Clear Sky Science · en
Colonic spatial single-cell proteomics and murine models link mitochondrial dysfunction to dimeric IgA-secreting plasma cell deficiency in Crohn’s disease
Why gut antibodies matter in Crohn’s disease
Crohn’s disease is best known for painful flare‑ups of intestinal inflammation, but long before symptoms spike, subtle changes can occur in how the immune system protects the gut. This study asks a simple but important question: do people with Crohn’s disease in remission still make the right kind of antibodies to keep gut microbes peacefully in check? By combining patient samples, high‑resolution tissue profiling, metabolic analyses, and mouse models, the authors uncover a hidden weakness in the gut’s antibody shield that is tied to cellular energy production.
A protective coating that comes up short
Our intestines rely on a special antibody called secretory IgA that is released into the mucus lining the gut. IgA, especially in its “double” form called dimeric IgA, coats bacteria and other microbes, helping keep them at the surface rather than deep in the tissue. In this work, the researchers examined colonic biopsies, blood, and stool from people with Crohn’s disease in remission and from non‑inflamed controls. Inside the colon tissue, Crohn’s patients actually had more B cells and antibody‑producing cells and higher levels of IgA‑related genes and proteins. Yet, when the team measured IgA in stool, they found the opposite of what one might expect: secretory IgA in the gut lumen was significantly reduced. 
Stuck halfway: antibody‑producing cells that do not fully mature
The cells that secrete IgA—plasma cells—normally mature through defined stages from active, short‑lived precursors to long‑lived specialists that pump out large amounts of dimeric IgA. Using spatial single‑cell proteomics, which maps many proteins on individual cells within intact tissue, the authors could distinguish immature and fully mature plasma cell subsets in the colon. In Crohn’s patients, early plasmablasts and immature plasma cells were expanded, while the most mature plasma cells were reduced. Biochemical analyses confirmed that the ratio of dimeric to monomeric IgA was lower in Crohn’s colons, and each antibody‑producing cell secreted less dimeric IgA on average. When the team isolated switched memory B cells from the colon and pushed them to become plasma cells in culture, cells from Crohn’s patients failed to upregulate key maturation markers and produced less total and dimeric IgA, even though IgG production remained largely normal. This suggests an intrinsic block in the maturation program specifically for IgA‑secreting plasma cells.
Energy factories under strain
