Clear Sky Science · en
Regulating interfacial water for oxygen transfer to benzylic C(sp3)–H bonds via Ni-activated tungsten-oxygen covalency
Turning Water and Oil into Useful Chemicals
Chemists have long wanted a cleaner way to turn simple oil-based molecules into more valuable ingredients for plastics, medicines, and everyday products. This study shows how carefully “tuning” the thin layer of water that touches an electrode can let ordinary water supply oxygen to stubborn hydrocarbon molecules, cutting waste and energy use in the process.
Why Oxygen from Water Matters
Many industrial chemicals are made by attaching oxygen to hydrocarbons—molecules built mostly from carbon and hydrogen. Today, that often means using strong oxidizing agents and high temperatures, which consume large amounts of energy and generate climate-warming emissions. Water is a cheap, safe, and abundant source of oxygen, but persuading it to share that oxygen directly with tough carbon–hydrogen bonds usually requires very high voltages. Under those harsh conditions, most of the electricity is wasted splitting water into oxygen gas instead of making useful products.
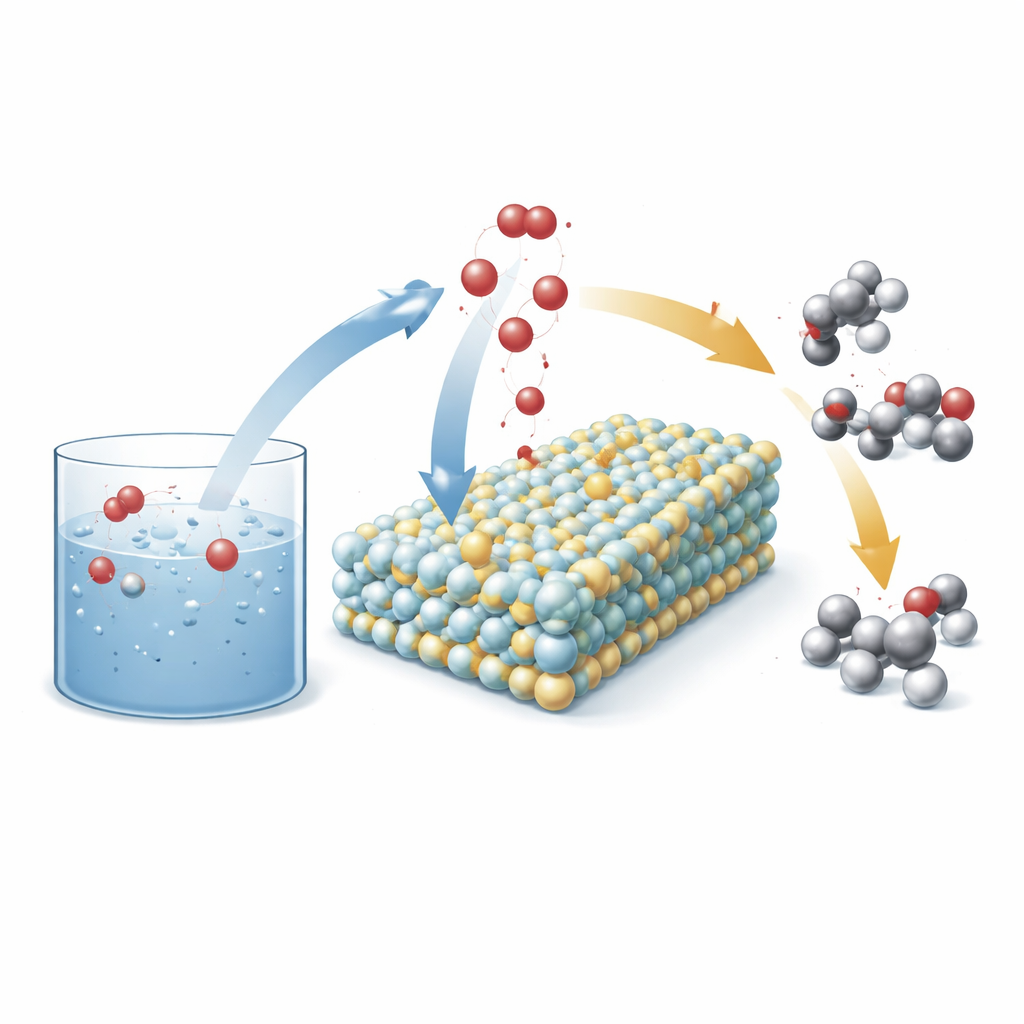
A Smart Electrode that Rearranges Water
The researchers designed a new kind of anode (the positive electrode in an electrochemical cell) built from tiny particles of tungsten carbide that transform during operation into an amorphous tungsten–oxygen material. Into this framework they sprinkle individual nickel atoms. Under reaction conditions, oxygen from water migrates into the solid, turning it into a nickel-activated tungsten oxide surface with many exposed reactive sites. This rebuilt surface then serves as a platform where water and hydrocarbon molecules meet and exchange oxygen in a controlled way, favoring valuable products such as alcohols and ketones instead of over-oxidized acids and other byproducts.
Loosening the Water Cage at the Surface
A key discovery is that nickel atoms subtly disturb the way water molecules pack against the electrode. Normally, water at a charged surface forms a dense web of hydrogen bonds, like a tight net that slows movement. Computer simulations and sensitive infrared measurements show that near nickel-doped regions this network becomes looser, with fewer hydrogen bonds and more “free” water molecules. This opens up channels through which water can more easily diffuse to the reactive tungsten–oxygen sites. As a result, oxygen-bearing fragments from water can reach the surface more quickly and more often, feeding the reaction instead of escaping as oxygen gas.
