Clear Sky Science · en
AACRNL evolved from virulence factor to epigenetic parasite driving genome expansion in free-living eukaryotes
When Old Toxins Learn New Tricks
Our genomes are not quiet instruction manuals. They are crowded arenas where genes, viruses, and mobile DNA constantly jostle for space and control. This study uncovers a surprising player in that struggle: a protein that began life as a toxin used by microbes to attack hosts, but has since been repurposed inside free‑living animals as a kind of "epigenetic parasite" that helps expand its own footprint in the genome.
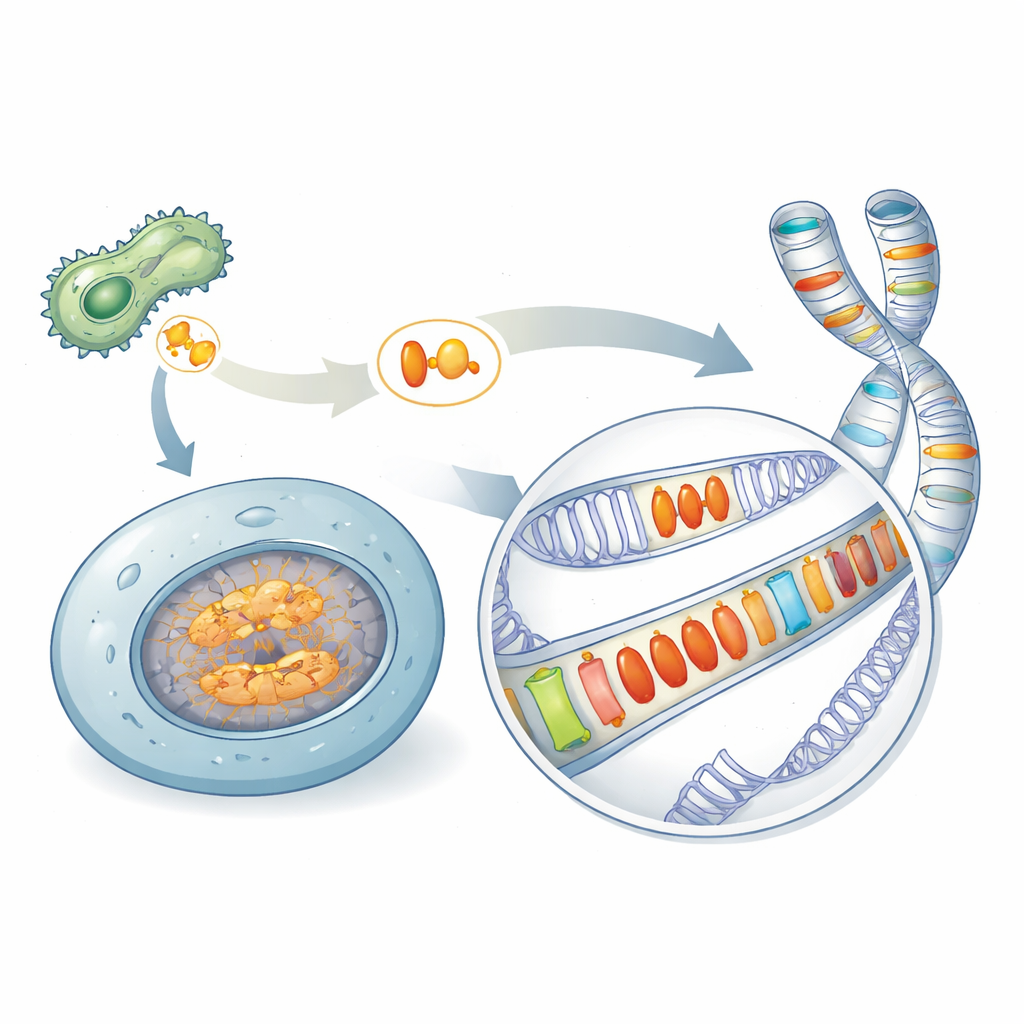
A Hidden Legacy of Microbial Weapons
Many disease‑causing microbes rely on special proteins, called effectors, to sabotage the defenses of the organisms they infect. One famous family, known as Crinklers, was thought to exist only in pathogens. By scanning a wide variety of species, the authors discovered related proteins—here grouped under the name AACRNL—in unexpected places, including sponges, corals, sea urchins, and bony fish that are not classical pathogens. These AACRNL proteins keep the toxin‑like core that can chemically modify other proteins, but their front "delivery" portion, which pathogens use to invade host cells, has largely crumbled. That structural change suggests a shift from attacking other organisms to acting within the host’s own cells.
A Selfish Switch in the Genome
In bony fish, AACRNL genes occur in multiple copies scattered across different chromosomes. By comparing neighboring stretches of DNA, the researchers showed that some AACRNL lineages have stopped copying themselves, while others have multiplied with the help of nearby jumping DNA segments called transposons. These transposons act like genomic shuttles: when they move or duplicate, they can drag AACRNL along for the ride. The result is a patchwork of repeated AACRNL copies embedded among transposon sequences, a hallmark of a gene that is spreading for its own benefit rather than for the host’s.
Breaking the Genome’s Mute Button
Ordinarily, cells keep such mobile DNA on a tight leash. One key safeguard is a chemical tag on DNA packaging proteins, known as H3K27me3, which helps keep dangerous or noisy regions switched off. The authors found that an active version of AACRNL in fish, called AACRNLβ, can chemically modify EZH2, the enzyme that lays down this repressive tag. When AACRNLβ alters EZH2, the amount of H3K27me3 at its own genetic address and at nearby transposons drops, the local DNA opens up, and both AACRNLβ and its transposon neighbors become more strongly expressed. In effect, the protein presses its own "on" button and lifts the brakes on the very mobile elements that can carry it to new spots in the genome.