To understand what drives this block, the researchers turned to cell metabolism. They found that plasma cells in Crohn’s tissue expressed higher levels of CD38, an enzyme on the cell surface that consumes NAD+, a central fuel for mitochondrial energy production. Genes involved in mitochondrial oxidative phosphorylation—the main way cells generate ATP from nutrients—were broadly downregulated in colonic tissue from Crohn’s patients, and plasma metabolite patterns were consistent with a shift away from efficient mitochondrial respiration. In ex vivo experiments, Crohn’s‑derived plasma cells consumed little glucose, released less lactate, and showed reduced expression of mitochondrial energy genes, indicating a metabolically “quiescent” or weakened state. 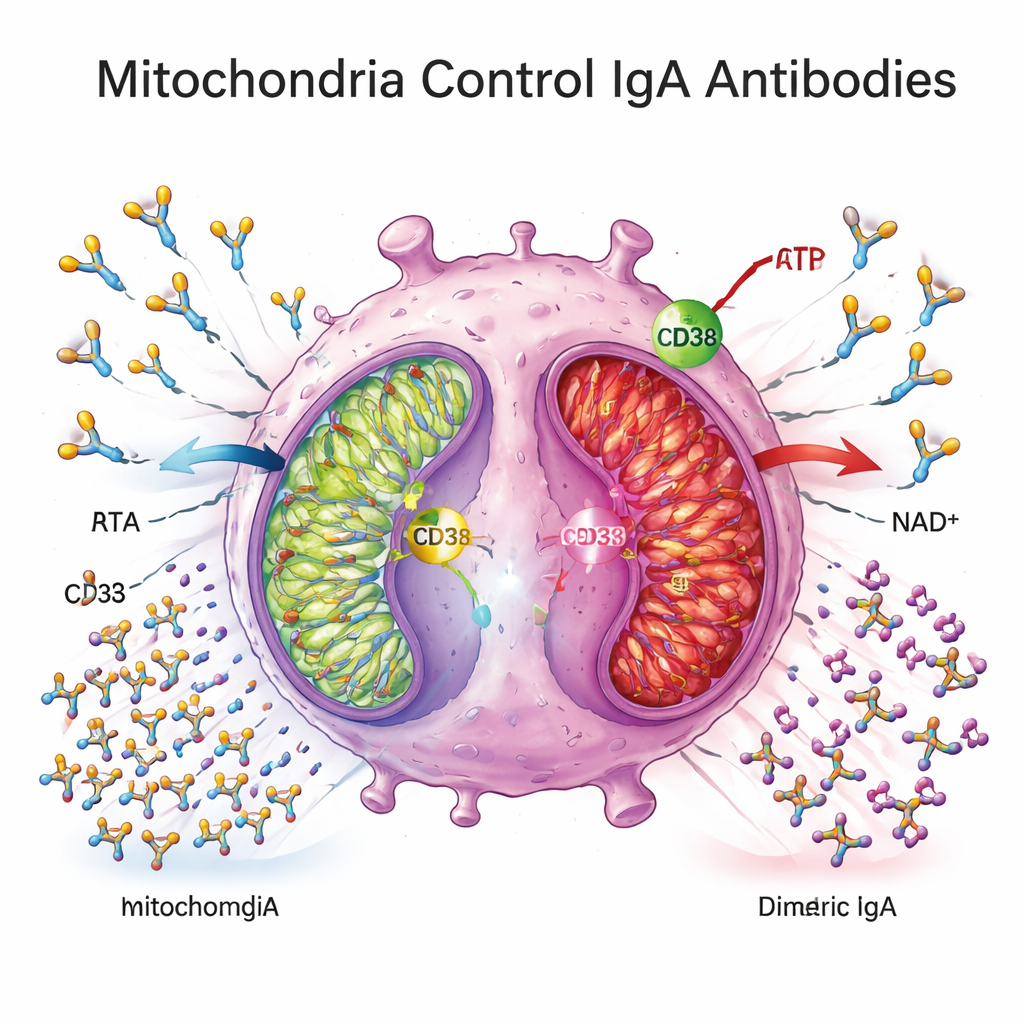
Clues from mouse models and diet
Mouse models lent further support. Mice carrying a defined mitochondrial mutation that impairs the ATP‑producing enzyme complex V had fewer mature plasma cells in gut‑associated lymphoid tissues, reduced expression of plasma‑cell markers in the colon, and lower levels of both monomeric and dimeric IgA in colonic tissue. Conversely, healthy mice fed a glucose‑free, high‑protein diet that enhances mitochondrial activity in the colon developed more IgA‑producing cells and higher fecal IgA. These complementary experiments tie mitochondrial performance directly to the ability of plasma cells to mature and maintain a robust IgA barrier in the gut.
What this means for people with Crohn’s disease
Taken together, the study paints a picture in which Crohn’s disease, even during clinical remission, is associated with an overabundance of immature B‑lineage cells in the colon and a shortage of fully mature, energy‑demanding plasma cells that secrete dimeric IgA into the mucus. The gut may look calm, but its antibody shield is thinner and less effective. By linking this defect to mitochondrial dysfunction—possibly driven in part by heightened CD38 activity—the work suggests that therapies aimed at restoring cellular energy metabolism or fine‑tuning CD38 signaling could help rebuild the mucosal IgA barrier. For patients, this raises the prospect that future treatments might not only suppress inflammation when it appears, but also strengthen the gut’s frontline antibody defenses to prevent flare‑ups from arising in the first place.
Citation: Raschdorf, A., de Almeida, L.N., Solbach, P. et al. Colonic spatial single-cell proteomics and murine models link mitochondrial dysfunction to dimeric IgA-secreting plasma cell deficiency in Crohn’s disease. Nat Commun 17, 1590 (2026). https://doi.org/10.1038/s41467-026-69069-w
Keywords: Crohn’s disease, gut antibodies, IgA, mitochondria, intestinal immunity