Clear Sky Science · en
Solution-synthesized stable triaza[4]triangulene triradical with a quartet ground state
A New Kind of Tiny Magnet
Magnets are not just the bars that stick to your fridge—they can also be individual molecules whose unpaired electrons act like tiny spinning tops. If chemists can tame such molecular magnets so they stay stable in air and at room temperature, they could become building blocks for future technologies such as ultra‑small memory devices or components in quantum computers. This article reports a rare success story: a triangular carbon‑based molecule that behaves as a robust, high‑spin magnet and can be handled like an ordinary chemical in solution.
Why Triangles Matter for Tiny Spins
Most molecules prefer to pair up their electrons, cancelling out any magnetic behavior. In certain flat, carbon‑rich structures with zigzag edges, however, some electrons remain unpaired and give rise to magnetism. A family of such molecules called triangulenes is especially interesting because theory predicts that as the triangular framework grows, more unpaired electrons appear and they all tend to align in the same direction. That alignment creates a high‑spin state, essentially turning the molecule into a stronger tiny magnet. Unfortunately, as the number of unpaired electrons increases, these molecules usually become extremely reactive and fall apart, especially when chemists try to make them in ordinary liquid solutions.
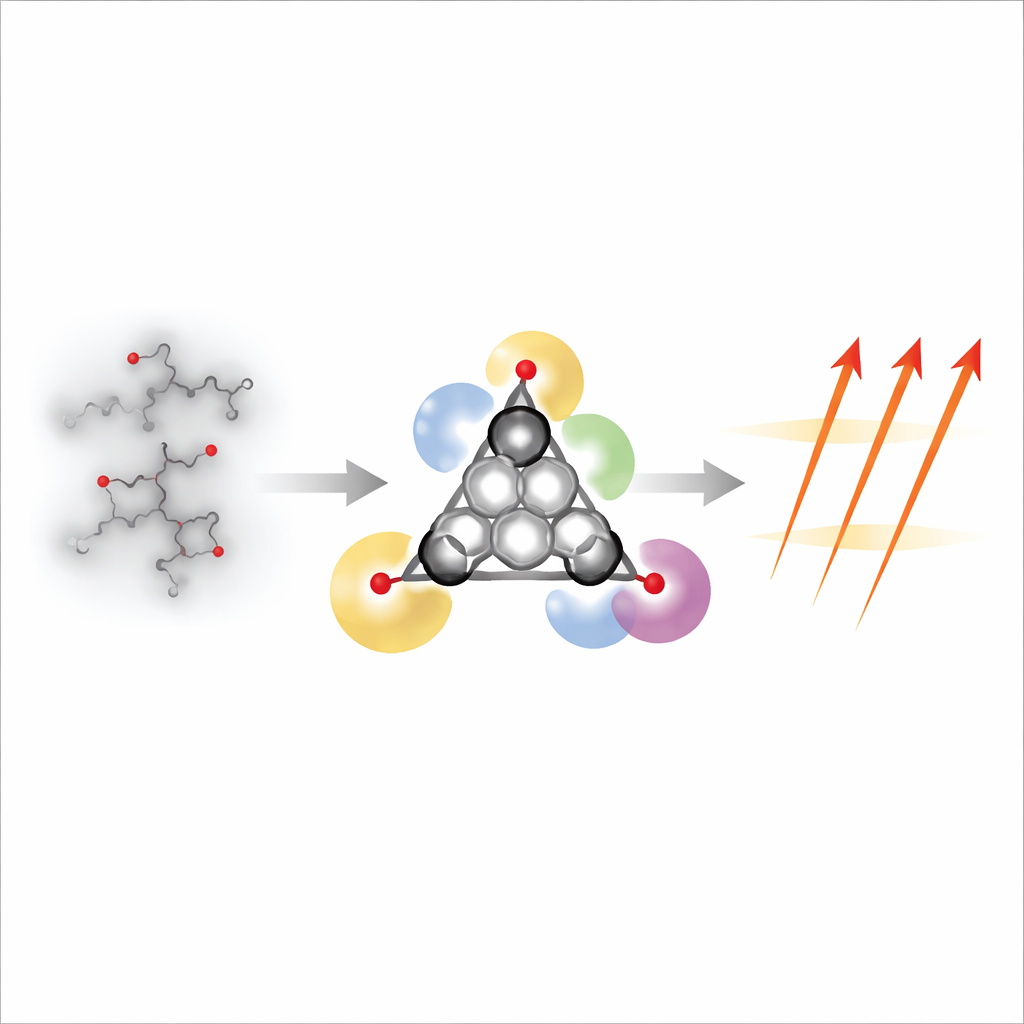
Designing a Tough Triangular Triradical
The authors set out to build a larger, tougher member of this family: a version of a [4]triangulene, which naturally hosts three unpaired electrons. They modified the classic all‑carbon triangle by replacing three edge positions with nitrogen atoms arranged symmetrically, and by surrounding the core with bulky carbon‑rich side groups. The nitrogen atoms help spread out the unpaired electrons over the entire framework, while the bulky groups act like shields that keep neighboring molecules from getting close enough to react. Together, these features make the resulting "triaza[4]triangulene" remarkably resistant to air and light. In solid form, half of the material remains intact even after about nine days in open air; in an oxygen‑rich solution, it lasts for more than a day—exceptional lifetimes for a molecule with three active spins.
Building and Seeing the Molecular Triangle
To construct this complex triangle, the team used a multi‑step organic synthesis. They first stitched together three aromatic building blocks using a cross‑coupling reaction, then folded them into a fused ring system through classic ring‑forming steps, and finally performed an oxidation that converted three carbon‑hydrogen bonds into three carbon‑centered radicals. X‑ray crystallography on single crystals confirmed that the central triangular framework is nearly perfectly flat and symmetric, with bond lengths that signal strong electron sharing across the whole structure. The protective side groups protrude above and below the triangle, forcing neighboring molecules to stay about 7.7 angstroms apart—far enough that their spins barely "feel" each other, so each molecule behaves as an independent magnet.
