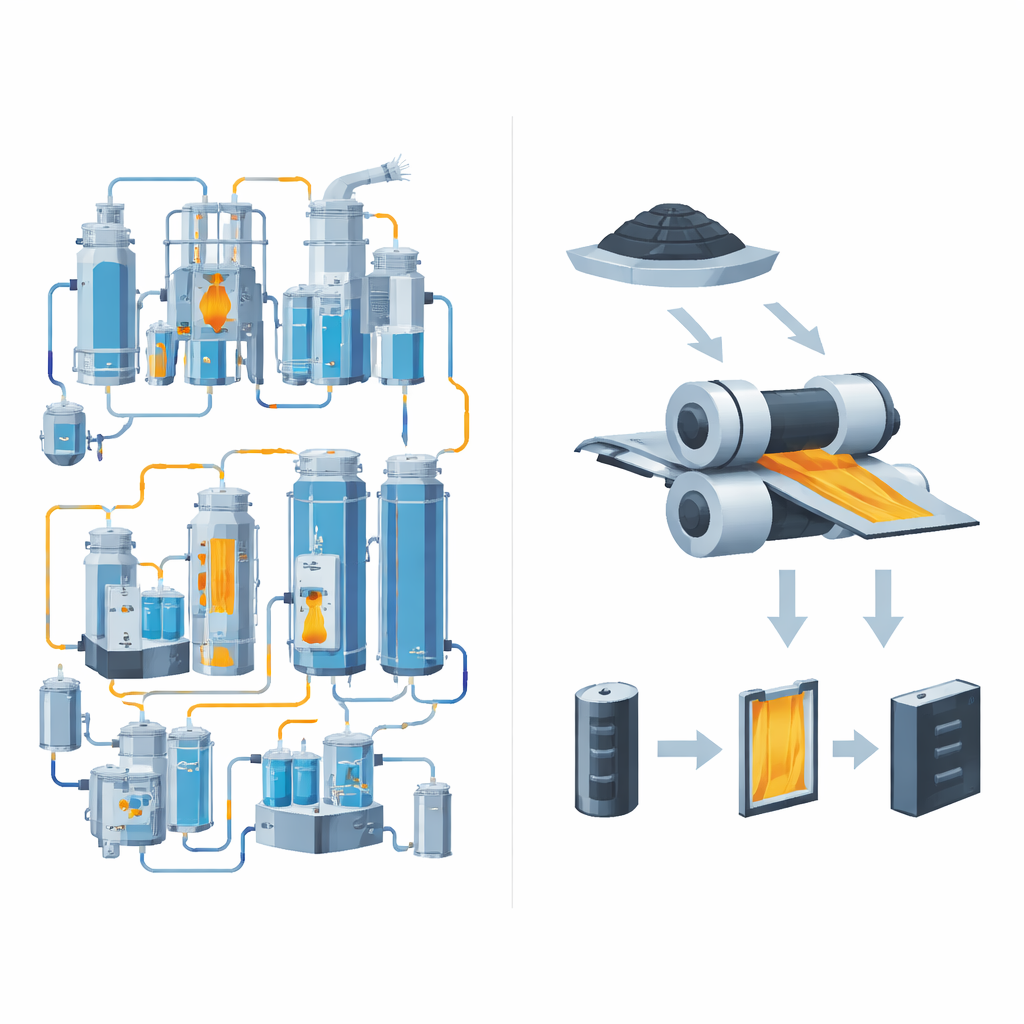
The Problem With Today’s Battery Factories
Most commercial batteries are made using a wet “slurry casting” process. Powders that store and conduct electricity are mixed with a polymer binder and dissolved in a solvent to form a thick paint, which is then spread on metal foil and dried in huge ovens. For lithium–sulfur batteries, this method brings several drawbacks. The solvent is often toxic and costly to recover, drying the slurry consumes large amounts of energy, and the polymer binder itself does not conduct electricity or ions, adding dead weight and resistance. In addition, the drying and re‑wetting cycles can damage the delicate porous structure that sulfur needs to work well, undermining the very advantages of this promising chemistry.
