Clear Sky Science · en
A combinatorial synthetic strategy for developing genome-editing protein-delivery agents targeting mouse retina
New hope for treating inherited blindness
Many forms of inherited blindness begin with a single faulty gene in the light-sensing cells at the back of the eye. Modern gene-editing tools such as CRISPR can, in principle, correct these mistakes, but getting the editing machinery safely into the right cells has been a major obstacle. This study describes a new way to ferry powerful gene editors directly into the mouse retina using specially designed fat-like particles, raising the prospect of one-time treatments that restore vision in people with genetic eye diseases.
Why delivering gene editors is so hard
CRISPR-based tools can now change individual letters of DNA without cutting both strands, an advance that makes them attractive for treating genetic disorders. But these tools are bulky protein–RNA machines, electrically charged and fragile in the body. Current delivery methods rely largely on modified viruses or on particles that carry genetic blueprints for the editor rather than the editor itself. Viruses can provoke immune reactions and have strict size limits, while delivering messenger RNA works very well in the liver but remains inefficient in many other tissues, including the eye. Directly injecting the pre-assembled protein–RNA complexes is conceptually cleaner and safer, yet they have lacked a suitable vehicle to help them cross cell membranes and reach their targets.
A dye-inspired shuttle for proteins
The researchers turned to an unexpected starting point: Coomassie Brilliant Blue, a deep-blue dye commonly used in biology labs and already approved for certain eye surgeries. This dye sticks tightly to many different proteins. The team chemically linked Coomassie "heads" to fat-like "tails" to create a family of new molecules called lipidoids. In water, these lipidoids tend to cluster into particles, with the oily parts buried inside and the Coomassie groups exposed outside, where they can grab onto proteins. By varying the length, branching and charge of the tails, the scientists built dozens of candidate compounds designed to bind gene-editing proteins on one side and interact with cell membranes on the other.
Testing protein delivery to eye cells
To see which designs actually worked, the group first used a simple test protein called Cre recombinase that flips a genetic switch from green to red or red to green fluorescence in engineered cells and mice. Several Coomassie lipidoids carried Cre efficiently into cultured cells, turning large fractions of cells from green to red. When injected under the retina of reporter mice, selected compounds triggered striking color changes in both the pigment layer that supports vision and in the light-sensing photoreceptors, showing that the proteins reached the very cells most affected in many blinding diseases. These early experiments also revealed that subtle changes in the lipidoid structure could shift which cell types were targeted, hinting that future versions might be tuned for particular retinal layers.
Boosting gene editing and rescuing vision
Next, the team loaded their system with an adenine base editor, a refined CRISPR variant that can correct the specific single-letter mutation causing vision loss in rd12 mice, a model of a severe childhood blindness. On its own, the editor protein–RNA complex penetrated cells poorly. One lipidoid, dubbed CBB11, improved delivery but tended to clump in solution. To stabilize it, the researchers embedded CBB11 into small, well-defined liposomes—hollow spheres made of several types of lipids, including clinically used components from mRNA vaccines. In these formulations, CBB11 on the liposome surface grabbed the protein part of the editor, while other lipids helped bind the RNA guide, creating a cooperative shell that held the whole complex on the particle exterior.
