Clear Sky Science · en
Combined targeted and epigenetic-based therapy enhances antitumor immunity by stabilizing GATA6-dependent MHCI expression in pancreatic ductal adenocarcinoma
Why this research matters for pancreatic cancer
Pancreatic ductal adenocarcinoma is one of the deadliest cancers, in large part because it hides from the immune system and resists most drugs. This study uncovers a particular tumor cell state, driven by a gene called GATA6, that makes pancreatic cancer cells more visible to immune attack. The authors also show how combining two types of drugs can preserve this vulnerable state and boost the ability of killer T cells to attack tumors in experimental models.
A tumor cell state that invites the immune system
Not all pancreatic cancer cells behave the same. Some keep a more orderly, epithelial-like identity, while others shift toward a more mobile, aggressive, mesenchymal-like state. The transcription factor GATA6 helps maintain the epithelial identity and is associated with better outcomes in patients. In large patient cohorts, tumors with high GATA6 levels contained more immune cells, including CD8 “killer” T cells, than tumors with little or no GATA6. Using advanced spatial imaging on human tumor samples, the researchers found that GATA6-positive cancer cells more often sat next to CD8 T cells and displayed higher levels of a molecule called MHCI, which acts like a flag showing T cells what is inside the cell. These observations suggested that GATA6 marks a tumor cell population that is naturally more visible to the immune system. 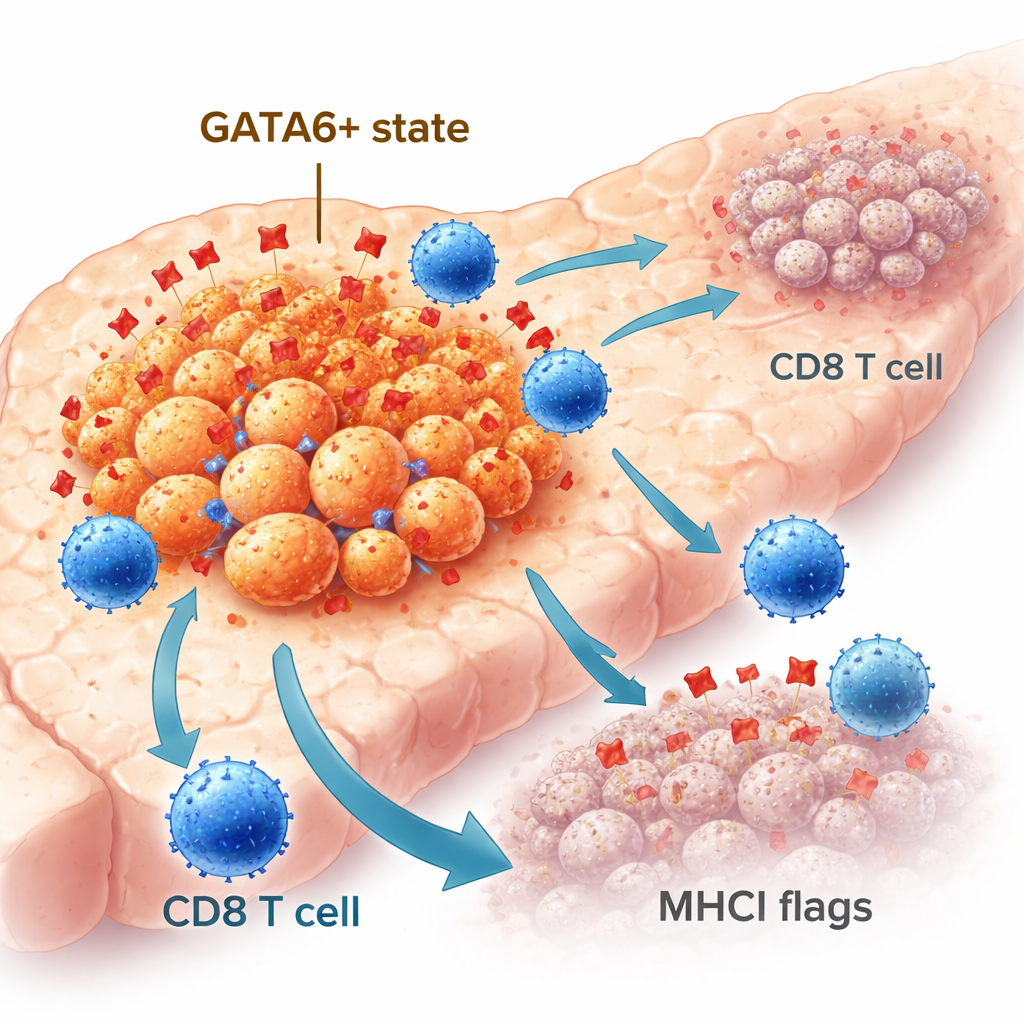
Targeted therapy that boosts tumor visibility—but at a cost
The team next asked whether targeted drugs could further enhance this visibility. They focused on MEK inhibitors, a class of drugs that block a growth signaling pathway frequently active in pancreatic cancer. In mouse pancreatic cancer cell lines and in tumors grown in mice and in patient-derived xenografts, MEK inhibition increased MHCI on the surface of GATA6-high tumor cells, making them potentially better targets for T