Clear Sky Science · en
In vivo dual RNA-Seq uncovers key effectors of epithelial barrier disruption by an extracellular pathogen
How Some Germs Sneak Past Our Cellular Fences
Our organs are lined with tightly packed cells that act like walls, keeping blood, microbes and toxins where they belong. Yet certain germs still slip between these cells and spread throughout the body. This study reveals in vivid detail how the bacterium that causes leptospirosis, a serious worldwide disease, silently reaches organs like the liver and kidneys, then uses a clever trick involving calcium inside our cells to prise open these protective barriers.
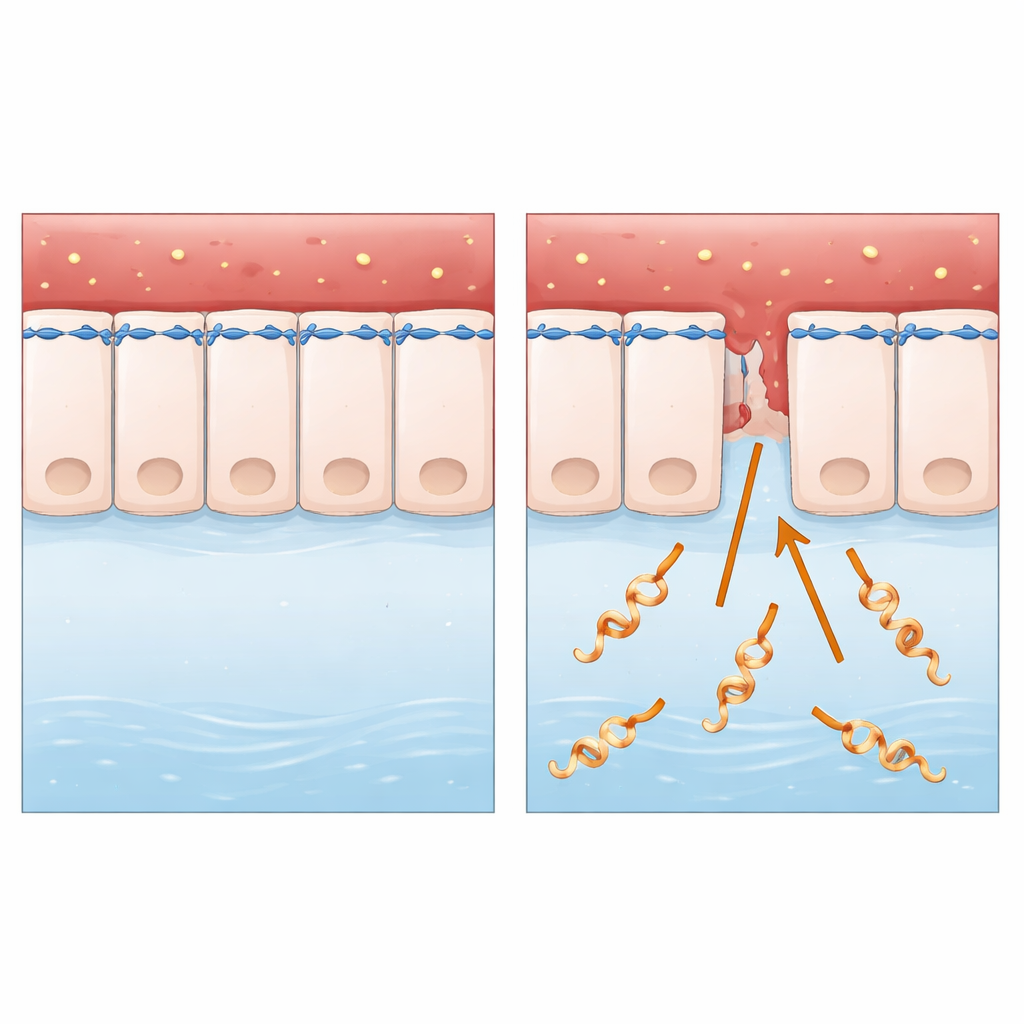
A Stealthy Invader of Animal and Human Tissues
The research focuses on Leptospira interrogans, a spiral-shaped bacterium spread mainly from animals to humans through contaminated water or soil. After entering through small skin breaks or mucous membranes, it travels through the bloodstream and can trigger severe illness, including liver and kidney failure and internal bleeding. Unlike many well-studied microbes, Leptospira lives outside cells and lacks some of the classic “molecular weapons” scientists normally look for. That made it an ideal test case to ask a basic question: how does an apparently simple, free‑living bacterium cross intact cell layers so efficiently?
Watching Host and Germ Genes Talk at the Same Time
To capture the infection process inside a living animal, the team used dual RNA sequencing in hamsters, a model for acute leptospirosis. This method reads out which genes are switched on or off in both host and pathogen simultaneously. The scientists infected hamsters and collected liver and kidney tissue one and three days later. Early on, despite high numbers of bacteria, the animals’ cells barely changed their gene activity, suggesting Leptospira initially slips under the immune system’s radar. By day three, however, both organs showed a strong reaction, especially the liver: genes involved in inflammation, cell–cell junctions and the internal scaffolding of cells were all sharply altered, pointing to active reshaping of tissue barriers.
Breaking the Cellular Zipper from the Outside In
Cell layers are sealed by tight and adherens junctions, which link neighboring cells together and anchor them to a ring of actin and myosin, the same protein pair that helps muscles contract. Microscopy of human epithelial cells infected with Leptospira showed that their shape changed, gaps opened between cells and key junction proteins became weaker or were misplaced. Importantly, the cells did not die, meaning the barrier was being dismantled, not destroyed. The researchers also saw signs of stress in the cells’ protein‑folding factories, but blocking this stress did not restore the junctions, pointing them toward another culprit.