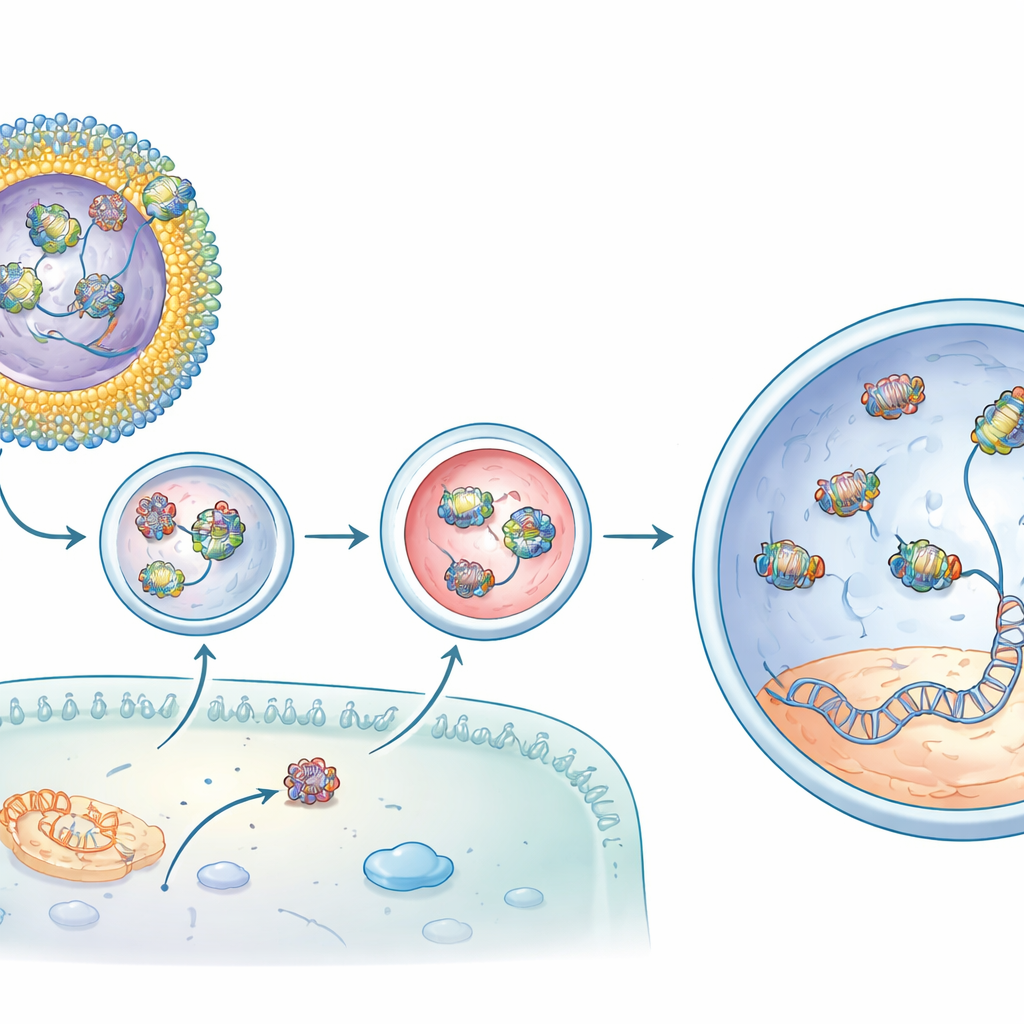
From molecular repair to restored light responses
When these CBB11-containing liposomes carrying the base editor were injected beneath the retina of rd12 mice, the results were striking. Compared with editor injected alone, the optimized formulation increased the desired DNA correction in the target gene by well over one order of magnitude and produced even higher levels of corrected messenger RNA. Chemical analysis showed recovery of the light-sensitive retinal pigment that is missing in the disease. Most compelling, electrical recordings from the animals’ eyes revealed that their rod cells regained much of their ability to respond to dim light—up to about three quarters of a normal mouse’s signal in the best cases. Off-target changes elsewhere in the genome were at background levels, suggesting that the editing was both potent and precise.
What this could mean for future eye therapies
To a non-specialist, the key message is that the authors have built a reusable "shuttle bus" for gene-editing proteins that works directly in the living eye. By decorating safe, vaccine-like fat particles with a protein-gripping dye, they created a system that can capture different editors on its surface, deliver them into retinal cells, correct disease-causing mutations, and measurably restore vision in mice after a single treatment. Although more work is needed to fully assess safety, improve targeting, and adapt the method for human use, this platform points toward future one-time precision medicines for inherited retinal diseases and possibly other genetic conditions where careful, localized DNA repair could make the difference between blindness and sight.
Citation: Zhang, J., Hołubowicz, R., Smidak, R. et al. A combinatorial synthetic strategy for developing genome-editing protein-delivery agents targeting mouse retina. Nat Commun 17, 2479 (2026). https://doi.org/10.1038/s41467-026-69077-w
Keywords: retinal gene therapy, CRISPR base editing, liposomes, protein delivery, inherited blindness