Clear Sky Science · en
Insights into the structure and modulation of human TWIK-2
Why this tiny gate in our cells matters
Deep inside our cells, microscopic gates made of protein control the flow of charged atoms, shaping how nerves fire, how blood vessels tighten, and how our immune system reacts. One such gate, called TWIK-2, has been linked to high blood pressure in the lungs and harmful inflammation, but until now scientists knew very little about how it is built or how to control it with medicines. This study reveals the detailed structure of human TWIK-2, shows how it behaves electrically, and outlines how it could be targeted to develop new anti‑inflammatory drugs.
A quiet channel with big health implications
TWIK-2 belongs to a large family of “leak” potassium channels that help set the resting voltage of cells by letting potassium ions drift across the cell membrane. While other members of this family, such as TREK and TASK channels, have been intensively studied and linked to pain and mood, TWIK-2 has remained obscure because it is hard to record and expresses poorly in standard lab systems. Yet animal and cell studies have tied TWIK-2 to pulmonary hypertension, acute lung injury, hearing loss, and activation of an immune complex called the NLRP3 inflammasome, which drives damaging inflammation in sepsis and other diseases. Understanding how TWIK-2 works is therefore essential both for basic biology and for designing targeted therapies.
Seeing the gate at atomic detail
The researchers expressed full-length human TWIK-2 in human cell lines and used single-particle cryo–electron microscopy to determine its three‑dimensional structure at a resolution of about 3.7 angstroms. The channel forms a two-part assembly that together creates a central pore for potassium ions to pass through. Each half contains four membrane-spanning segments and two “pore helices” that shape a narrow region called the selectivity filter, where potassium is distinguished from other ions. Above the pore, two arching “cap” structures create two separate entry paths for ions from outside the cell. The structure also reveals subtle asymmetries between the two sides of the pore that give TWIK-2 a pseudo–four-part arrangement, a hallmark of this channel family.
Hidden lipids and a finely tuned ion pathway
Along the ion conduction path, the team mapped how potassium ions move from the cell interior, through a hydrophobic “cuff,” up the selectivity filter, and out beneath the cap. The narrowest point is still wide enough for potassium to travel in single file. Although the protein was purified without added fat molecules, the cryo‑EM maps showed tube-like densities nestled near the lower part of the pore and in side pockets between helices. These likely represent lipids or detergent tails that stuck to TWIK-2 during purification, hinting that natural membrane fats may lodge in these grooves and subtly shift the shape of nearby helices. Such shifts, especially in a helix called M4 and in the selectivity filter loops, are thought to tune whether the channel is open, closed, or somewhere in between.
