Clear Sky Science · en
Engineered internal architecture of core-shell lipid nanoparticles promotes efficient mRNA endosomal release
Why this new delivery trick matters
Many of the most exciting new medicines, from COVID-19 vaccines to experimental cancer treatments, are built from messenger RNA, or mRNA. But there is a stubborn problem: once these fragile strands enter our cells, most of them get trapped and destroyed in small internal sacs called endosomes. This study introduces a clever redesign of the tiny fat bubbles, known as lipid nanoparticles, that carry mRNA. By reorganizing their inner structure around a solid gold core, the researchers dramatically improve how much mRNA escapes into the cell’s working space, where it can actually make useful proteins.
Giving tiny couriers a better body plan
Current mRNA vaccines and therapies mostly rely on soft, blob-like particles made of a mix of fats that wrap around the mRNA. In theory, special “ionizable” fats in these particles should become positively charged in the acidic interior of endosomes and tear open the endosomal membrane, freeing the mRNA. In practice, the mRNA’s negative charge cancels out much of this effect, so only about 2 percent of the delivered mRNA ever reaches the cell’s protein-making machinery. The authors asked a simple but powerful question: instead of only changing the chemistry of the fats, what if they re‑engineered the internal architecture of the particle to control where the mRNA and lipids sit relative to each other?
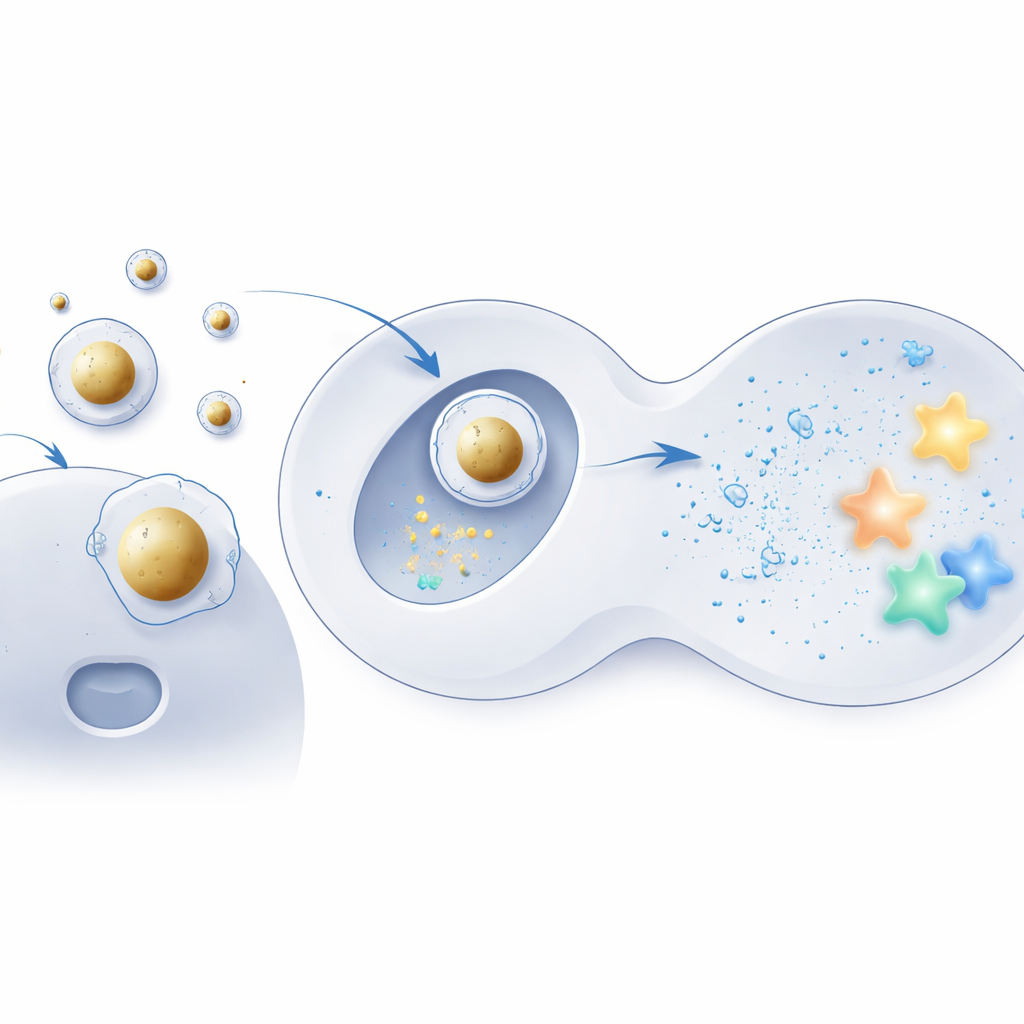
Building a gold-centered delivery shell
To test this idea, the team used tiny gold spheres as rigid cores and coated them with the same ionizable lipids used in conventional formulations. The negatively charged mRNA was first condensed onto these cores, and only then was the assembly wrapped in a familiar mixture of helper fats and stabilizing molecules, creating an orderly core–shell structure. Electron microscopy and computer simulations showed that these “Au‑LNPs” were highly uniform, thermodynamically stable, and kept their shape well in watery, body-like conditions. Importantly, their overall fat makeup remained the same as that of standard particles, isolating internal structure—not ingredients—as the key change.
