Clear Sky Science · en
Mosaic partial epidermal reprogramming remodels neighbors and niches to refine skin homeostasis and repair
Helping Skin Heal Itself
Anyone who has ever dealt with a slow-healing cut or a lasting scar has wished skin could repair itself more quickly and cleanly. This study explores a way to gently “reset” some skin cells so that the whole tissue becomes better at healing. By briefly nudging a small fraction of cells toward a more flexible, youthful state, the authors show that mouse skin can be primed to close wounds faster and form smaller, softer scars—without turning cells into dangerous, tumor-like cells.
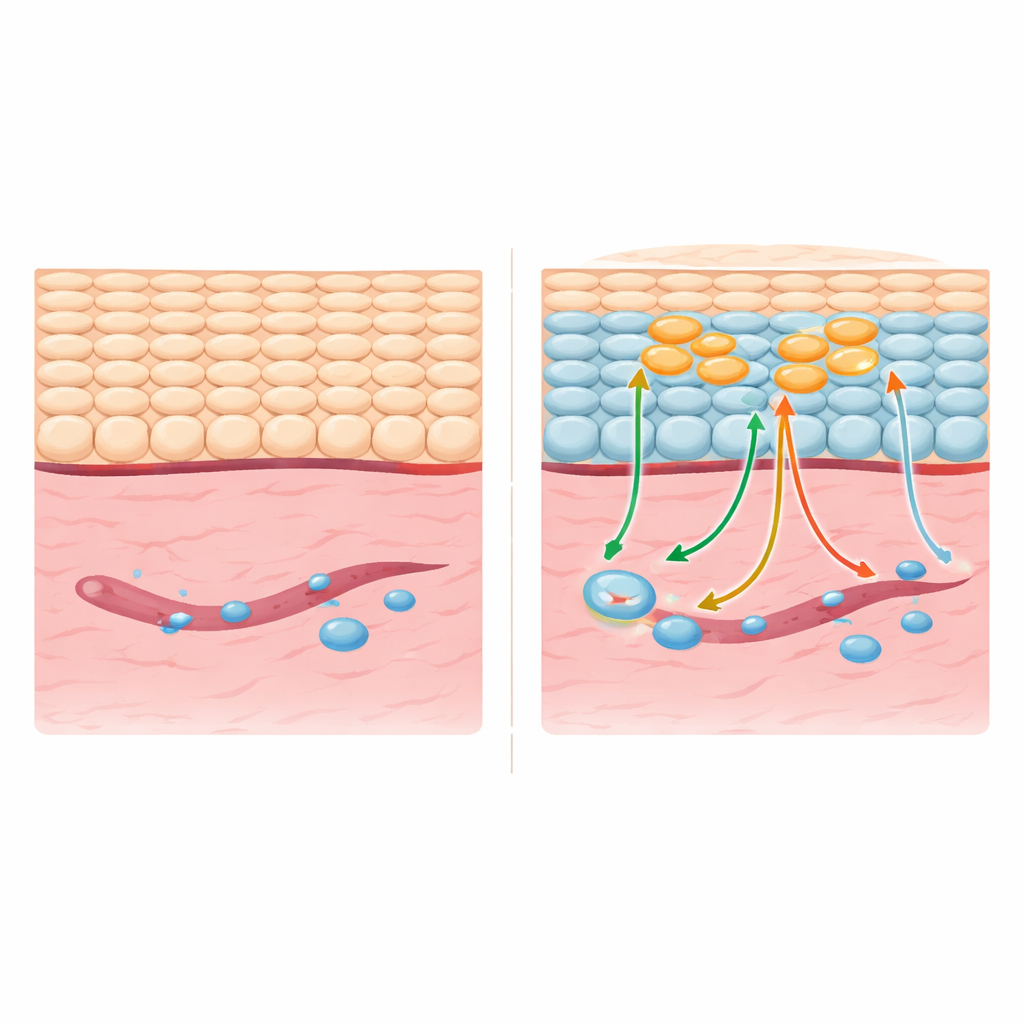
Gently Turning Back the Cellular Clock
The researchers focused on the outer layer of the skin, the epidermis, which is constantly renewed by adult stem cells. Instead of trying to reprogram every cell, they engineered mice so that only scattered epidermal cells would briefly switch on four famous “reprogramming” proteins (often called Yamanaka factors). This short pulse did not erase the cells’ identity or make them fully stem-like; they did not express key pluripotency markers or form tumors. Instead, both the reprogrammed cells and some of their neighbors partially reversed their normal path from stem-like to fully mature cell, regaining features of younger, more flexible epidermal cells while staying within the skin lineage.
Ripple Effects Across Neighboring Cells
Using single-cell RNA sequencing, the team showed that this mosaic, partial reprogramming changed not only the targeted cells but also nearby cells that never expressed the reprogramming factors. Many epidermal cells entered a state that closely resembled cells seen after an actual injury: markers associated with active, migrating keratinocytes were switched on, and genes involved in sensing stress, low oxygen, and inflammatory signals were elevated. Importantly, only the directly reprogrammed cells showed a surge in cell division, whereas neighbors adopted a repair-ready state without excessive proliferation. This balance was traced to distinct signaling pathways: reprogrammed cells activated a growth-promoting PI3K pathway, which in turn boosted EGFR signaling and secreted factors that spread “healing cues” to surrounding cells, while other signals dampened WNT activity in neighbors to prevent runaway growth.