cells. When tumors with high GATA6 were treated with MEK inhibitors in mice, tumor growth slowed, MHCI went up, and more CD8 T cells carrying cytotoxic molecules entered the tumors. Blocking GATA6 directly, either by gene knockout or by rapidly degrading the protein, abolished this MEK inhibitor–induced rise in MHCI and the resulting tumor control, demonstrating that GATA6 is essential for this beneficial effect.
How tumors adapt: slipping into a harder-to-kill state
However, the story came with a twist. Long-term treatment with MEK inhibitors drove many tumor cells to shift from the GATA6-positive epithelial state into a more mesenchymal, GATA6-low state associated with treatment resistance. In both mouse models and patient-derived xenografts, MEK inhibitor–treated tumors eventually showed fewer GATA6-positive cells, more markers of epithelial-to-mesenchymal transition, and overall less durable benefit. Although individual GATA6-positive cells still upregulated MHCI in response to the drug, the shrinking size of this population limited the overall improvement in tumor antigen presentation, helping explain why MEK inhibitors alone have disappointed in clinical trials of pancreatic cancer.
Epigenetic drugs that hold tumors in the vulnerable state
To counter this adaptive escape, the researchers turned to histone deacetylase inhibitors, or HDAC inhibitors, a class of epigenetic drugs known to influence cell identity and immune visibility. In cell cultures and mouse models, combining MEK inhibitors with class I HDAC inhibitors such as domatinostat restored GATA6 expression that had been suppressed by MEK inhibition, reversed signs of epithelial-to-mesenchymal transition, and further boosted MHCI levels on tumor cells. Importantly, this combination therapy led to stronger infiltration and activation of CD8 T cells and more tumor cell death in orthotopic mouse tumors and in a genetically engineered mouse model that closely mimics human pancreatic cancer. Depleting CD8 T cells eliminated much of the benefit, confirming that the combination works largely by empowering killer T cells.
