Clear Sky Science · en
Paclitaxel drives TREM2+ macrophage expansion underlying its inferior therapeutic efficacy compared to Nab-paclitaxel
Why this study matters
Many women with breast cancer receive chemotherapy drugs called taxanes, most commonly paclitaxel or a newer version attached to tiny albumin particles, known as nab‑paclitaxel. Doctors have long suspected that the newer drug often works better, but the reasons were unclear. This study digs into both patient data and animal experiments to show that standard paclitaxel can unintentionally help cancer spread to the lungs by reshaping the body’s immune cells, and it points to a way to block this harmful side effect.
Two similar drugs, two different outcomes
The researchers first compared the real‑world performance of paclitaxel and nab‑paclitaxel in thousands of women with breast cancer. By pooling results from 17 clinical studies involving 6,486 patients, they found that nab‑paclitaxel produced higher tumor shrinkage rates and more complete disappearance of cancer in the breast and nearby lymph nodes before surgery. This advantage was especially strong in patients treated after earlier therapies had failed. Yet both drugs were given at similar doses and are designed to attack dividing cancer cells in much the same way, hinting that something beyond direct tumor killing must be different between them.
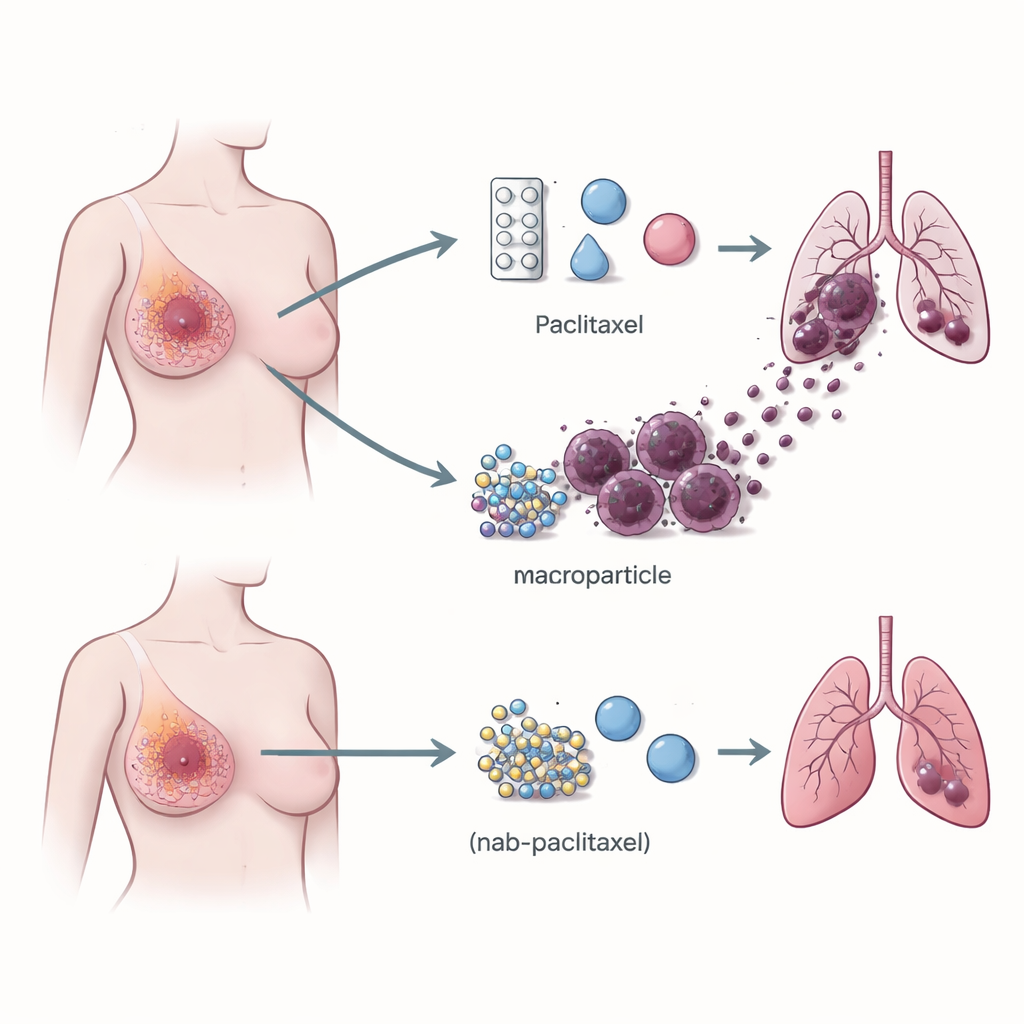
Immune cells that help cancer travel
Attention turned to the tumor’s immune environment—the mix of white blood cells that can either fight cancer or, surprisingly often, help it grow. Using single‑cell RNA sequencing, which reads the activity of genes in thousands of individual cells, the team compared tumors from patients treated with paclitaxel versus nab‑paclitaxel. They found that paclitaxel‑treated tumors contained many more macrophages carrying a receptor called TREM2 on their surface. These TREM2‑positive macrophages were enriched at tumor edges and were far more abundant in patients who later developed lung metastases than in those whose cancer stayed put. The pattern held up in larger patient groups and in mouse models, establishing a strong link between this macrophage type and the risk of cancer spread.
When treatment shrinks tumors but fuels spread
In mice with breast tumors, paclitaxel did what it is supposed to do: it slowed or shrank primary tumors. But at the same time it increased the number of cancer deposits in the lungs and boosted TREM2‑positive macrophages in tumors and in the bloodstream. Nab‑paclitaxel, in contrast, reduced tumor size without driving lung metastasis or expanding these macrophages, even though it was just as effective at shrinking the main tumor. When the gene for TREM2 was deleted in mice, or when TREM2 was reduced with antisense oligonucleotides (short pieces of DNA that silence specific genes), paclitaxel still shrank primary tumors but largely lost its ability to fuel lung spread. This shows that TREM2‑bearing macrophages are not just bystanders; they are required partners in paclitaxel‑driven metastasis.