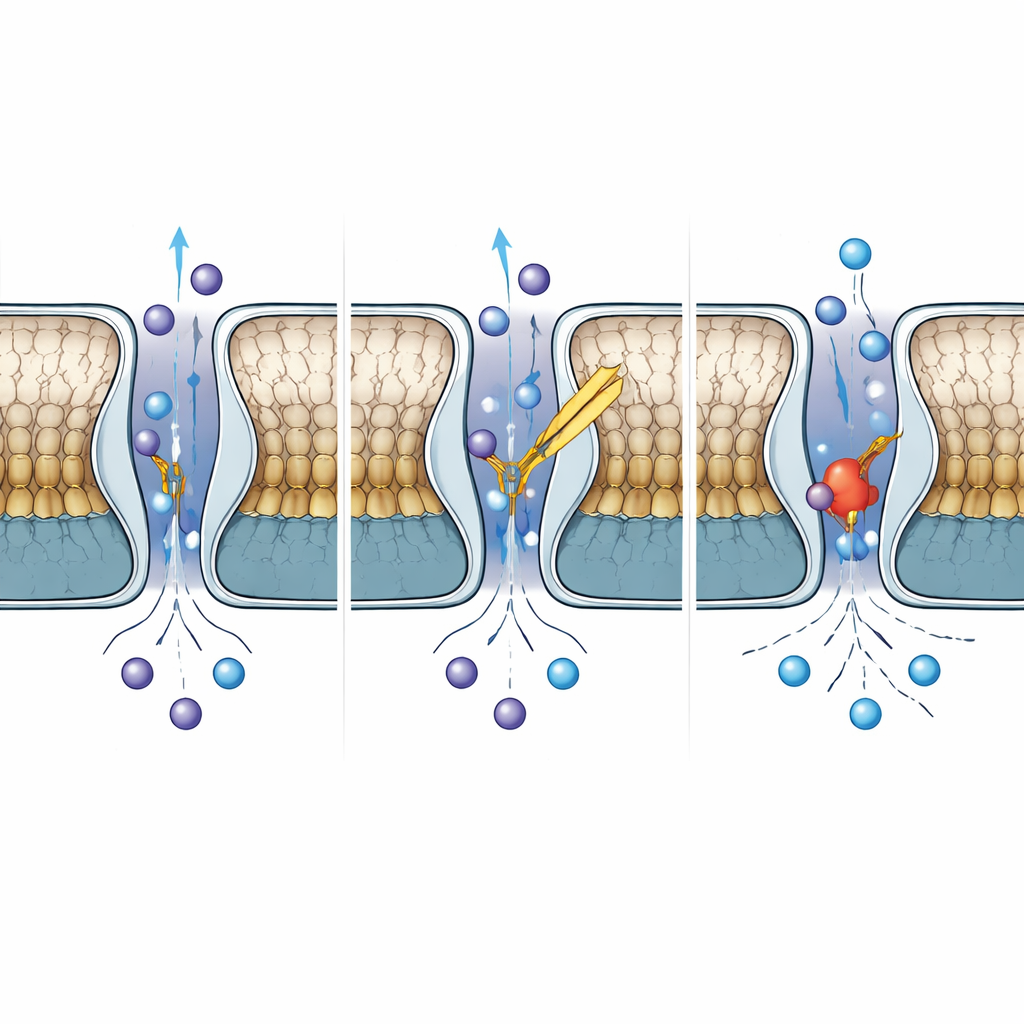
Probing the gate with mutations and blockers
To connect structure to function, the scientists used an automated patch‑clamp system to measure currents from cells expressing either normal TWIK-2 or versions with specific amino acids altered. They found that TWIK-2 is strongly voltage‑dependent but, unlike its cousin TWIK-1, largely insensitive to changes in external acidity. Mutations at two conserved threonine residues at the base of the selectivity filter made the channel open faster and carry more current, suggesting these sites act as key hinges that stabilize the closed state. Changes at the entrance and exit of the pore altered how quickly the channel turned on and off, revealing how tightly controlled the ion pathway is. The team then tested four known small molecules that modulate related channels. Two of them, ML365 and NPBA, potently blocked TWIK-2, while the others had no effect. By comparing how various mutations changed drug sensitivity, the authors infer that these compounds likely bind just beneath the selectivity filter and in nearby cavities, and that different drugs use overlapping but distinct binding modes.
How TWIK-2 differs from its close relatives
Although TWIK-2 shares more than half of its sequence with TWIK-1, their behavior diverges in important ways. When the authors compared TWIK-2 with published TWIK-1 structures at neutral and acidic pH, they found that TWIK-2’s cap region resembles the acid‑inhibited form of TWIK-1, yet its selectivity filter remains arranged like the conducting, open form. A single histidine in TWIK-1, known to sense protons and swing into the pore to block ion flow at low pH, is replaced by a tyrosine in TWIK-2. Even when the team reintroduced a histidine at this position in TWIK-2, the channel did not become pH-sensitive, showing that pH control in these channels relies on a broader network of residues and movements, not just one “switch” amino acid.
From structure to future therapies
Taken together, this work provides the first high‑resolution picture of human TWIK-2 and maps out how specific structural features shape its ion flow, response to voltage, and susceptibility to small‑molecule blockers. The discovery that existing compounds can selectively inhibit TWIK-2, combined with a robust automated screening platform, opens the door to large‑scale searches for safer and more potent drugs. Because TWIK-2 activity in immune cells has been tied to runaway inflammation, especially in the lungs, such drugs could form the basis of new anti‑inflammatory treatments that act by gently “turning down” this tiny but powerful gate in the cell membrane.
Citation: Ma, Q., Hernandez, C.C., Navratna, V. et al. Insights into the structure and modulation of human TWIK-2. Nat Commun 17, 2201 (2026). https://doi.org/10.1038/s41467-026-69072-1
Keywords: TWIK-2 potassium channel, two-pore-domain K2P channels, cryo-EM ion channel structure, potassium channel modulators, pulmonary inflammation