Clear Sky Science · en
Tenascin C+ myofibroblasts exacerbate vascular neointimal hyperplasia by propagation of nerve-macrophage interactions in mice
When Blood Vessels Heal the Wrong Way
Procedures that reopen clogged arteries, such as angioplasty and bypass surgery, save countless lives. Yet they often leave behind a hidden problem: over time, the treated vessel can scar inward, narrowing again and putting patients back at risk. This study in mice uncovers an unexpected three-way conversation between support cells, nerves, and immune cells in the vessel wall that drives this harmful over-healing, and points to a molecule called tenascin C as a promising target to break the cycle.
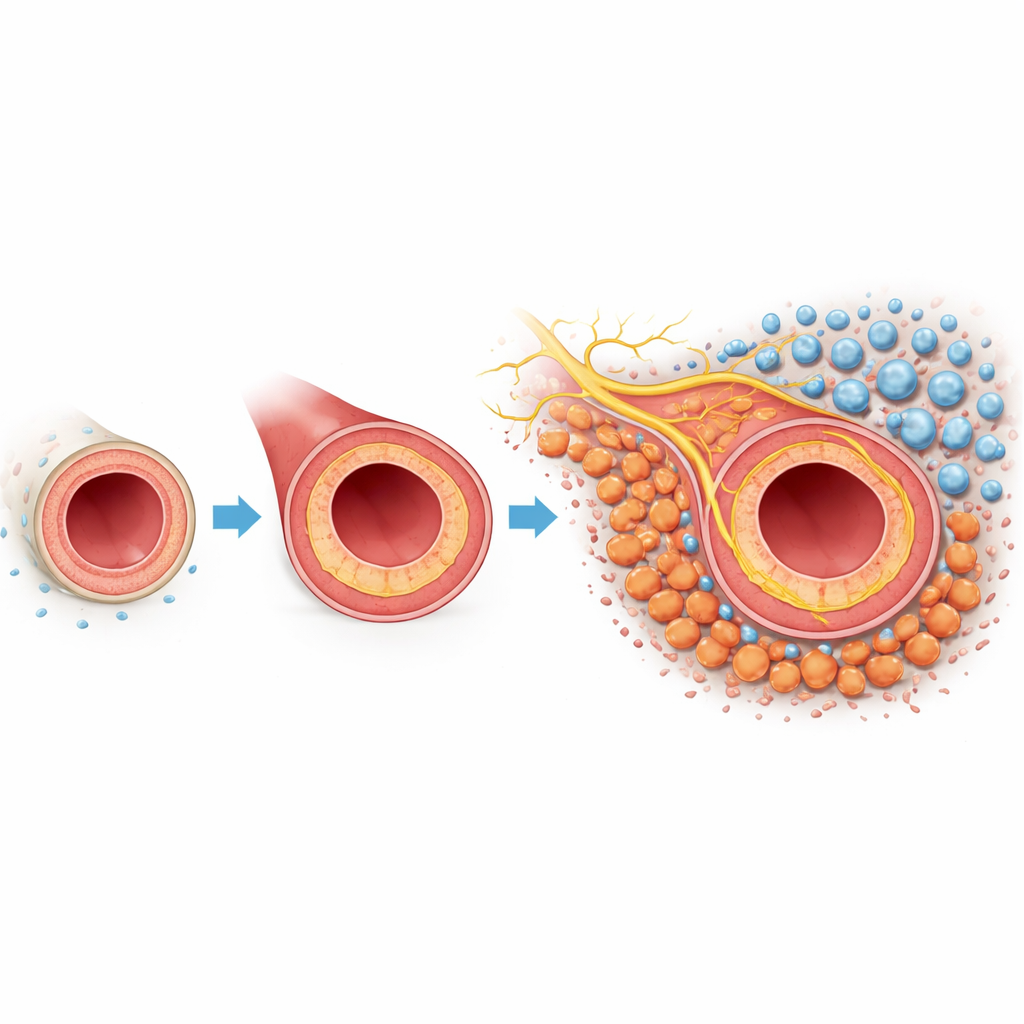
A Closer Look at Problem Scarring in Arteries
When an artery is injured or manipulated during surgery, the inner lining can grow back too aggressively. This inward growth, called neointimal hyperplasia, thickens the vessel wall and shrinks the channel where blood flows. The outermost layer of the vessel, the adventitia, contains fibroblasts—support cells that normally maintain the vessel’s structure. In this mouse model, where the carotid artery was partially tied off to mimic injury, the researchers used single-cell genetic profiling and spatial mapping to catalog every major cell type involved. They found that fibroblasts were especially abundant and changed their behavior dramatically after injury, hinting that these cells might be central players in the scarring response.
The Rise of Scar-Forming Tenascin C Cells
Diving deeper into the fibroblast population, the team discovered a distinct subset that appeared only after injury. These cells carried hallmarks of myofibroblasts—high levels of the contractile protein α-smooth muscle actin and another marker called periostin—signaling that they had switched into a scar-making mode. Crucially, this subset also produced large amounts of tenascin C, a matrix protein usually scarce in healthy adult tissues but abundant in disease and tissue repair. Lab experiments showed that exposing normal adventitial fibroblasts to transforming growth factor beta or extra tenascin C pushed them to become these tenascin C–positive myofibroblasts. The cells then secreted even more tenascin C, creating a self-reinforcing loop through a specific surface receptor pair, integrin αvβ1, that steadily expanded the scar-forming population.
