Clear Sky Science · en
Surface hole polaron site tuning governs charge carrier separation in BiVO4 photoanodes
Turning Sunlight into Usable Fuel
Sunlight is abundant, but storing its energy for use at night or on cloudy days remains a major challenge. One promising answer is to split water into hydrogen fuel and oxygen using special light-absorbing electrodes dipped in water. This paper explores why a leading material for such electrodes, called BiVO4, wastes much of the sunlight it absorbs—and shows a clever way to redesign its surface so that far more of the captured light is converted into useful chemical energy.
Why Good Materials Still Waste Light
In photoelectrochemical water splitting, an illuminated electrode generates tiny positive and negative charges that must travel to the surface and drive the splitting of water molecules. In metal-oxide materials like BiVO4, many of these charges get stuck in place instead of moving freely. They become trapped in small pockets within the crystal, forming so‑called polarons—localized distortions where a charge pulls nearby atoms slightly out of position. These trapped charges move sluggishly and readily recombine, meaning fewer are available to power water splitting. The problem is especially severe for positively charged holes at the surface, which are exactly the charges needed to oxidize water into oxygen.
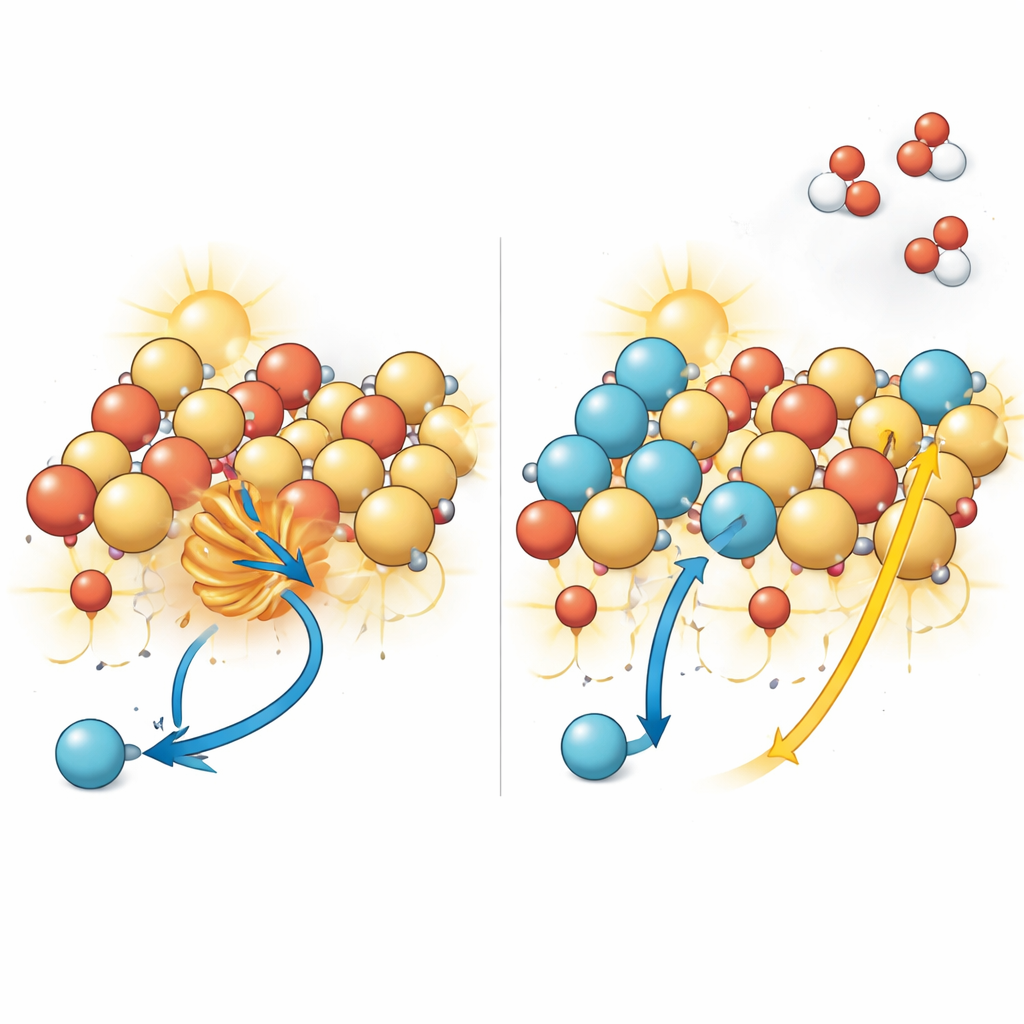
Redesigning the Surface Atoms
The researchers set out to change how the surface of BiVO4 handles these holes without disturbing the rest of the material. Using advanced quantum‑mechanical calculations, they predicted that swapping some of the surface bismuth atoms for indium atoms would make it harder for hole polarons to form. Indium attracts electrons more strongly, which weakens the coupling between charges and the vibrations of the lattice that normally encourage self‑trapping. The team then developed a liquid‑phase cation exchange method, a kind of gentle ion trade at the solid–liquid interface, to selectively replace bismuth with indium only near the surface while leaving the inner structure of BiVO4 intact.
Seeing Atoms and Charges in Action
To confirm that the surface was truly rebuilt as planned, the authors used a suite of high‑resolution techniques. Electron microscopy images showed isolated indium atoms dispersed at the surface rather than clumped into separate particles, while X‑ray–based measurements verified that indium sits in nearly the same type of local environment that bismuth once occupied. Further experiments probed how charges behaved after the modification. Magnetic resonance signals associated with trapped holes nearly vanished, temperature‑dependent light emission revealed weaker coupling between charges and lattice vibrations, and time‑resolved optical measurements showed that the formation of trapped hole states slowed down while the lifetime of mobile charges increased. Together, these observations paint a consistent picture: surface indium sites strongly discourage hole trapping and allow more charges to remain free and active.
From Better Charges to Better Water Splitting
The real test is whether these microscopic improvements translate into better device performance. When used as a photoanode in mildly alkaline water, the indium‑modified BiVO4 generated almost three times more photocurrent than the unmodified version. Adding a simple iron‑oxide co‑catalyst on top pushed the current even higher and greatly improved stability during many hours of operation. Measurements of efficiency showed that a much larger fraction of incoming sunlight was turned into electrical current and that nearly all of those charges went into actually producing hydrogen and oxygen. When paired with a commercial silicon solar cell in a tandem configuration, the system delivered an overall solar‑to‑hydrogen efficiency of about six percent with no external electrical bias, demonstrating a practical route toward stand‑alone solar fuel production.
What This Means for Future Solar Fuels
At its heart, this work shows that tiny changes in which atoms sit at a material’s surface can have outsized effects on how well it handles light‑generated charges. By deliberately suppressing the formation of trapped hole states, the researchers freed more charges to do useful work and significantly boosted water‑splitting performance. Because similar trapped‑charge problems plague many metal‑oxide electrodes, the same strategy of targeted surface substitution could be applied widely, helping to turn more of the sun’s energy into clean, storable hydrogen fuel.
Citation: Liu, H., Cong, H., Yang, G. et al. Surface hole polaron site tuning governs charge carrier separation in BiVO4 photoanodes. Nat Commun 17, 2562 (2026). https://doi.org/10.1038/s41467-026-69039-2
Keywords: solar water splitting, photoanode, hydrogen fuel, charge carrier trapping, surface engineering