Toward smarter combination therapies for a tough cancer
Putting these findings together, the study proposes a strategy in which targeted MEK inhibition and epigenetic HDAC inhibition are used together to stabilize a GATA6-driven tumor cell state that is rich in MHCI and therefore highly recognizable to the immune system. By both increasing the number of GATA6-positive cells and enhancing their MHCI “flags,” the combination promotes deeper CD8 T cell infiltration, stronger cytotoxic activity, and longer survival in aggressive mouse models of pancreatic cancer. While HDAC inhibitors can carry significant side effects and the work is preclinical, this research points toward future treatment approaches that might pair pathway-targeted drugs and carefully chosen epigenetic modifiers to make pancreatic tumors more vulnerable to the body’s own immune defenses. 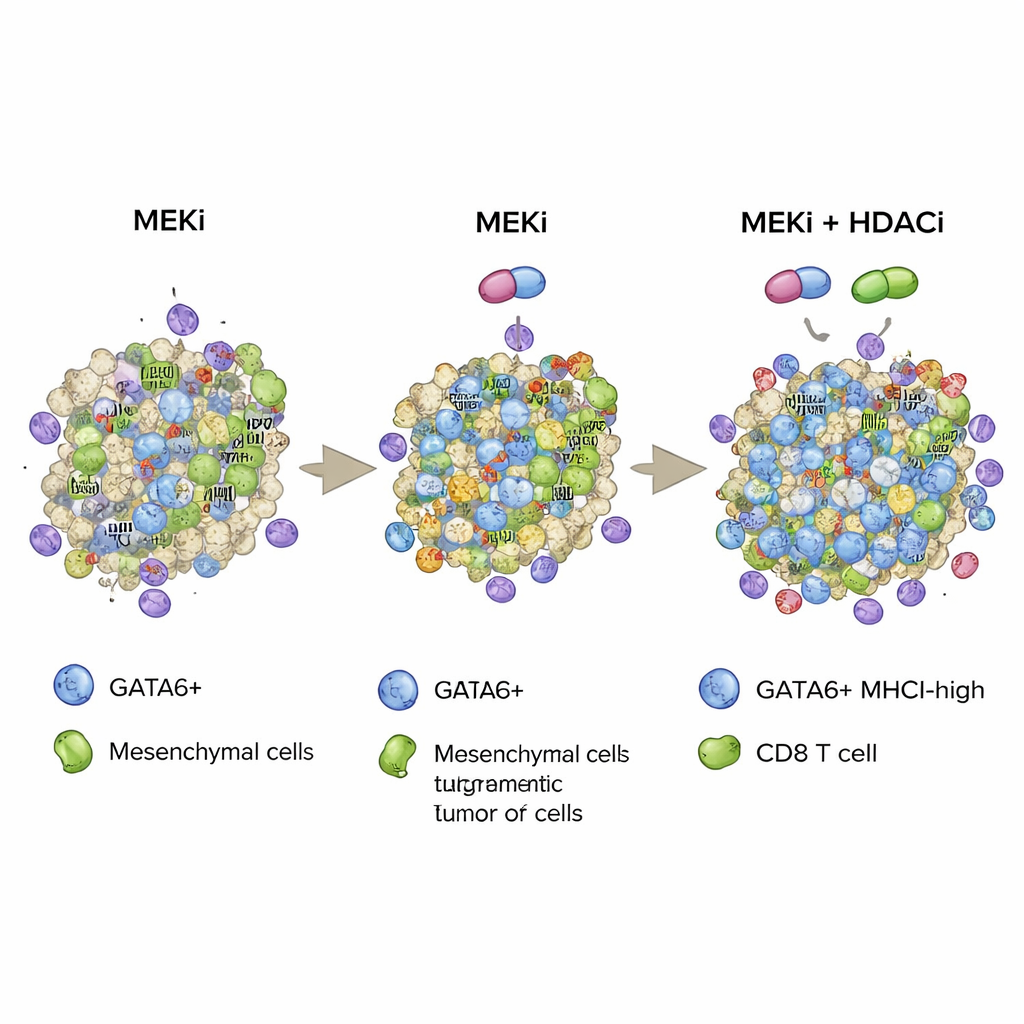
Citation: Peng, J., Yang, J., Antonopoulou, G. et al. Combined targeted and epigenetic-based therapy enhances antitumor immunity by stabilizing GATA6-dependent MHCI expression in pancreatic ductal adenocarcinoma. Nat Commun 17, 1476 (2026). https://doi.org/10.1038/s41467-026-69013-y
Keywords: pancreatic cancer, tumor immunity, GATA6, MEK inhibitor, HDAC inhibitor