Clear Sky Science · en
Stronger when wet: Aquatically robust chitinous objects via zero-waste coordination with metal ions
Why getting stronger in water matters
Most of the plastic objects we rely on every day—from food containers to medical devices—are designed to shrug off water. That same toughness, however, means they linger for decades or centuries as waste. This research explores a very different kind of material: a plastic-like film made from a natural molecule found in shrimp shells that actually becomes stronger when it gets wet, yet still breaks down harmlessly in the environment. It points to a future where we can have durable products without creating permanent pollution.
From seafood waste to useful materials
The study is built around chitin, a structural substance that gives insects and crustaceans their hard outer shells and is second only to cellulose in natural abundance. When chitin is slightly modified, it becomes chitosan, a biopolymer that can already be processed into films and molded objects. The authors looked to nature’s own tricks—especially the way metal ions help harden the cuticles of arthropods—for inspiration. They asked a simple question with big implications: could tiny amounts of metal, combined with water, turn this common biological material into something as strong and reliable as everyday plastics, but without the environmental cost?
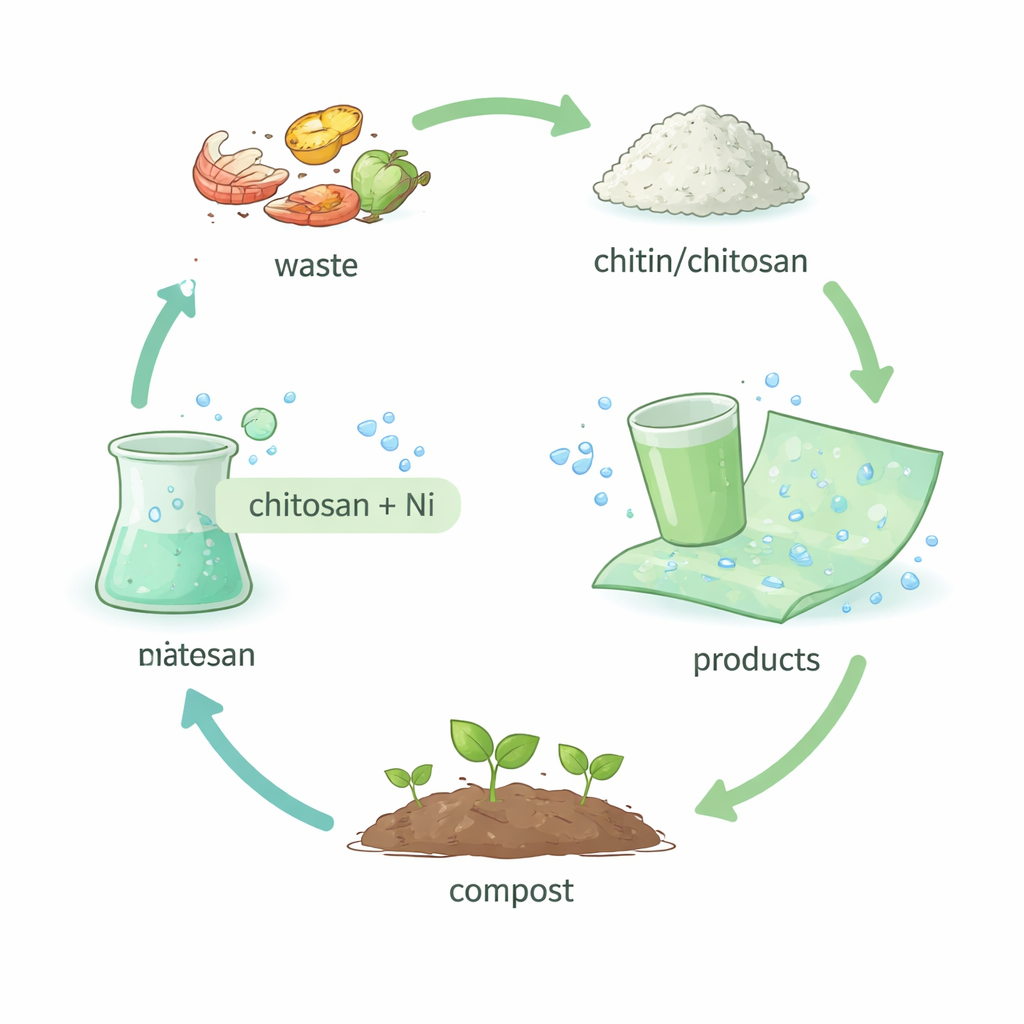
Adding a little metal and a lot of water
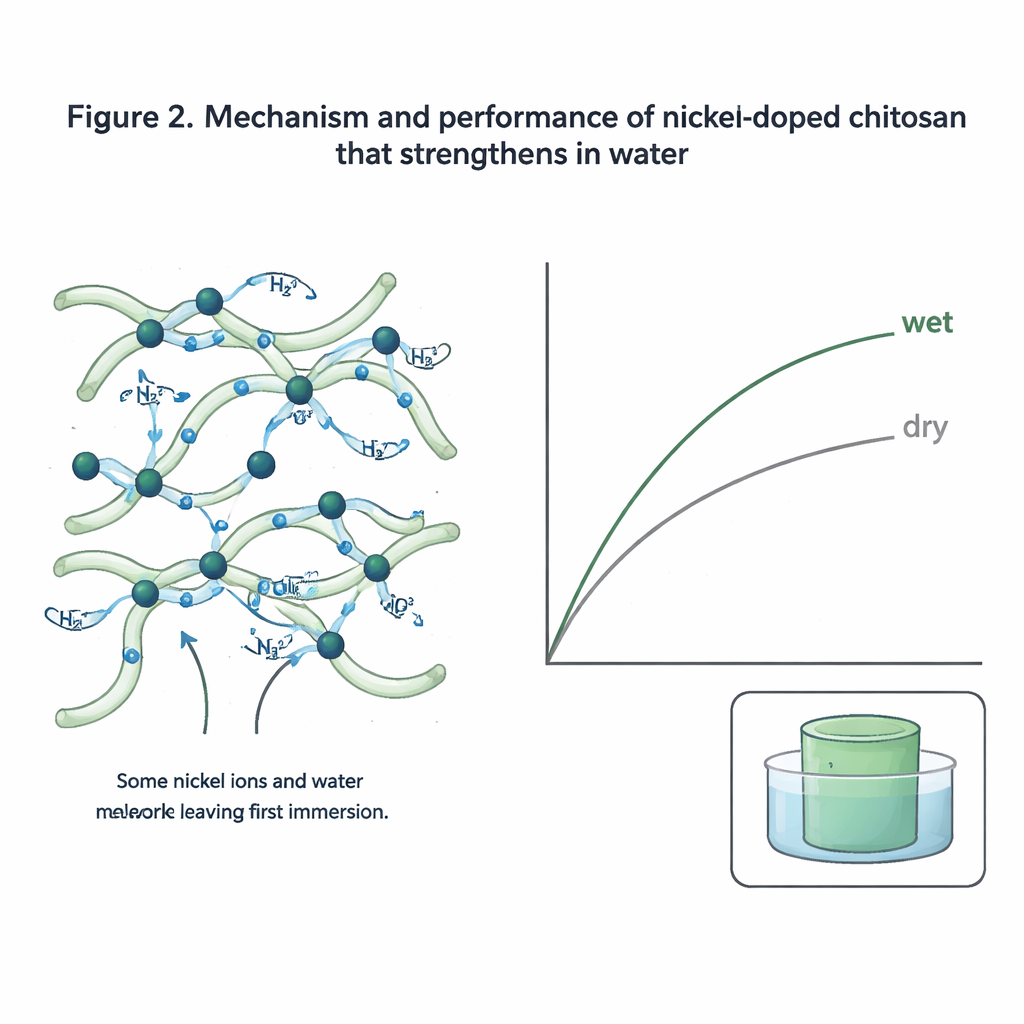
To test this, the researchers dissolved chitosan obtained from discarded shrimp shells in a mild vinegar-and-water solution—no harsh organic solvents required. They then added small amounts of nickel salt and let the water evaporate, forming thin, glassy green films. On a molecular level, the nickel ions nestle between parts of the chitosan chains and attract extra water molecules. Rather than locking everything into a rigid crystal, this combination creates a partly disordered network where chains are linked both directly and through constantly shifting bridges of water and nickel. Spectroscopy and X-ray measurements showed that these films contained more loosely organized regions and substantially more water than pure chitosan, yet still held together as robust solids.
Stronger when soaked, not weaker