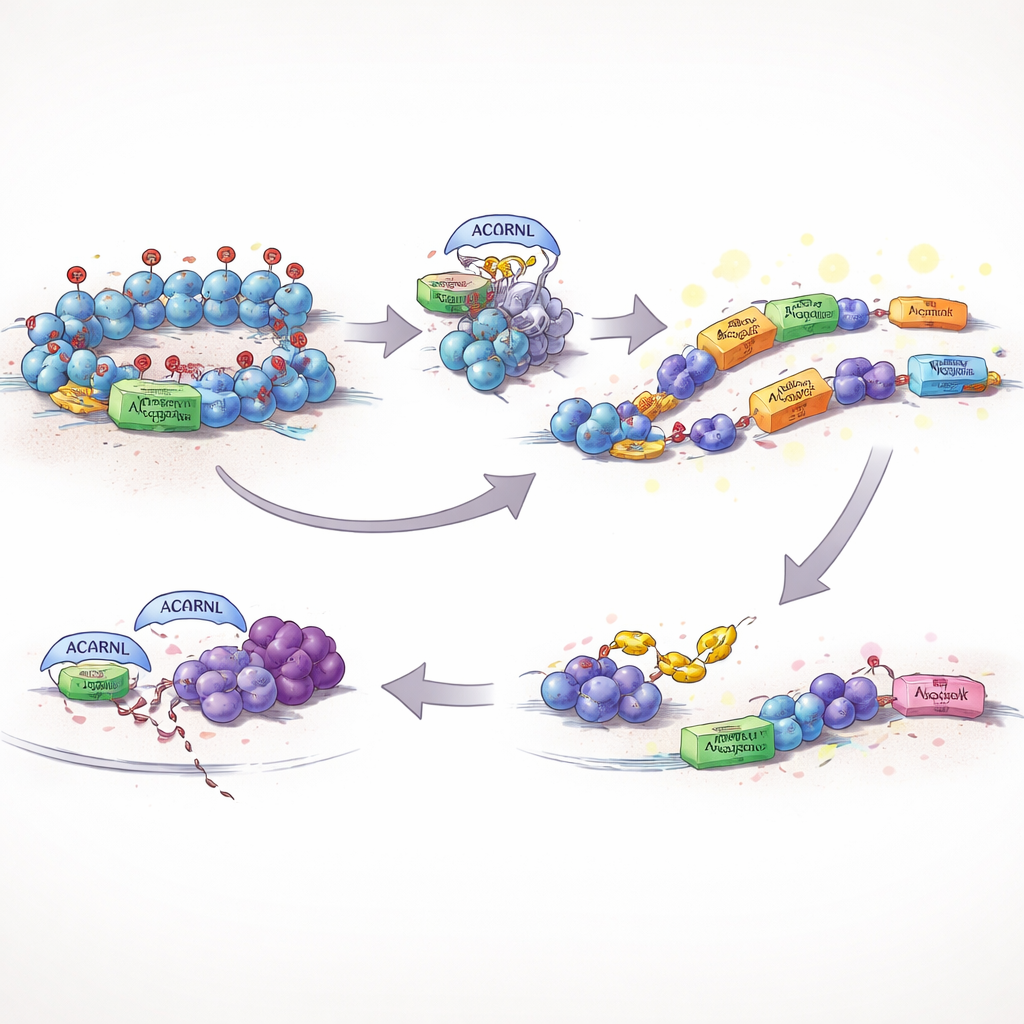
Evading the Immune Watchdogs
Unleashing transposons and unusual genetic activity risks drawing the attention of the immune system. The study shows that AACRNLβ tackles this problem too. It attaches a small chemical mark to TRAF6, a central hub protein that helps turn on antiviral and inflammatory signals. Marked in this way, TRAF6 becomes less stable and is increasingly tagged for disposal by the cell’s waste‑handling machinery. With this signal hub weakened, key defense pathways fire less strongly, creating a more tolerant environment in which AACRNLβ and its associated transposons can persist and copy themselves with fewer consequences for being detected.
A Delicate Arms Race Inside the Cell
The story does not end with AACRNLβ running unchecked. The same TRAF6 protein that is targeted by AACRNLβ can, in turn, attach its own marks to AACRNLβ, flagging the parasite‑like protein for destruction. This back‑and‑forth resembles a molecular arms race: AACRNLβ uses toxin‑derived chemistry to bend chromatin and immunity in its favor, while host proteins push back by degrading it and keeping its activity sporadic and risky. The authors argue that this tug‑of‑war has allowed a former microbial weapon to survive as a selfish resident of free‑living genomes, expanding DNA content and reshaping the rules of genetic conflict from within.
Why This Matters for Our View of Genomes
To a non‑specialist, the key message is that genomes are not just blueprints optimized for the host’s survival. They also harbor opportunists. This work shows that even classical toxins can be domesticated into such elements, using their original destructive chemistry to slip past epigenetic locks and immune alarms, and to spread through the genome over evolutionary time. Understanding this type of hidden conflict helps explain why genomes are so large, complex, and dynamic—and suggests that other "retired" toxins may be quietly rewriting DNA in many species, including perhaps our own.
Citation: Xu, T., Geng, S., Lv, X. et al. AACRNL evolved from virulence factor to epigenetic parasite driving genome expansion in free-living eukaryotes. Nat Commun 17, 2130 (2026). https://doi.org/10.1038/s41467-026-69012-z
Keywords: selfish genetic elements, genome evolution, epigenetic regulation, transposons, innate immunity