Clear Sky Science · en
Chlorination-controlled aggregation and film-formation kinetics enabling high-efficiency organic solar cells with low-cost linear conjugated polymers
Why Cheaper Solar Cells Matter
Solar panels are getting better every year, but many of the highest-performing designs still depend on complex, expensive materials. This study tackles a key question: can we build highly efficient organic solar cells from simpler, cheaper plastics without sacrificing performance? The researchers show that by carefully tuning how plastic-like molecules clump together and arrange themselves while a thin film dries, they can make low-cost materials that rival far more sophisticated ones, pushing organic solar cell efficiency just above 20%.
Simple Materials, Big Ambition
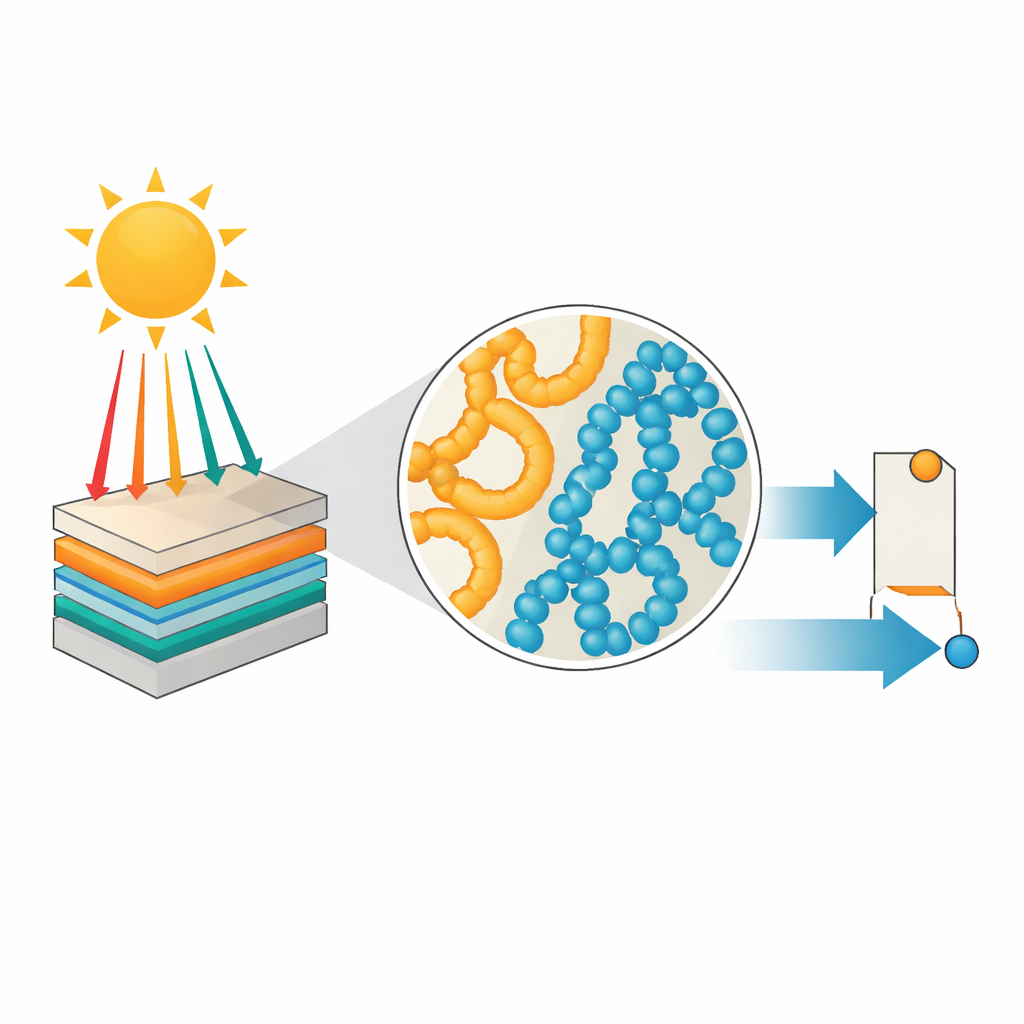
Organic solar cells use carbon-based materials instead of rigid silicon wafers. They are light, flexible, and can be printed from solution, making them attractive for portable devices, building facades, or even wearables. But there has been a mismatch: highly optimized “acceptor” molecules have advanced rapidly, while equally strong “donor” polymers have lagged behind and often require complicated, expensive chemistries. The authors focus on a simpler family called linear conjugated polymers, which are easier to synthesize but usually give lower power output. Their goal is to close this performance gap without giving up the cost and manufacturing advantages of these simpler structures.
A Chlorine Knob for Tuning Behavior
At the heart of the work is a subtle change: adding chlorine atoms along the polymer backbone. The team created three related polymers, identical except for how many chlorine-bearing building blocks they contained—none, half, or all. Although chlorine is a tiny chemical tweak, it acts like a powerful control knob. It strengthens attractive forces between polymer chains, encourages them to lie flatter and stack more neatly, and alters how readily the polymer mixes with the partner acceptor material in the solar cell. These shifts affect not only how the polymers behave in solution, but also how they freeze into a solid film as the solvent evaporates.