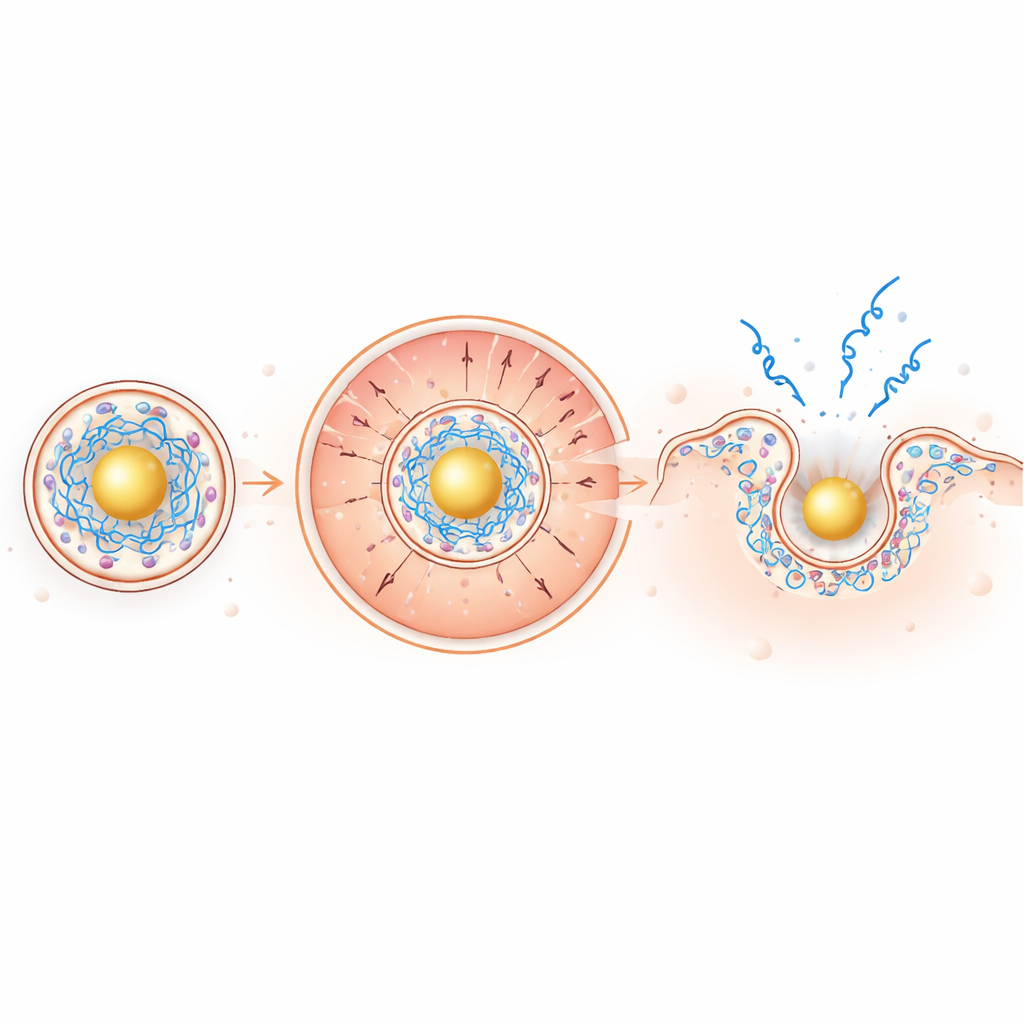
How a rearranged interior boosts escape
Inside the mildly acidic endosomal environment, the gold-based core becomes strongly positive, pulling the negatively charged mRNA inward while pushing the positively charged ionizable lipids outward toward the surrounding membrane. This charge-driven segregation concentrates membrane-disrupting lipids exactly where they are needed. In model membrane tests and in red blood cells, the new particles were more effective at puncturing membranes at low pH while remaining quiet at neutral pH, a desirable safety feature. In living cells, imaging of fluorescent mRNA revealed about a twofold increase in endosomal escape and an astonishing ~100-fold increase in how far mRNA spread through the cytoplasm compared with conventional particles.
From dishes to mice: stronger signals and stronger protection
These physical advantages translated into real biological gains. Across several cell types, the gold-core particles drove much higher production of reporter proteins than standard formulations, especially at low doses. In mice, they boosted mRNA-directed protein output by up to five- to sevenfold after injections into muscle or veins, without changing where in the body the particles tended to accumulate. When used to deliver a SARS‑CoV‑2 spike mRNA vaccine, the redesigned particles roughly doubled antibody levels after the first shot and further increased them after a booster, while also producing more potent virus-neutralizing antibodies. In a challenging triple‑negative breast cancer model, mRNA cancer vaccines carried by the new particles shrank tumors more effectively, lowered tumor activity signals, and notably prolonged survival.
Safety and a path toward future medicines
Despite using gold at their core, the particles appeared well tolerated in mice. Body weights, inflammatory markers, and tissue examinations showed no major toxicity, and gold levels in organs dropped back toward baseline within two days. The authors stress that gold serves mainly as a proof-of-concept scaffold: the same internal design principle could be applied to other safe or biodegradable materials. The central message for non-specialists is that how we pack mRNA inside its tiny carrier can matter as much as what we pack it with. By giving lipid nanoparticles a more disciplined internal layout, this work offers a new way to overcome a longstanding bottleneck in mRNA delivery—unlocking more effective vaccines and therapies across infectious disease, cancer, and beyond.
Citation: Li, T., Zhang, J., Guo, J. et al. Engineered internal architecture of core-shell lipid nanoparticles promotes efficient mRNA endosomal release. Nat Commun 17, 2187 (2026). https://doi.org/10.1038/s41467-026-69017-8
Keywords: mRNA delivery, lipid nanoparticles, endosomal escape, nanomedicine, RNA vaccines