Clear Sky Science · en
Extended Shine-Dalgarno motifs govern translation initiation in Staphylococcus aureus
How a Hospital Germ Fine-Tunes Its Protein Factories
Staphylococcus aureus is a common germ that can cause anything from mild skin infections to life‑threatening disease. To survive in the body and resist treatment, it must constantly rewire which proteins it makes. This paper uncovers how S. aureus controls the very first step of protein production in a way that differs from textbook bacteria like Escherichia coli, and shows how this control is linked to biofilm formation, a major contributor to chronic and antibiotic‑tolerant infections.
A Hidden “Handshake” Between Gene and Ribosome
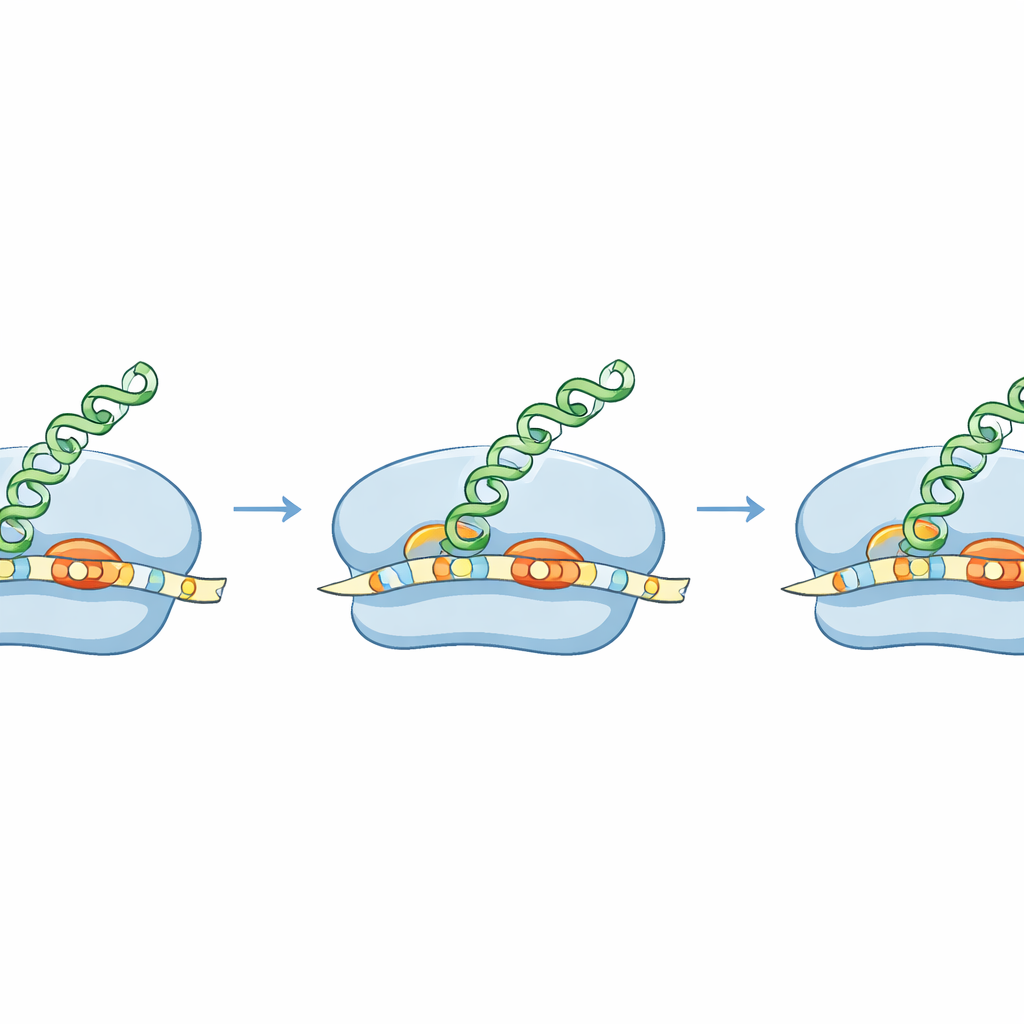
To turn a gene into a protein, bacteria use ribosomes, molecular machines that scan a messenger RNA (mRNA) to find where to start reading. Many bacterial mRNAs contain a short sequence, long known as the Shine–Dalgarno region, that helps the ribosome dock in the right place. By freezing ribosomes at the moment they begin translation and sequencing the protected RNA fragments, the authors created a high‑resolution map of start sites across the S. aureus genome. They discovered that S. aureus often uses an unusually long version of this docking sequence, positioned very close to the start signal, forming an extended “zipper” between mRNA and ribosomal RNA. This extended pairing is much more prominent than in E. coli and appears to be a hallmark of how S. aureus chooses where to begin.