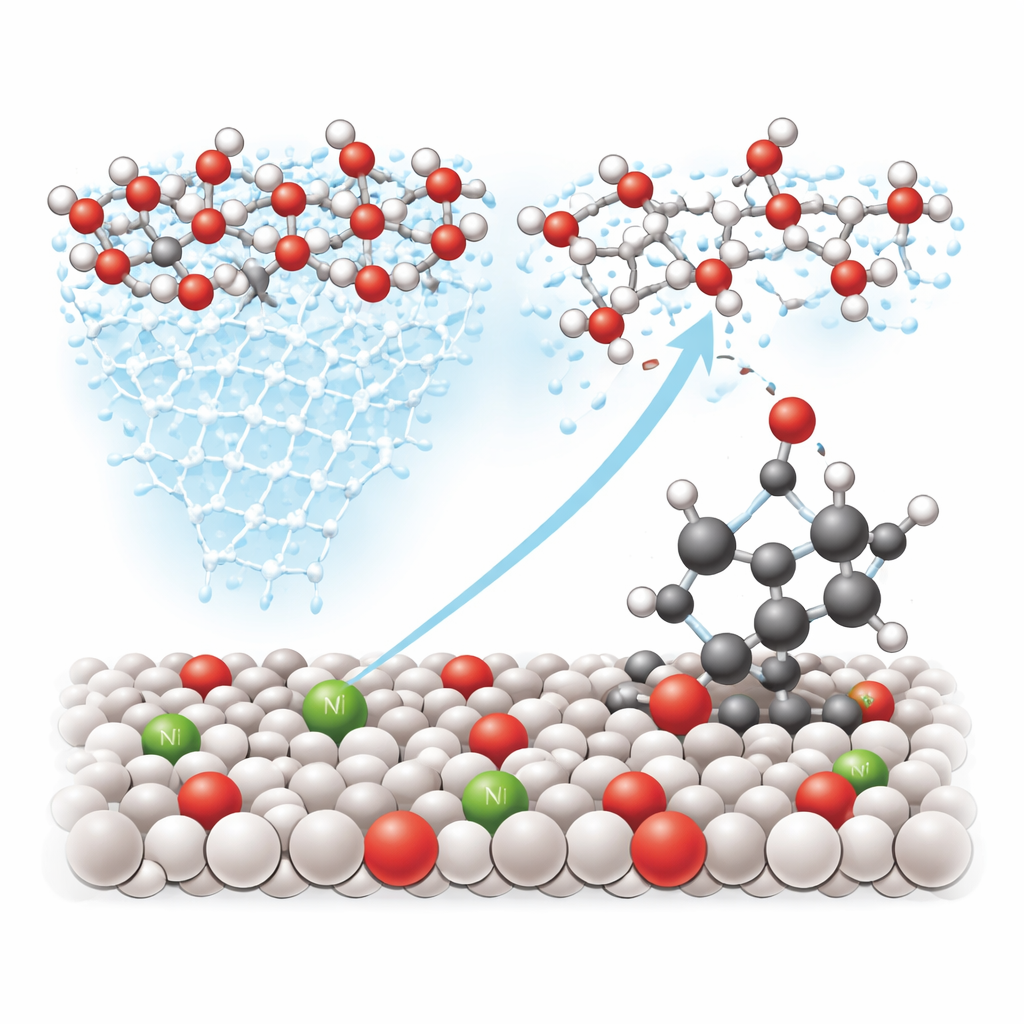
Guiding Oxygen to the Right Bonds
Beyond improving traffic at the surface, nickel also tunes the electronic structure of neighboring tungsten atoms, making certain spots especially good at grabbing and activating water. The team’s experiments with isotopes—special versions of oxygen and hydrogen that can be tracked—show that the active oxygen comes from freshly split water rather than from the bulk of the oxide itself. Spectroscopic “snapshots” capture a short-lived bridge between tungsten, oxygen, and the benzylic carbon on molecules like ethylbenzene. Theoretical calculations confirm that on the nickel-modified surface it is easier for an oxygen atom to attack this carbon–hydrogen site than to follow the usual water-splitting route that produces oxygen gas, effectively steering the reaction toward useful oxygenated products.
Efficient, Selective, and Built to Last
In tests, the nickel-activated electrode converts a range of benzylic carbon–hydrogen bonds into alcohols and ketones with Faradaic efficiencies—how much of the electrical charge goes into the desired chemistry—typically above 50%, reaching over 56% in optimized cases. The system runs for many cycles without losing activity, while maintaining its nickel–tungsten structure. Because it uses water as the only oxygen source and avoids added chemical oxidants, it offers competitive energy efficiency and lower material costs compared with other advanced methods. Although the current scope is focused on benzylic sites, the underlying principle—using atomic additives to reshape interfacial water and redirect reaction pathways—points toward a broader toolkit for greener, electricity-driven oxidation of otherwise inert carbon–hydrogen bonds.
What This Means for Cleaner Chemistry
This work shows that small changes at the atomic level can have outsized effects on how water behaves at an electrode and where its oxygen ends up. By using nickel atoms to both loosen the interfacial water structure and fine-tune reactive sites, the authors turn water into an efficient oxygen donor for hard-to-activate hydrocarbon bonds, while suppressing unwanted oxygen gas formation. In the long run, strategies like this could help redesign oxidation chemistry in the chemical industry, making it more energy-efficient and climate-friendly by using electricity and water instead of harsh oxidants and fossil-derived reagents.
Citation: Leng, BL., Lin, X., Dong, HY. et al. Regulating interfacial water for oxygen transfer to benzylic C(sp3)–H bonds via Ni-activated tungsten-oxygen covalency. Nat Commun 17, 2355 (2026). https://doi.org/10.1038/s41467-026-69054-3
Keywords: electrocatalysis, water oxidation, benzylic C–H activation, nickel-doped tungsten oxide, green chemistry