Letting Sulfur Become the Glue
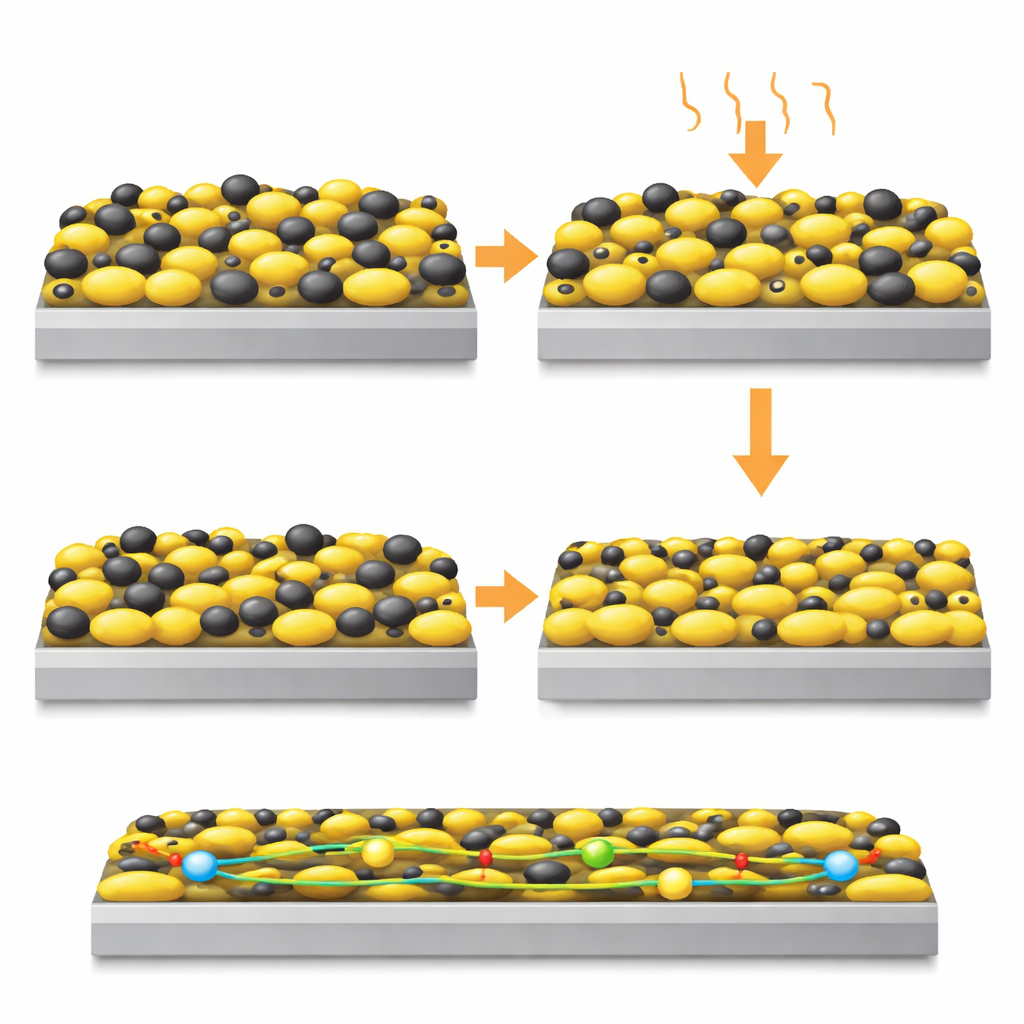
The team set out to remove both solvent and polymer binder from the equation entirely. Their key insight is that sulfur, normally thought of only as the active ingredient that stores energy, can also serve as the structural binder if treated correctly. Sulfur softens at relatively low temperatures, well below its melting point. By gently heating a mixture of sulfur and porous carbon and then pressing it onto aluminum foil, the softened sulfur flows just enough to lock particles together and stick them firmly to the metal. Careful experiments and computer simulations show that around 80 °C the sulfur particles deform and pack closely, dramatically reducing voids and forming a smooth, cohesive layer—without ever needing a separate glue.
Building Stronger Electrodes With a Simple Dry Press
To put this idea into practice, the researchers first prepared a sulfur–carbon powder in which some sulfur is tucked into tiny pores of the carbon, while additional sulfur forms slightly larger particles. This “dual” structure helps both electrical contact and mechanical binding. They then spread the dry powder directly onto aluminum foil and passed it through heated rollers. At room temperature, the result was a fragile, uneven film. At 80 °C, however, the film became mechanically robust, with a more uniform internal pore structure and straighter pathways for ions to move through. X‑ray imaging and microscopy revealed that electrodes pressed at the higher temperature had better contact among particles and with the foil, and absorbed liquid electrolyte more quickly and evenly than conventionally cast, binder‑containing films.
How the New Electrodes Perform in Real Cells
The team then tested these dry‑pressed sulfur–carbon electrodes inside coin‑cell and pouch‑cell batteries. Under challenging conditions—fast charging and discharging, and hundreds of cycles—the 80 °C‑pressed electrodes clearly outperformed both room‑temperature‑pressed versions and traditional slurry‑cast electrodes with polymer binder. At a moderate sulfur loading, the optimized dry electrodes delivered about 1300 to 600 milliamp‑hours per gram across a wide range of charge rates, and retained a reversible capacity of 932 milliamp‑hours per gram even after 500 cycles. In contrast, the slurry‑cast electrodes lost capacity far more quickly and showed rising internal resistance. Microscopy during operation showed that the dry‑pressed electrodes expanded and contracted more uniformly, avoiding cracking and delamination that plague traditional designs.
What This Means for Future Batteries
For non‑specialists, the core message is simple: this work shows a way to use sulfur itself as both the energy‑storing ingredient and the structural glue in a lithium–sulfur battery electrode. By relying on a straightforward dry‑pressing step instead of solvent‑based coating and polymer binders, the method could cut electrode fabrication costs by more than half, sharply reduce energy use and emissions, and avoid hazardous chemicals. At the same time, the resulting electrodes last longer and store more energy per gram than their conventional counterparts. If adapted to large‑scale production, this solvent‑ and binder‑free process could help turn high‑energy lithium–sulfur batteries into practical, sustainable power sources for electric vehicles and grid storage.
Citation: An, Y., Kim, K., Lee, YJ. et al. Binding properties of sulfur to enable solvent-free fabrication of high-performance polymer-free sulfur-carbon positive electrodes. Nat Commun 17, 2360 (2026). https://doi.org/10.1038/s41467-026-69097-6
Keywords: lithium–sulfur batteries, dry electrode fabrication, sulfur–carbon cathodes, solvent-free processing, energy storage materials