Species-Specific Rules for Starting Protein Synthesis
The team showed that this extended docking helix has real consequences. In several natural S. aureus genes, the first few letters can be read in more than one way, offering rival places to start translation. Using purified ribosomes, cell‑free systems, and live cells, the researchers compared how S. aureus and E. coli handle these ambiguous starts. S. aureus ribosomes reliably chose the same, “correct” start used in the bacterium itself, guided by the long docking helix and the preferred spacing between this helix and the start site. E. coli ribosomes, in contrast, favored a different nearby start that would produce an alternative protein. This means the same mRNA can drive different proteins in different species, opening the door to engineering messages that are readable only by selected bacteria.
New Tiny Proteins and Weak Start Signals
Because their method pinpoints the exact arrival of ribosomes at start sites, the authors could also uncover many short, previously missed genes. They identified dozens of small open reading frames, including short “upstream” sequences that sit just before larger, known genes. Some of these short regions likely encode tiny proteins with their own functions; others seem to act mainly as regulators. The study further revealed that S. aureus sometimes begins translation at “non‑canonical” start triplets that are usually considered poor signals. In these cases, strong and extended docking motifs appear to compensate for the weak start, throttling protein production so that certain factors, including metabolic regulators, are made only sparingly or in response to growth conditions.
A Molecular Sensor Linking Nutrients to Biofilm Growth
One short upstream element, named rbfL, emerged as a striking example of translational control tied to virulence. It lies just before rbf, a gene that encodes a transcription factor promoting biofilm formation. The rbfL sequence encodes a tiny “leader” peptide rich in arginine, including very rare arginine codons that are decoded by scarce transfer RNAs. When arginine is limited, these codons are translated slowly and ribosomes stall over the region that overlaps the docking site for rbf itself, physically blocking new ribosomes from starting rbf translation. When arginine or its corresponding transfer RNA is abundant, the stall is relieved and rbf is produced more efficiently. In growth experiments, adding extra arginine boosted biofilm formation, tying this molecular sensor directly to a community‑lifestyle that resists antibiotics.
Why These Findings Matter
This work shows that S. aureus has evolved extended docking motifs and tiny upstream elements to fine‑tune where and when protein synthesis begins. These features not only distinguish it from model bacteria like E. coli but also couple nutrient sensing to the control of biofilm regulators. For a lay reader, the key takeaway is that the bacterium’s “start buttons” for protein production are more elaborate than previously thought—and that these specialized switches help determine when S. aureus becomes more dangerous. Understanding these unique initiation rules could guide the design of species‑specific antibiotics or genetic tools that selectively disrupt harmful bacteria while sparing beneficial ones.
Citation: Kohl, M.P., Bahena-Ceron, R., Chane-Woon-Ming, B. et al. Extended Shine-Dalgarno motifs govern translation initiation in Staphylococcus aureus. Nat Commun 17, 2678 (2026). https://doi.org/10.1038/s41467-026-69079-8
Keywords: Staphylococcus aureus, translation initiation, Shine-Dalgarno, small ORFs, biofilm regulation