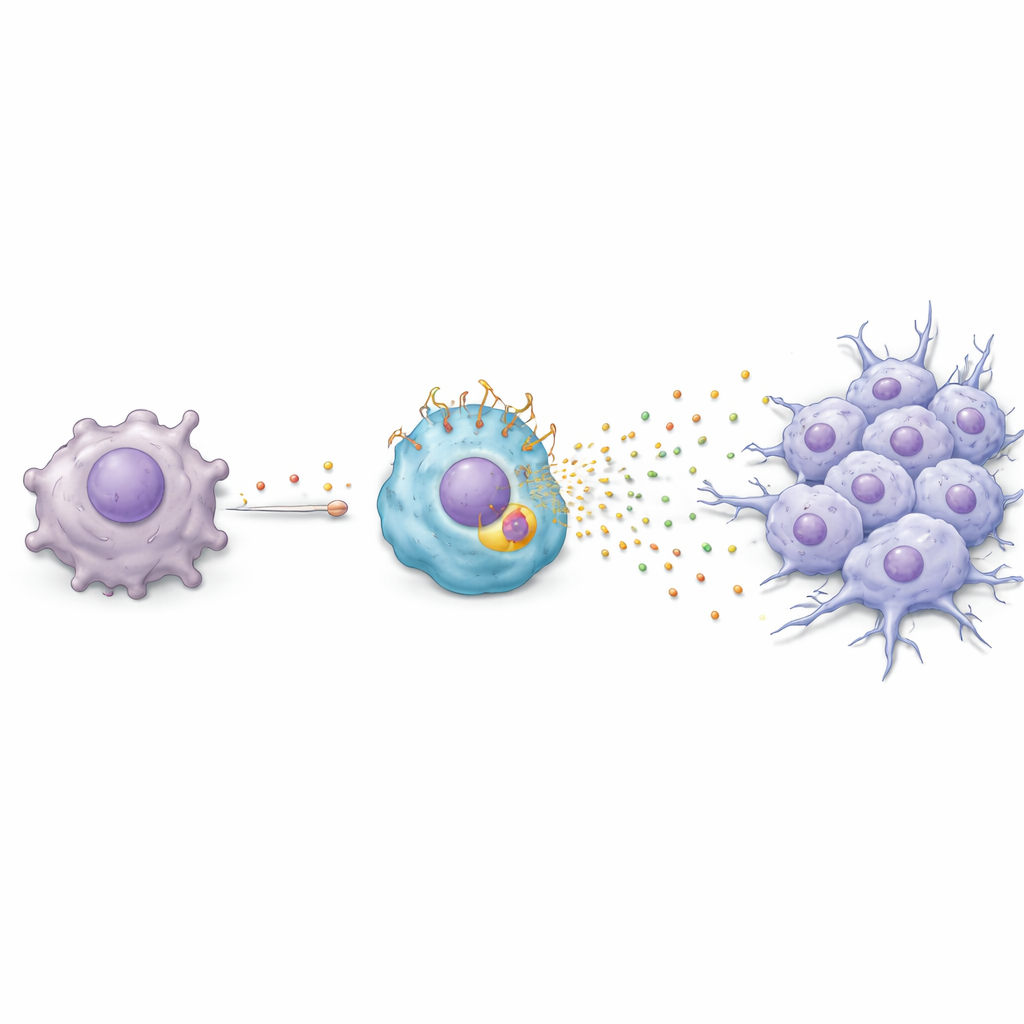
A chemical chain reaction from tumor to immune cell
The team next asked how paclitaxel causes macrophages to turn into this harmful TREM2‑rich state. They discovered a chain reaction: paclitaxel prompts cancer cells to switch on a stress‑response gene called ATF3, which in turn boosts production of a growth factor called FGF2. Cancer cells then release FGF2 into their surroundings, where it acts on nearby macrophages. In those macrophages, FGF2 activates another regulator, EGR1, which raises TREM2 levels. Once TREM2 is high, macrophages begin secreting a cocktail of proteins—including Serpin E1, HGF, CCL3, and CXCL2—that push cancer cells into a more mobile, invasive form known as epithelial‑mesenchymal transition. In lab dishes and in mice, this chemical crosstalk made tumor cells more likely to migrate and invade, key steps on the road to metastasis.
Turning a double‑edged sword into a safer tool
From a lay perspective, the key message is that paclitaxel can act like a double‑edged sword: while it attacks tumors, it also instructs certain immune cells to help the cancer escape to distant organs. Nab‑paclitaxel seems to avoid triggering this particular immune misfire. The encouraging news is that blocking TREM2—or parts of the FGF2–EGR1–TREM2 signaling chain—can separate paclitaxel’s tumor‑shrinking benefits from its metastasis‑promoting risks in animal models. If similar strategies prove safe and effective in people, doctors might be able to keep using a familiar, widely available chemotherapy drug while curbing its tendency to encourage cancer spread, leading to better long‑term outcomes for patients.
Citation: Xing, Y., Zhong, R., Li, Q. et al. Paclitaxel drives TREM2+ macrophage expansion underlying its inferior therapeutic efficacy compared to Nab-paclitaxel. Nat Commun 17, 2272 (2026). https://doi.org/10.1038/s41467-026-69060-5
Keywords: breast cancer, paclitaxel, tumor microenvironment, macrophages, metastasis