From Liquid Mixture to Tiny Pathways
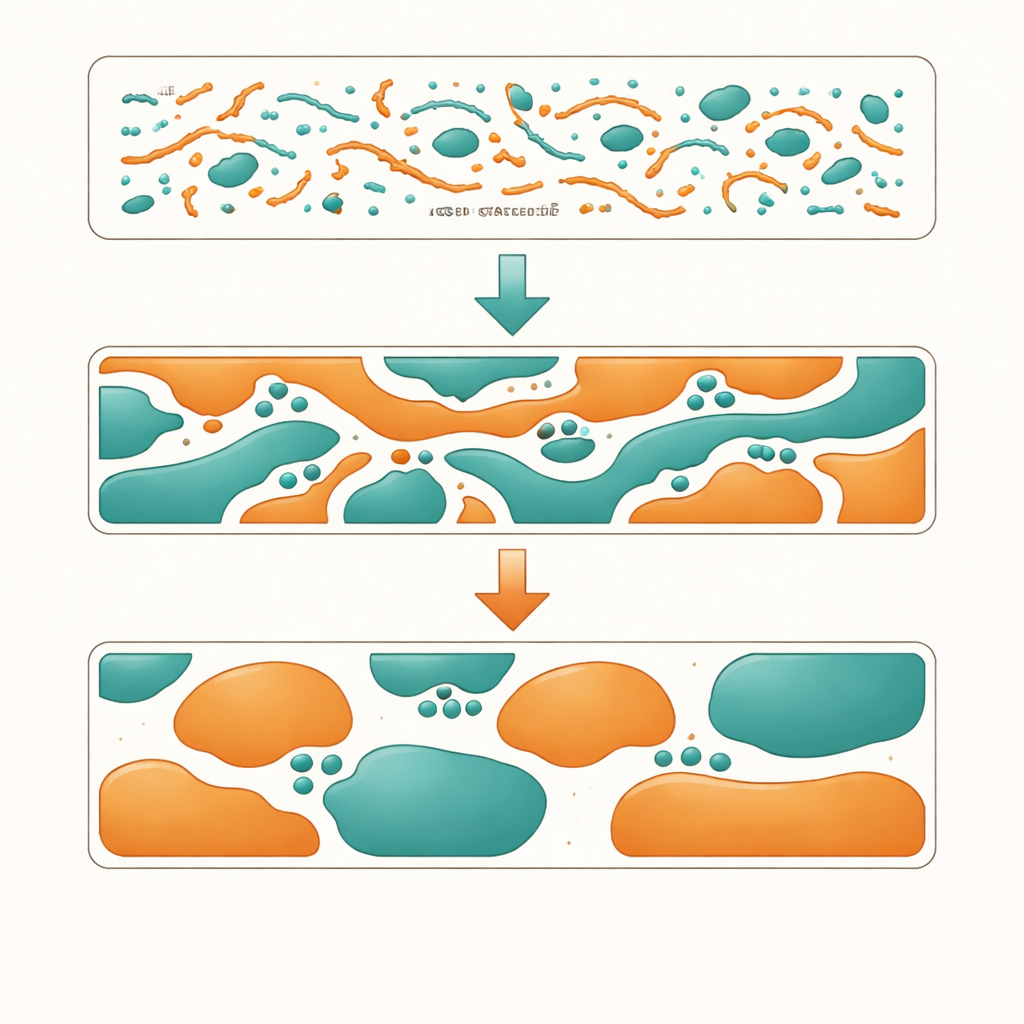
When a solar cell layer dries, countless nanometer-scale events determine its final structure. The polymers may form thin, rope-like bundles in solution that survive into the solid; the acceptor molecules may crystallize earlier or later; the two components may either mix intimately or split into separate regions. Using scattering methods, electron microscopy, and in-situ optical measurements, the researchers show that chlorine content controls the size and shape of polymer bundles and how the two materials separate. With no chlorine, the mixture is too uniform, offering many interfaces for light-generated excitons to split, but not enough clean pathways for charges to travel out. With too much chlorine, the materials repel each other, breaking into large, purer regions that transport charges well inside each region but provide too few interfaces for splitting excitons efficiently.
The “Goldilocks” Polymer
The version with an intermediate level of chlorination lands in the sweet spot. In solution, it forms pre-aggregated polymer bundles of just the right size. As the film dries, these bundles and the acceptor molecules organize into a finely interwoven, bicontinuous network: two intertwined highways of donor and acceptor with many junctions between them. Time-resolved measurements reveal that excitons split rapidly at these junctions, charges move along continuous paths with balanced speeds for positive and negative carriers, and fewer charges fall into traps or recombine. Devices made from this optimized polymer reach a power conversion efficiency of 20.42%, an impressive figure for such a structurally simple material, and they maintain performance well under continuous illumination.
What This Means for Future Solar Tech
For non-specialists, the key message is that smart control over how molecules gather and solidify can make “simple” plastics behave like much more elaborate materials. By tuning chlorination, the authors show that one can steer the self-assembly of an organic solar cell’s active layer to create just the right nanostructure for capturing sunlight and moving charges. This approach keeps synthesis routes short and inexpensive while delivering efficiencies that approach the best in the field, helping bring flexible, printable solar technologies closer to real-world, large-scale use.
Citation: Yin, B., Chen, Z., Wu, B. et al. Chlorination-controlled aggregation and film-formation kinetics enabling high-efficiency organic solar cells with low-cost linear conjugated polymers. Nat Commun 17, 2340 (2026). https://doi.org/10.1038/s41467-026-69051-6
Keywords: organic solar cells, polymer photovoltaics, thin-film solar, material self-assembly, chlorinated polymers