Clear Sky Science · en
Overcoming the trade-off in reverse osmosis membranes through homologous matching
Cleaning salty water more efficiently
Supplying enough clean drinking water is one of the biggest challenges of this century. Most of Earth’s water is salty, so we increasingly rely on filters that push seawater through thin membranes to remove salt. But these membranes usually face a stubborn trade-off: if you make them let water through faster, they tend to let more salt leak through as well. This study presents a new way to design membranes that break this compromise, opening the door to cleaner water using less energy and more sustainable materials. 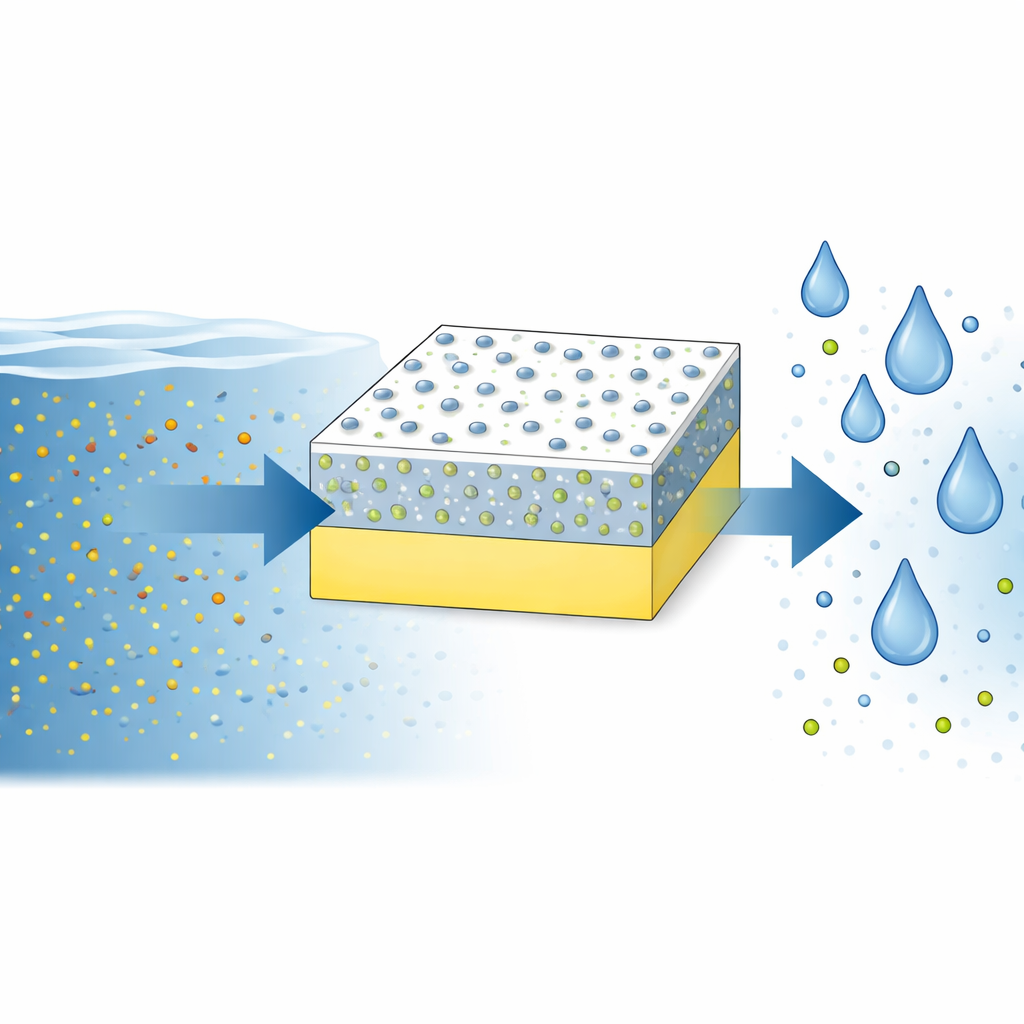
Why current filters hit a wall
Modern desalination plants often use reverse osmosis, where pressure forces seawater through a dense plastic-like film that holds back salt while letting water squeeze through. Cellulose triacetate (CTA), made from plant-based cellulose, is attractive because it is abundant, biodegradable, and relatively environmentally friendly. However, CTA-based membranes still suffer from the classic problem: improving water flow usually reduces their ability to reject salt, and they can also be damaged by chlorine, a common disinfectant. Researchers have tried adding various nanomaterials to fine-tune membrane structure, but these particles often clump together or form defects, which can create leaks for salt or block the flow of water.
Matching building blocks at the nanoscale
The authors tackle this by introducing ultra-small carbon dots—nanoparticles less than ten billionths of a meter across—made from wood and a family of molecules called phenylenediamines. Crucially, one type, called M-CDs, is built from m-phenylenediamine, the same kind of building block used to form the membrane’s ultra-thin polyamide (PA) separation layer. Because these carbon dots and the PA-forming monomer are structurally similar, they “fit” each other at the molecular level. During a process called interfacial polymerization, where an aqueous layer and an oil layer meet to form the PA film, the M-CDs act as nano-intercalators: they slip into the forming layer, guide how the molecules link up, and help build a thinner, smoother, more uniform barrier coated on top of the CTA support.