Enlisting the Immune System and Blood Vessels
The skin’s repair program involves more than just epidermal cells, and partial reprogramming reshaped these “niches” as well. The altered epidermis produced chemokines that attracted and activated specific T cells known to aid wound closure. One signaling route, driven by the chemokine CCL20 and its receptor CCR6, drew in RORγt-positive T cells, which normally appear near wound edges and help stimulate re-epithelialization. At the same time, a key stress sensor, HIF-1α, was activated in the epidermis, influencing the pattern of new blood vessel growth in the underlying dermis. This led to blood vessels forming closer to the original wound edges and, over time, helped organize vascular growth in a way associated with better-quality repair rather than thick, fibrotic scarring.
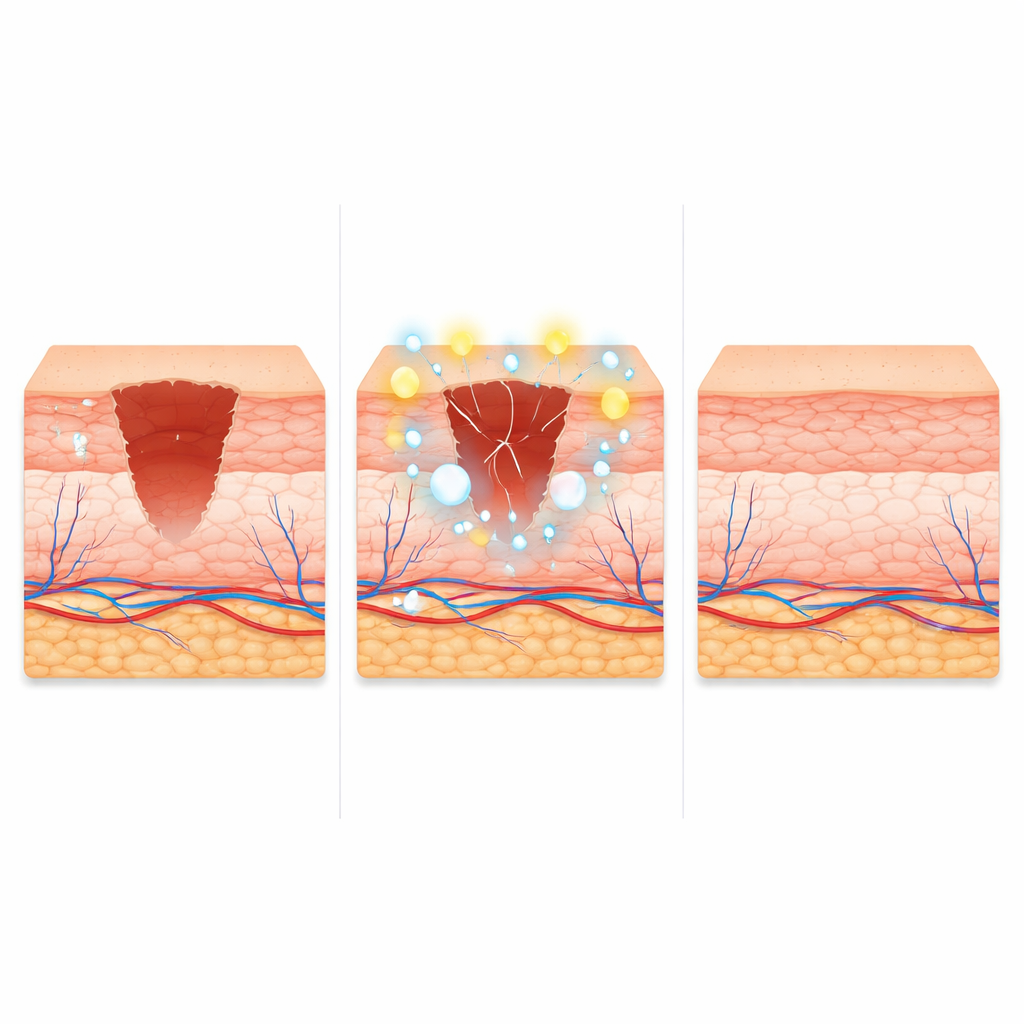
Better Healing and Less Scarring in Healthy and Diabetic Skin
When the researchers made full-thickness wounds in these mice while briefly inducing partial reprogramming, the skin closed more quickly. The migrating edge of the epidermis advanced farther, the barrier recovered sooner, and the resulting scars were thinner, with collagen fibers that more closely resembled unwounded skin and a lower proportion of immature collagen. Temporarily blocking HIF-1α erased much of this benefit, underscoring its central role. Strikingly, the same strategy improved healing in diabetic mice, which normally heal poorly: wounds closed faster, proliferative defects in epidermal cells were restored, and blood vessel growth in the wound bed partially recovered. However, when reprogramming was prolonged beyond the brief window, the skin developed inflammatory changes and thickening, highlighting the need for tight control of dose and timing.
What This Could Mean for Future Therapies
Overall, the work shows that lightly and temporarily “loosening up” a subset of skin cells can rewire their communication with neighbors and support cells, nudging the whole tissue into a coordinated, pro-healing mode. By separating beneficial partial reprogramming from dangerous full reprogramming, and by relying on mosaic rather than global changes, the study points toward future treatments that might one day speed up wound closure and reduce scarring—even in people with conditions like diabetes—by safely boosting the skin’s own regenerative instincts.
Citation: Kwak, M., Choi, E., Jo, Y. et al. Mosaic partial epidermal reprogramming remodels neighbors and niches to refine skin homeostasis and repair. Nat Commun 17, 2191 (2026). https://doi.org/10.1038/s41467-026-69047-2
Keywords: skin regeneration, wound healing, cellular reprogramming, stem cell niches, scar reduction