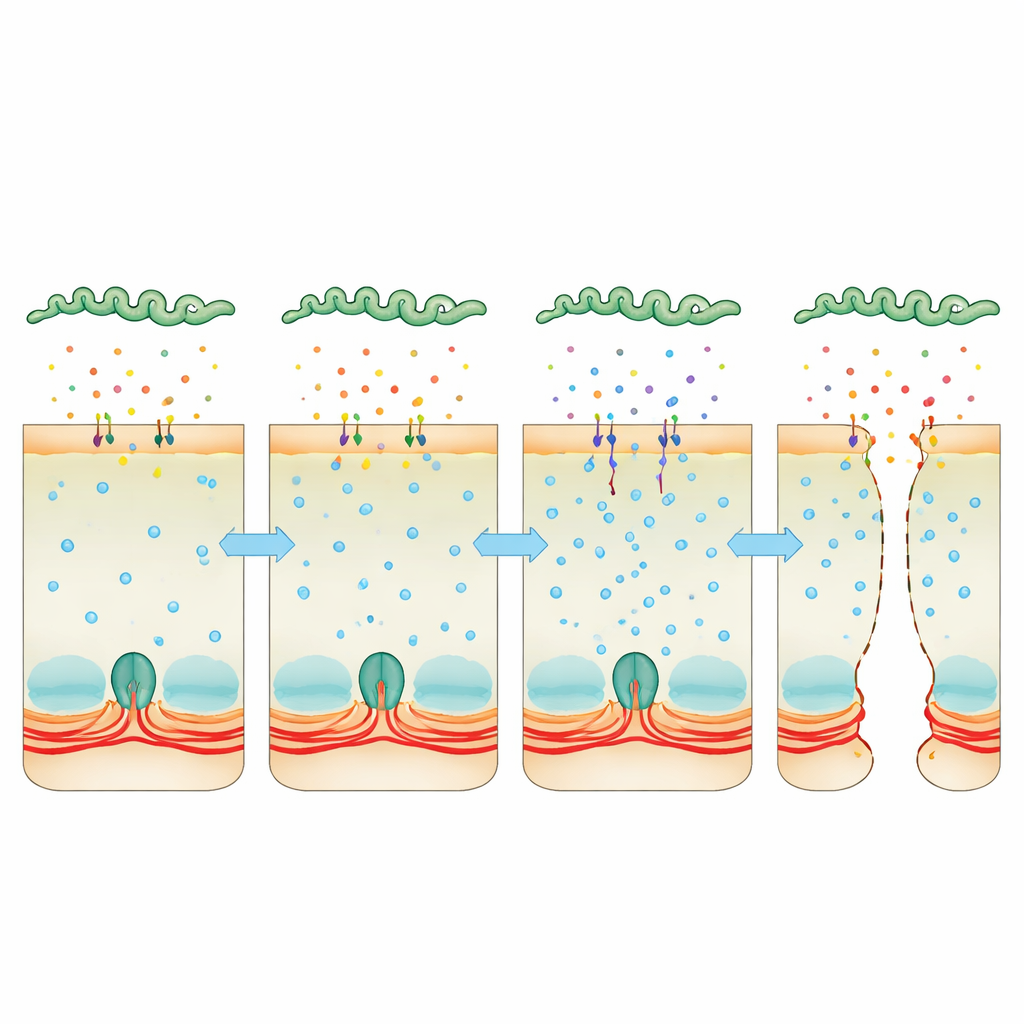
Hijacking Calcium as a Hidden Crowbar
Attention turned to calcium, a universal signal inside cells that can tighten or relax the actin–myosin ring. Infected cells accumulated more calcium, and this was linked to higher activity of myosin light chain kinase, an enzyme that makes the contractile ring tug on junctions. When the team used a compound that mops up calcium, or drugs that inhibit calmodulin or this kinase, the cell layers stayed more intact and bacteria had a harder time squeezing through. At the same time, deep sequencing of bacterial RNA revealed that, inside the host, Leptospira strongly boosts production of two “Virulence‑Modifying” proteins that resemble toxins. These proteins are secreted into the surroundings, stick to host cells and are found inside them. When bacteria lacked one or both of these proteins, they were far less able to cross cell layers or cause lethal disease, and their secreted broth no longer raised calcium levels or dismantled junctions as effectively.
What This Means for Disease and Future Defenses
Taken together, the results outline a clear story. After slipping into the body almost unnoticed, Leptospira begins to secrete toxin‑like Virulence‑Modifying proteins. These proteins bind to epithelial cells and disturb calcium balance inside, which in turn switches on a contractile pathway that pulls apart the cellular “zipper” holding neighboring cells together. As the tight and adherens junctions loosen, microscopic gaps appear, allowing the bacteria to pass between cells, colonize organs and trigger severe illness. By pinpointing this calcium‑driven mechanism and the bacterial proteins behind it, the study opens a path toward new strategies—such as blocking these proteins or their signaling route—to reinforce our cellular fences and limit the spread of leptospirosis and possibly other infections that exploit similar tactics.
Citation: Giraud-Gatineau, A., Haustant, G., Monot, M. et al. In vivo dual RNA-Seq uncovers key effectors of epithelial barrier disruption by an extracellular pathogen. Nat Commun 17, 2274 (2026). https://doi.org/10.1038/s41467-026-69033-8
Keywords: leptospirosis, epithelial barrier, calcium signaling, bacterial toxins, host–pathogen interactions