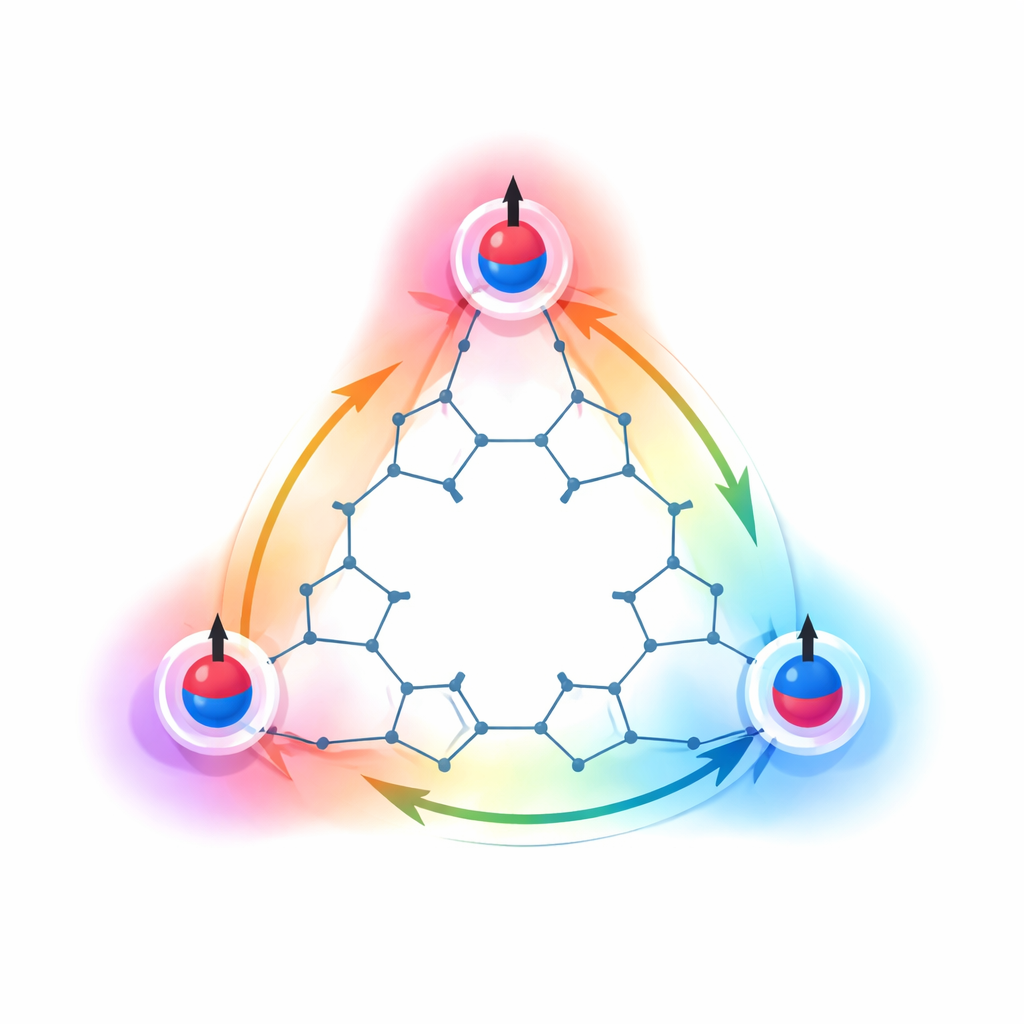
Proving the High‑Spin Magnetic State
To find out how the three spins interact, the researchers turned to sensitive magnetic techniques. Electron paramagnetic resonance (EPR) experiments showed patterns of splitting that can only be explained if the three unpaired electrons are spread out and rapidly exchanging positions across the triangle, rather than being trapped at single atoms. Cooling the material revealed signatures—such as zero‑field splitting and special transitions in the spectrum—that clearly point to a quartet ground state, meaning all three spins align to give a total spin of three‑halves. Measurements using a superconducting quantum interference device (SQUID) magnetometer showed that the energy gap between this high‑spin state and the nearest lower‑spin state is unusually large for an organic molecule. This large gap indicates very strong internal cooperation among the spins, so thermal fluctuations at ordinary temperatures cannot easily flip them into a weaker magnetic state.
Toward Molecular Building Blocks for Quantum Devices
Beyond simply being stable, this triangular triradical behaves in a way that could make it useful for quantum technologies. Pulsed EPR experiments revealed that its spins can maintain their phase coherence for microseconds and relax back to equilibrium over milliseconds at low temperatures—timescales long enough to manipulate and read out spin states using microwave pulses. Because the quartet state offers four distinct energy levels, the molecule can in principle serve as a small multi‑level quantum unit, or "qudit," rather than a simple two‑level qubit. Altogether, the work shows how careful molecular design—combining a triangular carbon framework, strategic nitrogen atoms, and bulky protective groups—can turn a fragile radical system into a robust, well‑defined molecular magnet, paving the way for families of similar molecules to power future spin‑based electronics and quantum information devices.
Citation: Bai, X., Zhang, D., Zhang, Y. et al. Solution-synthesized stable triaza[4]triangulene triradical with a quartet ground state. Nat Commun 17, 2297 (2026). https://doi.org/10.1038/s41467-026-69048-1
Keywords: molecular magnets, organic radicals, triangulene, spintronics, quantum information