How the new design boosts water flow and salt blocking
Experiments show that when the concentration of M-CDs is just right, the resulting composite membrane lets more water through while blocking more salt than the original CTA membrane. At the optimal loading, the new membrane increases salt rejection from 96.5% to 99.1% and raises water flux from 15.2 to 18.3 liters per square meter per hour. Microscopy reveals that the M-CDs make the surface more wrinkled and rough on the nanoscale, but also thinner and more hydrophilic, meaning it more readily attracts water. Molecular simulations give a microscopic explanation: the M-CDs slow and shape the way the PA network forms, producing smaller and more uniform pores. Water tends to travel as clusters through well-organized pathways, while ions must partially shed their surrounding water shell to enter the tiny channels—and are effectively turned away. 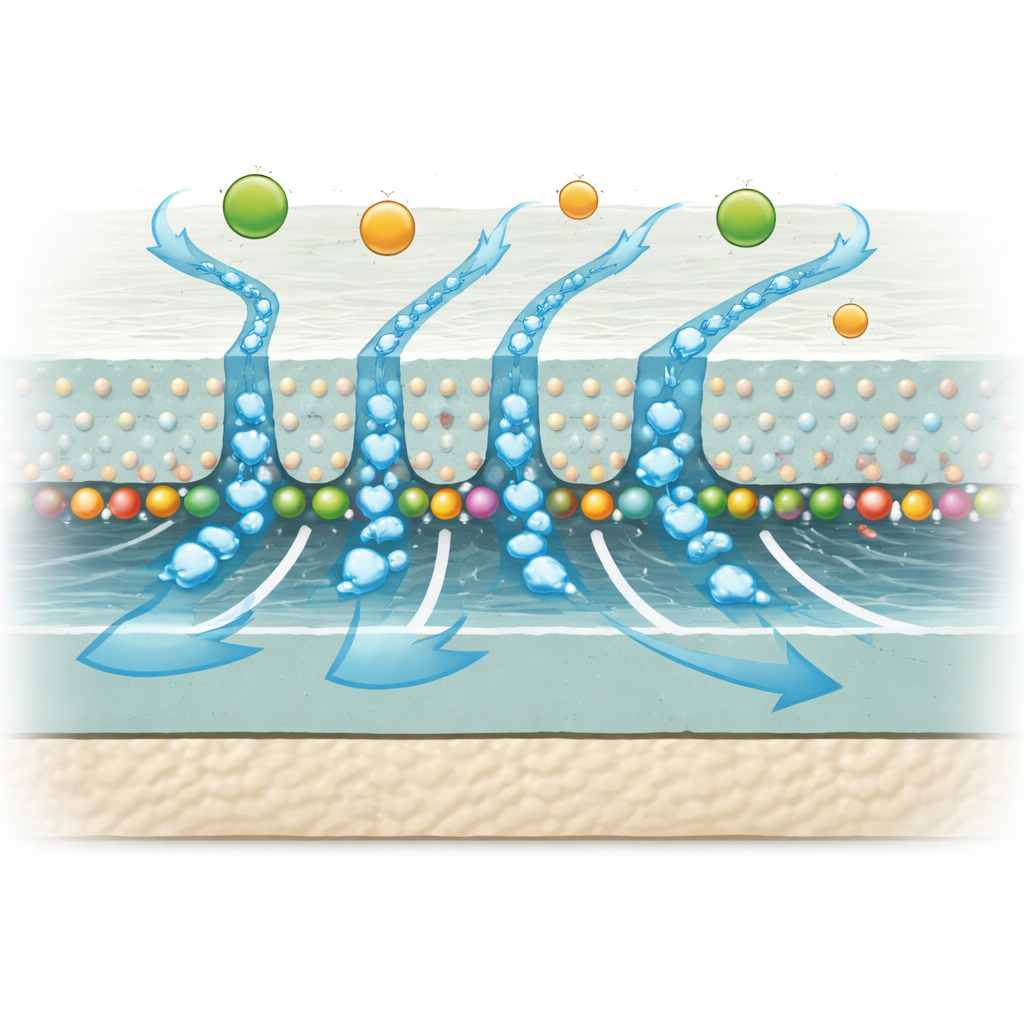
Stability, chlorine resistance, and long-term use
The benefits of the M-CDs go beyond initial performance. The carbon dots carry many oxygen- and nitrogen-containing groups that both attract water and make the membrane surface more negatively charged. This negative charge helps repel negatively charged chloride ions, improving salt rejection and also buffering the surface against chlorine attack. Tests show that after exposure to a strong chlorine solution, the new membrane maintains its high salt rejection far better than a comparable membrane without M-CDs. In long-term trials lasting more than 11 hours, the enhanced membranes keep their flux and salt removal steady, indicating a stable internal structure and strong bonding between the CTA base, the carbon dots, and the PA layer.
What this means for future clean water
To a non-specialist, the key message is that this work finds a smart way to “match” tiny additives with the membrane’s own building blocks so that everything locks together more cleanly at the molecular scale. By carefully tuning this match, the researchers create a plant-derived desalination membrane that sends more fresh water through while keeping more salt out, and that stands up better to harsh disinfectants. This strategy of using structurally compatible, biomass-based carbon dots could be extended to other types of filters, offering a more sustainable and efficient route to turning salty or polluted water into safe drinking water.
Citation: Shao, X., Lv, S., Qin, X. et al. Overcoming the trade-off in reverse osmosis membranes through homologous matching. Nat Commun 17, 2308 (2026). https://doi.org/10.1038/s41467-026-69044-5
Keywords: reverse osmosis, desalination membranes, carbon dots, cellulose triacetate, chlorine-resistant water filtration