Nerves and Immune Cells Move Into the Neighborhood
The story did not end with scar formation. Using 3D whole-mount imaging and spatial transcriptomics, the researchers observed that the tenascin C–rich myofibroblasts clustered near sprouting nerve fibers and immune cells called macrophages in the outer vessel wall. Injured arteries showed dense, tangled sensory nerves and more synapse-like structures than normal, both in mice and in human artery samples with neointimal hyperplasia. Macrophages in these regions bore an inflammatory signature, producing molecules such as IL-1β and TGF-β1 that can further fuel scarring. Together, the data painted a picture of a reorganized microenvironment in which scar-forming cells, nerves, and macrophages physically intermingle and influence each other.
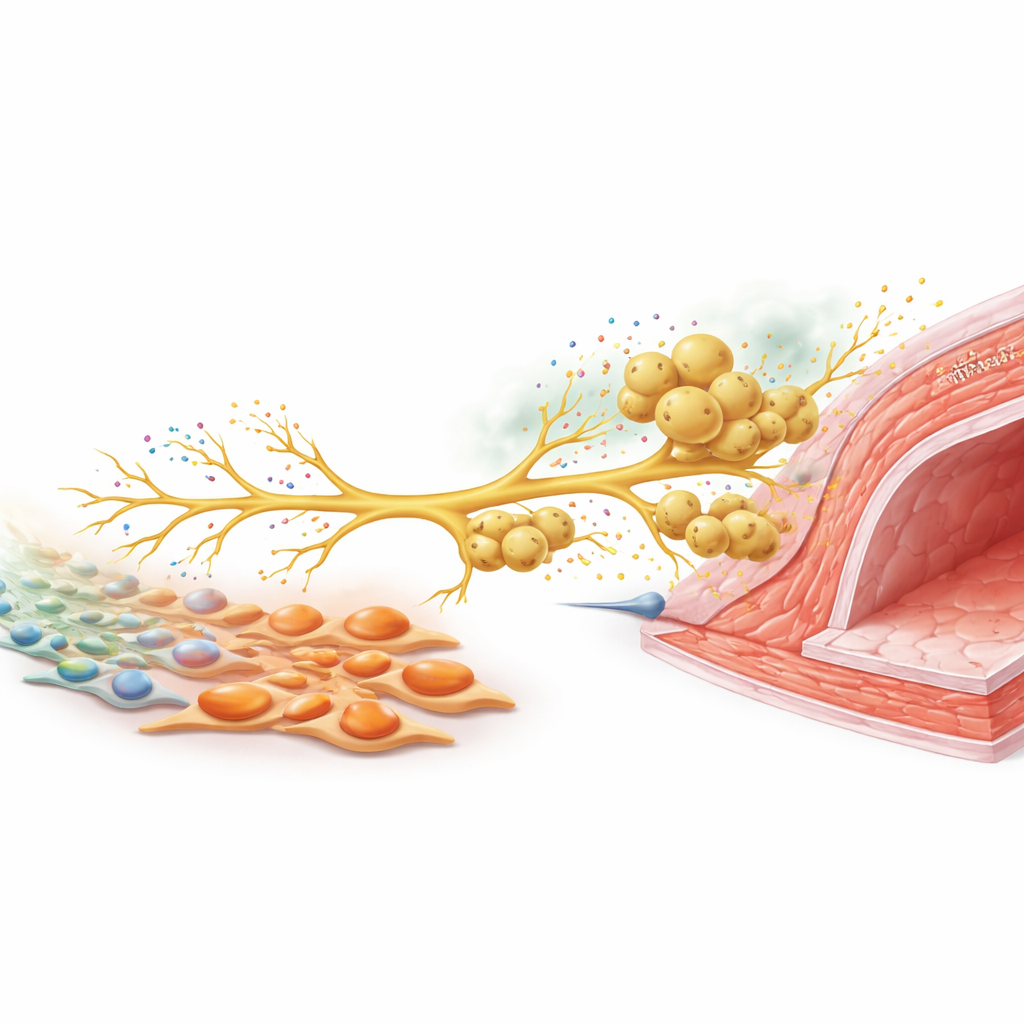
A Nerve Signal That Calls in More Defenders
To understand how these cell types communicate, the team focused on sensory nerves that sense damaging stimuli. In culture, tenascin C–rich myofibroblasts boosted the outgrowth of nerve fibers from dorsal root ganglia neurons. In injured mice, these sensory neurons ramped up production of CCL2, a chemical signal well known for attracting macrophages. When neurons and macrophages were grown together, macrophages migrated along growing nerve fibers, much as they did around arteries in vivo. Silencing CCL2 in neurons sharply reduced macrophage recruitment. In animals, cutting back sensory nerve activity with a toxin, or selectively deleting tenascin C only in fibroblasts, both lowered nerve density, CCL2 levels, macrophage accumulation, and ultimately vessel wall thickening. Combining both interventions did not add extra benefit, suggesting they act along the same pathway.
Why This New Pathway Matters
Taken together, the findings reveal a vicious circle: injury prompts fibroblasts to become tenascin C–producing myofibroblasts; these cells reshape the matrix and stimulate sensory nerves; activated nerves release CCL2, which brings in macrophages; and macrophages secrete inflammatory and fibrotic factors that worsen scarring and narrow the artery. By identifying tenascin C and its downstream nerve–macrophage axis as central drivers of this loop, the work suggests that treatments targeting this molecule or its signaling partners could help keep repaired arteries open for longer, offering patients more durable protection after cardiovascular procedures.
Citation: Tong, X., Shi, G., Fang, Z. et al. Tenascin C+ myofibroblasts exacerbate vascular neointimal hyperplasia by propagation of nerve-macrophage interactions in mice. Nat Commun 17, 2199 (2026). https://doi.org/10.1038/s41467-026-69062-3
Keywords: neointimal hyperplasia, tenascin C, vascular remodeling, sensory nerves, macrophage inflammation