Mechanically, the nickel–chitosan films behave in an unusual and valuable way. In air, they reach strengths comparable to common plastics like polypropylene. Above a certain nickel content, they become tougher and more stretchable without losing strength—two qualities that engineers usually have to trade off. The real surprise appears when the films are immersed in water: instead of softening, most versions either maintain their strength or become significantly stronger, with an optimal formulation nearly doubling its tensile strength when wet, reaching the range of engineering plastics. Tests showed that only a small fraction of the nickel is actually needed to achieve this effect; during a first soak, most of the “extra” nickel and its associated water are washed away, leaving behind just enough ions to help organize a dynamic web of water-mediated links that resist breaking under load.
Zero-waste shaping and real-world objects
Because water both builds and “tunes” the material, the authors designed a circular production process. The rinse water that removes surplus nickel from one object is reused as an ingredient for the next, so none of the metal is wasted. Using simple molds, they cast cups and containers that can hold water as reliably as plastic tumblers, yet are fully biodegradable in soil over a few months. A rotating molding machine allowed them to create smoother, closed shapes, and they demonstrated scalability by producing flexible films several square meters in size that remained strong even after a day underwater. Calculations suggest that the nickel content of a single small battery could reinforce more than a dozen drinking cups, keeping metal use extremely low.
A new way to think about durability
To a layperson, the most striking takeaway is that this material flips our usual expectations: instead of fighting water, it uses water as a partner. Tiny amounts of a common micronutrient metal and a naturally abundant biopolymer yield a tough, water-stable, and compostable material that can be formed into everyday objects. Because both nickel and chitosan are already accepted in certain medical uses, the authors foresee applications from medical devices to waterproof coatings, and ultimately to large-scale consumer goods. More broadly, the work hints at a manufacturing future based on regional organic waste, gentle chemistry, and materials that work with their surroundings instead of enduring as permanent debris.
Citation: Kompa, A., G. Fernandez, J. Stronger when wet: Aquatically robust chitinous objects via zero-waste coordination with metal ions. Nat Commun 17, 1397 (2026). https://doi.org/10.1038/s41467-026-69037-4
Keywords: biodegradable plastics, chitosan materials, nickel coordination, water-strengthened polymer, sustainable manufacturing